Lifshitz Theory Of Van Der Waals Force on:
[Wikipedia]
[Google]
[Amazon]
In
 The Lifshitz theory can be expressed as an effective
The Lifshitz theory can be expressed as an effective 
 Consider now, how the interaction energy will change if the right hand molecule is replaced with a medium of density of such molecules. According to the "classical" van der Waals theory, the total force will simply be the summation over individual molecules. Integrating over the volume of the medium (see the third figure), we might expect the total interaction energy with the charge to be
:
But this result cannot be correct, since It is well known that a charge in a medium of dielectric constant at a distance from the plane surface of a second medium of dielectric constant experiences a force as if there were an 'image' charge of strength at distance D on the other side of the boundary. The force between the real and image charges must then be
:
and the energy, therefore
:
Equating the two expressions for the energy, we define a new effective polarizability that must obey
:
Similarly, replacing the real charge with a medium of density and polarizability gives an expression for . Using these two relations, we may restate our theory in terms of an effective Hamaker constant. Specifically, using McLachlan's generalized theory of VDW forces the Hamaker constant for an interaction potential of the form between two bodies at temperature is
:
with , where and are Boltzmann's and Planck's constants correspondingly. Inserting our relations for and approximating the sum as an integral , the effective Hamaker constant in the Lifshitz theory may be approximated as
:
We note that are real functions, and are related to measurable properties of the medium; thus, the Hamaker constant in the Lifshitz theory can be expressed in terms of observable properties of the physical system.
Consider now, how the interaction energy will change if the right hand molecule is replaced with a medium of density of such molecules. According to the "classical" van der Waals theory, the total force will simply be the summation over individual molecules. Integrating over the volume of the medium (see the third figure), we might expect the total interaction energy with the charge to be
:
But this result cannot be correct, since It is well known that a charge in a medium of dielectric constant at a distance from the plane surface of a second medium of dielectric constant experiences a force as if there were an 'image' charge of strength at distance D on the other side of the boundary. The force between the real and image charges must then be
:
and the energy, therefore
:
Equating the two expressions for the energy, we define a new effective polarizability that must obey
:
Similarly, replacing the real charge with a medium of density and polarizability gives an expression for . Using these two relations, we may restate our theory in terms of an effective Hamaker constant. Specifically, using McLachlan's generalized theory of VDW forces the Hamaker constant for an interaction potential of the form between two bodies at temperature is
:
with , where and are Boltzmann's and Planck's constants correspondingly. Inserting our relations for and approximating the sum as an integral , the effective Hamaker constant in the Lifshitz theory may be approximated as
:
We note that are real functions, and are related to measurable properties of the medium; thus, the Hamaker constant in the Lifshitz theory can be expressed in terms of observable properties of the physical system.
condensed matter physics
Condensed matter physics is the field of physics that deals with the macroscopic and microscopic physical properties of matter, especially the solid and liquid phases which arise from electromagnetic forces between atoms. More generally, the s ...
and physical chemistry
Physical chemistry is the study of macroscopic and microscopic phenomena in chemical systems in terms of the principles, practices, and concepts of physics such as motion, energy, force, time, thermodynamics, quantum chemistry, statistical ...
, the Lifshitz theory of van der Waals forces, sometimes called the macroscopic theory of van der Waals forces, is a method proposed by Evgeny Mikhailovich Lifshitz
Evgeny Mikhailovich Lifshitz (russian: Евге́ний Миха́йлович Ли́фшиц; February 21, 1915, Kharkiv, Russian Empire – October 29, 1985, Moscow, Russian SFSR) was a leading Soviet physicist and brother of the physicist ...
in 1954 for treating van der Waals forces
In molecular physics, the van der Waals force is a distance-dependent interaction between atoms or molecules. Unlike ionic or covalent bonds, these attractions do not result from a chemical electronic bond; they are comparatively weak and t ...
between bodies which does not assume pairwise additivity of the individual intermolecular forces; that is to say, the theory takes into account the influence of neighboring molecules on the interaction between every pair of molecules located in the two bodies, rather than treating each pair independently.
Need for a non-pairwise additive theory
The van der Waals force between two molecules, in this context, is the sum of the attractive or repulsive forces between them; these forces are primarilyelectrostatic
Electrostatics is a branch of physics that studies electric charges at rest (static electricity).
Since classical times, it has been known that some materials, such as amber, attract lightweight particles after rubbing. The Greek word for am ...
in nature, and in their simplest form might consist of a force between two charges, two dipoles
In physics, a dipole () is an electromagnetic phenomenon which occurs in two ways:
*An electric dipole deals with the separation of the positive and negative electric charges found in any electromagnetic system. A simple example of this system ...
, or between a charge and a dipole. Thus, the strength of the force may often depend on the net charge, electric dipole moment, or the electric polarizability
Polarizability usually refers to the tendency of matter, when subjected to an electric field, to acquire an electric dipole moment in proportion to that applied field. It is a property of all matter, considering that matter is made up of elementar ...
() (see for example London force
London dispersion forces (LDF, also known as dispersion forces, London forces, instantaneous dipole–induced dipole forces, fluctuating induced dipole bonds or loosely as van der Waals forces) are a type of intermolecular force acting between ...
) of the molecules, with highly polarizable molecules contributing to stronger forces, and so on.
The total force between two bodies, each consisting of many molecules in the van der Waals theory is simply the sum of the intermolecular van der Waals forces, where pairwise additivity is assumed. That is to say, the forces are summed as though each pair of molecules interacts completely independently of their surroundings (See Van der Waals forces between Macroscopic Objects for an example of such a treatment). This assumption is usually correct for gasses, but presents a problem for many condensed materials, as it is known that the molecular interactions may depend strongly on their environment and neighbors. For example, in a conductor, a point-like charge might be screened by the electrons in the conductance band, and the polarizability of a condensed material may be vastly different from that of an individual molecule. In order to correctly predict the van der Waals forces of condensed materials, a theory that takes into account their total electrostatic response is needed.
General principle
The problem of pairwise additivity is completely avoided in the Lifshitz theory, where the molecular structure is ignored and the bodies are treated as continuous media. The forces between the bodies are now derived in terms of their bulk properties, such asdielectric constant
The relative permittivity (in older texts, dielectric constant) is the permittivity of a material expressed as a ratio with the electric permittivity of a vacuum. A dielectric is an insulating material, and the dielectric constant of an insulat ...
and refractive index
In optics, the refractive index (or refraction index) of an optical medium is a dimensionless number that gives the indication of the light bending ability of that medium.
The refractive index determines how much the path of light is bent, o ...
, which already contain all the necessary information from the original molecular structure.
The original Lifshitz 1955 paper proposed this method relying on quantum field theory
In theoretical physics, quantum field theory (QFT) is a theoretical framework that combines classical field theory, special relativity, and quantum mechanics. QFT is used in particle physics to construct physical models of subatomic particles a ...
principles, and is, in essence, a generalization of the Casimir effect
In quantum field theory, the Casimir effect is a physical force acting on the macroscopic boundaries of a confined space which arises from the quantum fluctuations of the field. It is named after the Dutch physicist Hendrik Casimir, who pred ...
, from two parallel, flat, ideally conducting surfaces, to two surfaces of any material. Later papers by Langbein, Ninham, Parsegian and Van Kampen showed that the essential equations could be derived using much simpler theoretical techniques, an example of which is presented here.
Hamaker constant
 The Lifshitz theory can be expressed as an effective
The Lifshitz theory can be expressed as an effective Hamaker constant
The Hamaker constant ''A'' can be defined for a van der Waals (vdW) body–body interaction:
:A=\pi^2C\rho_1\rho_2,
where \rho_1 and \rho_2 are the number densities of the two interacting kinds of particles, and ''C'' is the London coefficient in ...
in the van der Waals theory.
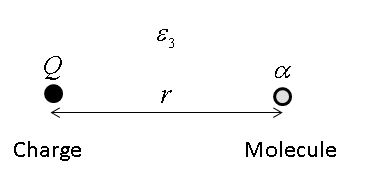
Consider, for example, the interaction between an ion of charge , and a nonpolar molecule
In chemistry, polarity is a separation of electric charge leading to a molecule or its chemical groups having an electric dipole moment, with a negatively charged end and a positively charged end.
Polar molecules must contain one or more pola ...
with polarizability at distance . In a medium with dielectric constant
The relative permittivity (in older texts, dielectric constant) is the permittivity of a material expressed as a ratio with the electric permittivity of a vacuum. A dielectric is an insulating material, and the dielectric constant of an insulat ...
, the interaction energy between a charge and an electric dipole is given by
:
with the dipole moment of the polarizable molecule given by , where is the strength of the electric field at distance from the ion. According to Coulomb's law:
:
so we may write the interaction energy as
:

 Consider now, how the interaction energy will change if the right hand molecule is replaced with a medium of density of such molecules. According to the "classical" van der Waals theory, the total force will simply be the summation over individual molecules. Integrating over the volume of the medium (see the third figure), we might expect the total interaction energy with the charge to be
:
But this result cannot be correct, since It is well known that a charge in a medium of dielectric constant at a distance from the plane surface of a second medium of dielectric constant experiences a force as if there were an 'image' charge of strength at distance D on the other side of the boundary. The force between the real and image charges must then be
:
and the energy, therefore
:
Equating the two expressions for the energy, we define a new effective polarizability that must obey
:
Similarly, replacing the real charge with a medium of density and polarizability gives an expression for . Using these two relations, we may restate our theory in terms of an effective Hamaker constant. Specifically, using McLachlan's generalized theory of VDW forces the Hamaker constant for an interaction potential of the form between two bodies at temperature is
:
with , where and are Boltzmann's and Planck's constants correspondingly. Inserting our relations for and approximating the sum as an integral , the effective Hamaker constant in the Lifshitz theory may be approximated as
:
We note that are real functions, and are related to measurable properties of the medium; thus, the Hamaker constant in the Lifshitz theory can be expressed in terms of observable properties of the physical system.
Consider now, how the interaction energy will change if the right hand molecule is replaced with a medium of density of such molecules. According to the "classical" van der Waals theory, the total force will simply be the summation over individual molecules. Integrating over the volume of the medium (see the third figure), we might expect the total interaction energy with the charge to be
:
But this result cannot be correct, since It is well known that a charge in a medium of dielectric constant at a distance from the plane surface of a second medium of dielectric constant experiences a force as if there were an 'image' charge of strength at distance D on the other side of the boundary. The force between the real and image charges must then be
:
and the energy, therefore
:
Equating the two expressions for the energy, we define a new effective polarizability that must obey
:
Similarly, replacing the real charge with a medium of density and polarizability gives an expression for . Using these two relations, we may restate our theory in terms of an effective Hamaker constant. Specifically, using McLachlan's generalized theory of VDW forces the Hamaker constant for an interaction potential of the form between two bodies at temperature is
:
with , where and are Boltzmann's and Planck's constants correspondingly. Inserting our relations for and approximating the sum as an integral , the effective Hamaker constant in the Lifshitz theory may be approximated as
:
We note that are real functions, and are related to measurable properties of the medium; thus, the Hamaker constant in the Lifshitz theory can be expressed in terms of observable properties of the physical system.
Experimental validation
The macroscopic theory of van der Waals theory has many experimental validations. Among which, some of the most notable ones are Derjaguin (1960); Derjaguin, Abrikosova and Lifshitz (1956) and Israelachvili and Tabor (1973), who measured the balance of forces between macroscopic bodies of glass, or glass and mica; Haydon and Taylor (1968), who measured the forces across bilayers by measuring their contact angle; and lastly Shih and Parsegian (1975), who investigated Van der Waals potentials between heavy alkali-metal atoms and gold surfaces using atomic-beam-deflection.References
{{Reflist Physical chemistry Condensed matter physics