Fast Atom Bombardment on:
[Wikipedia]
[Google]
[Amazon]
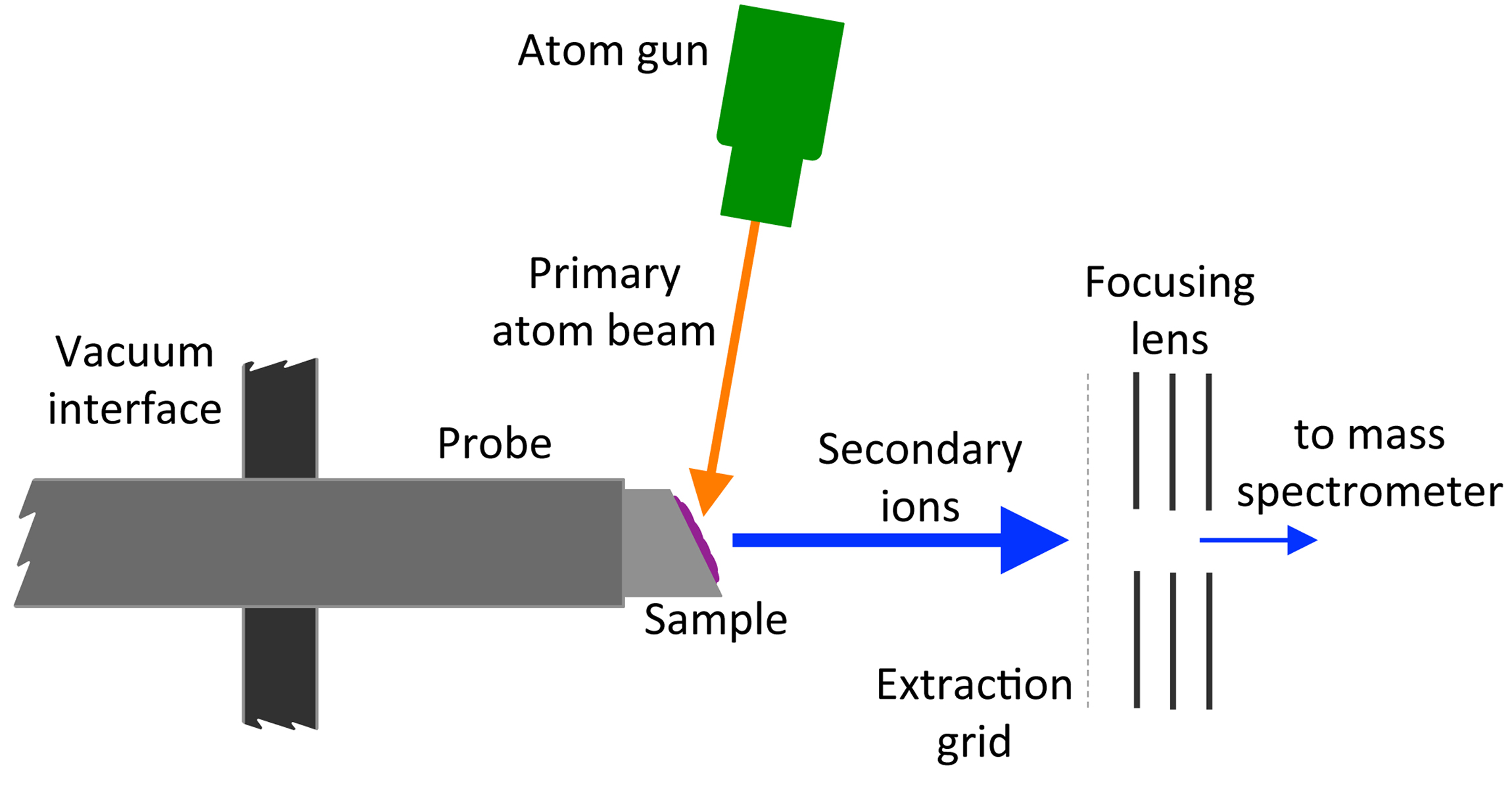 Fast atom bombardment (FAB) is an
Fast atom bombardment (FAB) is an
 FAB is a relatively low fragmentation (soft) ionization technique and produces primarily intact
FAB is a relatively low fragmentation (soft) ionization technique and produces primarily intact
 The first example of the practical application of this FAB was the elucidation of the amino acid sequence of the oligopeptide efrapeptin D. This contained a variety of very unusual amino acid residues. The sequence was shown to be: N-acetyl-L-pip-AIB-L-pip-AIB-AIB-L-leu-beta-ala-gly-AIB-AIB-L-pip-AIB-gly-L-leu-L-iva-AIB-X. PIP =
The first example of the practical application of this FAB was the elucidation of the amino acid sequence of the oligopeptide efrapeptin D. This contained a variety of very unusual amino acid residues. The sequence was shown to be: N-acetyl-L-pip-AIB-L-pip-AIB-AIB-L-leu-beta-ala-gly-AIB-AIB-L-pip-AIB-gly-L-leu-L-iva-AIB-X. PIP =
 Fast atom bombardment (FAB) is an
Fast atom bombardment (FAB) is an ionization
Ionization, or Ionisation is the process by which an atom or a molecule acquires a negative or positive charge by gaining or losing electrons, often in conjunction with other chemical changes. The resulting electrically charged atom or molecule i ...
technique used in mass spectrometry
Mass spectrometry (MS) is an analytical technique that is used to measure the mass-to-charge ratio of ions. The results are presented as a ''mass spectrum'', a plot of intensity as a function of the mass-to-charge ratio. Mass spectrometry is use ...
in which a beam of high energy atoms
Every atom is composed of a nucleus and one or more electrons bound to the nucleus. The nucleus is made of one or more protons and a number of neutrons. Only the most common variety of hydrogen has no neutrons.
Every solid, liquid, gas, an ...
strikes a surface to create ion
An ion () is an atom or molecule with a net electrical charge.
The charge of an electron is considered to be negative by convention and this charge is equal and opposite to the charge of a proton, which is considered to be positive by conve ...
s. It was developed by Michael Barber at the University of Manchester
, mottoeng = Knowledge, Wisdom, Humanity
, established = 2004 – University of Manchester Predecessor institutions: 1956 – UMIST (as university college; university 1994) 1904 – Victoria University of Manchester 1880 – Victoria Univer ...
in 1980. When a beam of high energy ions is used instead of atoms (as in secondary ion mass spectrometry
Secondary-ion mass spectrometry (SIMS) is a technique used to analyze the composition of solid surfaces and thin films by sputtering the surface of the specimen with a focused primary ion beam and collecting and analyzing ejected secondary ions. ...
), the method is known as liquid secondary ion mass spectrometry (LSIMS). In FAB and LSIMS, the material to be analyzed is mixed with a non-volatile chemical protection environment, called a matrix
Matrix most commonly refers to:
* ''The Matrix'' (franchise), an American media franchise
** ''The Matrix'', a 1999 science-fiction action film
** "The Matrix", a fictional setting, a virtual reality environment, within ''The Matrix'' (franchis ...
, and is bombarded under vacuum with a high energy (4000 to 10,000 electron volts
In physics, an electronvolt (symbol eV, also written electron-volt and electron volt) is the measure of an amount of kinetic energy gained by a single electron accelerating from rest through an electric potential difference of one volt in vacuum. ...
) beam of atoms. The atoms are typically from an inert gas such as argon
Argon is a chemical element with the symbol Ar and atomic number 18. It is in group 18 of the periodic table and is a noble gas. Argon is the third-most abundant gas in Earth's atmosphere, at 0.934% (9340 ppmv). It is more than twice as abu ...
or xenon
Xenon is a chemical element with the symbol Xe and atomic number 54. It is a dense, colorless, odorless noble gas found in Earth's atmosphere in trace amounts. Although generally unreactive, it can undergo a few chemical reactions such as the ...
. Common matrices include glycerol
Glycerol (), also called glycerine in British English and glycerin in American English, is a simple triol compound. It is a colorless, odorless, viscous liquid that is sweet-tasting and non-toxic. The glycerol backbone is found in lipids known ...
, thioglycerol, 3-nitrobenzyl alcohol (3-NBA), 18-crown-6
18-Crown-6 is an organic compound with the formula 2H4O and the IUPAC name of 1,4,7,10,13,16-hexaoxacyclooctadecane. It is a white, hygroscopic crystalline solid with a low melting point. Like other crown ethers, 18-crown-6 functions as a li ...
ether, 2-nitrophenyloctyl ether, sulfolane
Sulfolane (also ''tetramethylene sulfone'', systematic name: 1λ6-thiolane-1,1-dione) is an organosulfur compound, formally a cyclic sulfone, with the formula (CH2)4SO2. It is a colorless liquid commonly used in the chemical industry as a solvent ...
, diethanolamine
Diethanolamine, often abbreviated as DEA or DEOA, is an organic compound with the formula HN(CH2CH2OH)2. Pure diethanolamine is a white solid at room temperature, but its tendencies to absorb water and to supercool meaning that it is often encou ...
, and triethanolamine
Triethanolamine, or TEA is a viscous organic compound that is both a tertiary amine and a triol. A triol is a molecule with three alcohol groups. Approximately 150,000 tonnes were produced in 1999. It is a colourless compound although samples m ...
. This technique is similar to secondary ion mass spectrometry
Secondary-ion mass spectrometry (SIMS) is a technique used to analyze the composition of solid surfaces and thin films by sputtering the surface of the specimen with a focused primary ion beam and collecting and analyzing ejected secondary ions. ...
and plasma desorption mass spectrometry
An ion source is a device that creates atomic and molecular ions. Ion sources are used to form ions for mass spectrometers, optical emission spectrometers, particle accelerators, ion implanters and ion engines.
Electron ionization
Electron ...
.
Ionization mechanism
 FAB is a relatively low fragmentation (soft) ionization technique and produces primarily intact
FAB is a relatively low fragmentation (soft) ionization technique and produces primarily intact protonated molecule
In chemistry, protonation (or hydronation) is the adding of a proton (or hydron, or hydrogen cation), (H+) to an atom, molecule, or ion, forming a conjugate acid. (The complementary process, when a proton is removed from a Brønsted–Lowry acid, ...
s denoted as + Hsup>+ and deprotonated molecules such as - Hsup>−. Radical cations can also be observed in a FAB spectrum in rare cases. FAB was designed as an improved version of SIMS that allowed for the primary beam to no longer cause damaging effects to the sample. The major difference between the two techniques is the difference in the nature of the primary beam used; ions vs atoms. For LSIMS, Cesium
Caesium (IUPAC spelling) (or cesium in American English) is a chemical element with the symbol Cs and atomic number 55. It is a soft, silvery-golden alkali metal with a melting point of , which makes it one of only five elemental metals that ar ...
, Cs+ ions make up the primary beam and for FAB the primary beam is made up of Xe or Ar atoms. Xe atoms are used because they tend to be more sensitive than Argon atoms due to their larger masses and more momentum. For the molecules to be ionized by FAB, first the slow moving atoms (Xe or Ar) are ionized by colliding electrons. Those slow moving atoms are then ionized and accelerated to a certain potential where they develop into fast moving ions that become neutral in a dense cloud of excess natural gas atoms that make a flowing stream of high translational energy atoms. Although the exact mechanism of how the samples are ionized have not been fully discovered, the nature of its ionization mechanism is similar to matrix-assisted laser desorption/ionization
In mass spectrometry, matrix-assisted laser desorption/ionization (MALDI) is an ionization technique that uses a laser energy absorbing matrix to create ions from large molecules with minimal fragmentation. It has been applied to the analysis of ...
(MALDI) and chemical ionization
Chemical ionization (CI) is a soft ionization technique used in mass spectrometry. This was first introduced by Burnaby Munson and Frank H. Field in 1966. This technique is a branch of gaseous ion-molecule chemistry. Reagent gas molecules (often ...
.
Matrices and sample introduction
As previously stated, in FAB the samples are mixed with a non-volatile environment (matrix
Matrix most commonly refers to:
* ''The Matrix'' (franchise), an American media franchise
** ''The Matrix'', a 1999 science-fiction action film
** "The Matrix", a fictional setting, a virtual reality environment, within ''The Matrix'' (franchis ...
) in order to be analyzed. FAB uses a liquid matrix that is mixed with the sample in order to provide a sample ion current that is sustained, reduces damages made to the sample by absorbing the impact of the primary beam, and keeps the sample molecules form aggregating. The liquid matrix, like any other matrix, most importantly provides a medium that promotes sample ionization. The most widely accepted matrix for this type of ionization is glycerol
Glycerol (), also called glycerine in British English and glycerin in American English, is a simple triol compound. It is a colorless, odorless, viscous liquid that is sweet-tasting and non-toxic. The glycerol backbone is found in lipids known ...
. Choosing the appropriate matrix for the sample is crucial because the matrix can also influence the degree of fragmentation of the sample (analyte) ions.
The sample can then be introduced to FAB analysis. The normal method of introducing the sample-matrix mixture is through an insertion probe. The sample-matrix mixture is loaded on a stainless steel sample target on the probe, which is then placed in the ion source via a vacuum lock. The alternative method of introducing the sample is by using a device called continuous flow fast atom bombardment (CF)-FAB.
Continuous flow fast atom bombardment
In continuous flow fast atom bombardment (CF-FAB), the sample is introduced into the mass spectrometer insertion probe through a small diameter capillary. (CF)-FAB was developed to minimize the problem of poor detection sensitivity that is caused by an excess of the matrix background that results in a high matrix-to-sample ratio. When a metal frit is used to disperse the liquid on the probe, the technique is known as frit FAB. Samples can be introduced by flow injection, microdialysis, or by coupling with liquid chromatography. Flow rates are typically between 1 and 20 μL/min. CF-FAB has a higher sensitivity compared to static FABApplications
 The first example of the practical application of this FAB was the elucidation of the amino acid sequence of the oligopeptide efrapeptin D. This contained a variety of very unusual amino acid residues. The sequence was shown to be: N-acetyl-L-pip-AIB-L-pip-AIB-AIB-L-leu-beta-ala-gly-AIB-AIB-L-pip-AIB-gly-L-leu-L-iva-AIB-X. PIP =
The first example of the practical application of this FAB was the elucidation of the amino acid sequence of the oligopeptide efrapeptin D. This contained a variety of very unusual amino acid residues. The sequence was shown to be: N-acetyl-L-pip-AIB-L-pip-AIB-AIB-L-leu-beta-ala-gly-AIB-AIB-L-pip-AIB-gly-L-leu-L-iva-AIB-X. PIP = pipecolic acid
Pipecolic acid (piperidine-2-carboxylic acid) is an organic compound with the formula HNC5H9CO2H. It is a carboxylic acid derivative of piperidine and, as such, an amino acid, although not one encoded genetically. Like many other α-amino acids, p ...
, AIB = alpha-amino-isobutyric acid
2-Aminoisobutyric acid (also known as α-aminoisobutyric acid, AIB, α-methylalanine, or 2-methylalanine) is the non-proteinogenic amino acid with the structural formula H2N-C(CH3)2-COOH. It is rare in nature, having been only found in meteorites ...
, leu = leucine
Leucine (symbol Leu or L) is an essential amino acid that is used in the biosynthesis of proteins. Leucine is an α-amino acid, meaning it contains an α-amino group (which is in the protonated −NH3+ form under biological conditions), an α- ca ...
, iva = isovaline, gly = glycine
Glycine (symbol Gly or G; ) is an amino acid that has a single hydrogen atom as its side chain. It is the simplest stable amino acid (carbamic acid is unstable), with the chemical formula NH2‐ CH2‐ COOH. Glycine is one of the proteinogeni ...
. This is a potent inhibitor of mitochondrial
A mitochondrion (; ) is an organelle found in the cells of most Eukaryotes, such as animals, plants and fungi. Mitochondria have a double membrane structure and use aerobic respiration to generate adenosine triphosphate (ATP), which is use ...
ATPase activity. Another application of FAB includes its original use for the analysis of condensed-phase samples. FAB can be use for measurements of the molecular weight of samples below 5000 Da, as well as their structural characteristics. FAB can be paired with various mass spectrometers for data analysis, such as with a quadrupole mass analyzer
The quadrupole mass analyzer, originally conceived by Nobel Laureate Wolfgang Paul and his student Helmut Steinwedel, also known as quadrupole mass filter, is one type of mass analyzer used in mass spectrometry. As the name implies, it consists of ...
, liquid chromatography–mass spectrometry
Liquid chromatography–mass spectrometry (LC–MS) is an analytical chemistry technique that combines the physical separation capabilities of liquid chromatography (or HPLC) with the mass analysis capabilities of mass spectrometry (MS). Coupled ...
, and more.
Inorganic analysis
In 1983 a paper was published describing the use of fast atom bombardment mass spectrometry (FAB-MS) to analyze isotopes of calcium. Glycerol was not used; samples in aqueous solution were deposited on the sample target and dried prior to analysis. The technique was effectivelysecondary ion mass spectrometry
Secondary-ion mass spectrometry (SIMS) is a technique used to analyze the composition of solid surfaces and thin films by sputtering the surface of the specimen with a focused primary ion beam and collecting and analyzing ejected secondary ions. ...
using a neutral primary beam. This was a welcomed development for biomedical researchers studying the nutrition and metabolism of essential minerals but lacking access to inorganic mass spectrometry instrumentation such as thermal ionization mass spectrometry
Thermal ionization mass spectrometry (TIMS) is also known as surface ionization and is a highly sensitive isotope mass spectrometry characterization technique. The isotopic ratios of radionuclides are used to get an accurate measurement for the ele ...
or inductively-coupled plasma mass spectrometry (ICP-MS). In contrast, FAB mass spectrometers were widely found in biomedical research institutions. Multiple laboratories adopted this technique, using FAB-MS to measure isotope ratios in isotope tracer studies of calcium, iron, magnesium and zinc. The analysis of metals required minimal modification of the mass spectrometers, e.g.replacing the stainless steel sample targets with pure silver ones to eliminate background from ionization of stainless steel components. Signal acquisition systems were sometimes modified to perform peak jumping instead of scanning and to do ion counting detection. While satisfactory precision and accuracy were attained with FAB-MS, the technique was labor-intensive with a very low sample through-put rate due in part to the absence of auto-sampling options. By the early 2000's this severe sampling rate limitation had motivated users of FAB-MS for mineral isotope analysis to switch to conventional inorganic mass spectrometers, usually ICP-MS which also exhibited improved affordability and isotope ratio analysis performance by that time.
References
{{Mass spectrometry Ion source