End-group on:
[Wikipedia]
[Google]
[Amazon]
End groups are an important aspect of
/ref> In polymer synthesis, like


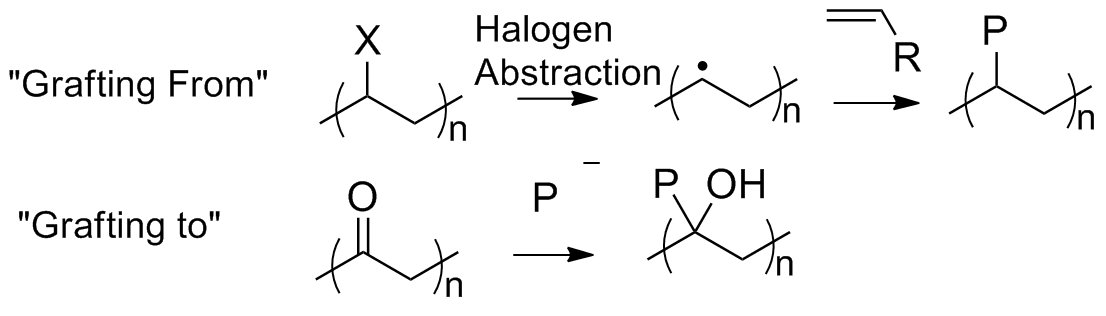 The "grafting from" technique involves the generation of radicals along the polymer backbone from an abstraction of a halogen, from either the backbone or a functional group along the backbone. Monomers are reacted with the radicals along the backbone and subsequently generate polymers which are grafted from the backbone of the first polymer. The schematic for "grafting to" shows an example using anionic polymerizations, the polymer containing the carbonyl functionalities gets attacked by the activated polymer chain and generates a polymer attached to the associated carbon along with an alcohol group, in this example. These examples show us the potential of fine tuning end groups of polymer chains to target certain copolymer structures.
The "grafting from" technique involves the generation of radicals along the polymer backbone from an abstraction of a halogen, from either the backbone or a functional group along the backbone. Monomers are reacted with the radicals along the backbone and subsequently generate polymers which are grafted from the backbone of the first polymer. The schematic for "grafting to" shows an example using anionic polymerizations, the polymer containing the carbonyl functionalities gets attacked by the activated polymer chain and generates a polymer attached to the associated carbon along with an alcohol group, in this example. These examples show us the potential of fine tuning end groups of polymer chains to target certain copolymer structures.

 The method for removal of thiocarbonyl containing end groups includes reacting the polymers containing the end-groups with en excess of radicals which add to the reactive C=S bond of the end group forming an intermediate
The method for removal of thiocarbonyl containing end groups includes reacting the polymers containing the end-groups with en excess of radicals which add to the reactive C=S bond of the end group forming an intermediate  Another method of end group removal for the thiocarbonyl containing end-groups of RAFT polymers is the addition of heat to the polymer; this is referred to as
Another method of end group removal for the thiocarbonyl containing end-groups of RAFT polymers is the addition of heat to the polymer; this is referred to as
polymer
A polymer (; Greek '' poly-'', "many" + ''-mer'', "part")
is a substance or material consisting of very large molecules called macromolecules, composed of many repeating subunits. Due to their broad spectrum of properties, both synthetic a ...
synthesis and characterization. In polymer chemistry
Polymer chemistry is a sub-discipline of chemistry that focuses on the structures of chemicals, chemical synthesis, and chemical and physical properties of polymers and macromolecules. The principles and methods used within polymer chemistry are ...
, they are functional groups
In organic chemistry, a functional group is a substituent or moiety in a molecule that causes the molecule's characteristic chemical reactions. The same functional group will undergo the same or similar chemical reactions regardless of the rest ...
that are at the very ends of a macromolecule
A macromolecule is a very large molecule important to biophysical processes, such as a protein or nucleic acid. It is composed of thousands of covalently bonded atoms. Many macromolecules are polymers of smaller molecules called monomers. The ...
or oligomer
In chemistry and biochemistry, an oligomer () is a molecule that consists of a few repeating units which could be derived, actually or conceptually, from smaller molecules, monomers.Quote: ''Oligomer molecule: A molecule of intermediate relativ ...
(IUPAC
The International Union of Pure and Applied Chemistry (IUPAC ) is an international federation of National Adhering Organizations working for the advancement of the chemical sciences, especially by developing nomenclature and terminology. It is ...
).IUPAC Gold Book, "end-group"/ref> In polymer synthesis, like
condensation polymerization
In polymer chemistry, condensation polymers are any kind of polymers whose process of polymerization involves a condensation reaction (i.e. a small molecule, such as water or methanol, is produced as a byproduct). Condensation polymers are form ...
and free-radical
In chemistry, a radical, also known as a free radical, is an atom, molecule, or ion that has at least one unpaired valence electron.
With some exceptions, these unpaired electrons make radicals highly chemically reactive. Many radicals spon ...
types of polymerization, end-groups are commonly used and can be analyzed by nuclear magnetic resonance (NMR
Nuclear magnetic resonance (NMR) is a physical phenomenon in which nuclei in a strong constant magnetic field are perturbed by a weak oscillating magnetic field (in the near field) and respond by producing an electromagnetic signal with ...
) to determine the average length of the polymer. Other methods for characterization of polymers where end-groups are used are mass spectrometry
Mass spectrometry (MS) is an analytical technique that is used to measure the mass-to-charge ratio of ions. The results are presented as a ''mass spectrum'', a plot of intensity as a function of the mass-to-charge ratio. Mass spectrometry is use ...
and vibrational spectrometry, like infrared
Infrared (IR), sometimes called infrared light, is electromagnetic radiation (EMR) with wavelengths longer than those of visible light. It is therefore invisible to the human eye. IR is generally understood to encompass wavelengths from around ...
and raman spectroscopy. These groups are important for the analysis of polymers and for grafting to and from a polymer chain to create a new copolymer
In polymer chemistry, a copolymer is a polymer derived from more than one species of monomer. The polymerization of monomers into copolymers is called copolymerization. Copolymers obtained from the copolymerization of two monomer species are some ...
. One example of an end group is in the polymer poly(ethylene glycol) diacrylate where the end-groups are circled.

End groups in polymer synthesis
End groups are seen on all polymers and the functionality of those end groups can be important in determining the application of polymers. Each type of polymerization (free radical, condensation or etc.) has end groups that are typical for the polymerization, and knowledge of these can help to identify the type of polymerization method used to form the polymer.Step-growth polymerization
Step-growth polymerization
Step-growth polymerization refers to a type of polymerization mechanism in which bi-functional or multifunctional monomers react to form first dimers, then trimers, longer oligomers and eventually long chain polymers. Many naturally occurring a ...
involves two monomers with bi- or multifunctionality to form polymer chains. Many polymers are synthesized via step-growth polymerization and include polyesters
Polyester is a category of polymers that contain the ester functional group in every repeat unit of their main chain. As a specific material, it most commonly refers to a type called polyethylene terephthalate (PET). Polyesters include natural ...
, polyamides
A polyamide is a polymer with repeating units linked by amide bonds.
Polyamides occur both naturally and artificially. Examples of naturally occurring polyamides are proteins, such as wool and silk. Artificially made polyamides can be made throug ...
, and polyurethanes
Polyurethane (; often abbreviated PUR and PU) refers to a class of polymers composed of organic units joined by carbamate (urethane) links. In contrast to other common polymers such as polyethylene and polystyrene, polyurethane is produced fro ...
. A sub-class of step-growth polymerization is condensation polymerization.
Condensation polymerization
Condensation polymerization is an important class of step-growth polymerization, which is formed simply by the reaction of two monomers and results in the release of a water molecule. Since these polymers are typically made up of two or more monomers, the resulting end groups are from the monomer functionality. Examples of condensation polymers can be seen with polyamides, polyacetals and polyesters. An example of polyester ispolyethylene terephthalate
Polyethylene terephthalate (or poly(ethylene terephthalate), PET, PETE, or the obsolete PETP or PET-P), is the most common thermoplastic polymer resin of the polyester family and is used in fibres for clothing, containers for liquids and foods ...
(PET), which is made from the monomers terephthalic acid
Terephthalic acid is an organic compound with formula C6H4(CO2H)2. This white solid is a commodity chemical, used principally as a precursor to the polyester PET, used to make clothing and plastic bottles. Several million tonnes are produced annua ...
and ethylene glycol
Ethylene glycol (IUPAC name: ethane-1,2-diol) is an organic compound (a vicinal diol) with the formula . It is mainly used for two purposes, as a raw material in the manufacture of polyester fibers and for antifreeze formulations. It is an odo ...
. If one of the components in the polymerization is in excess, then that polymers functionality will be at the ends of the polymers (a carboxylic acid or alcohol group respectively).

Free radical polymerization
The end groups that are found on polymers formed through free radical polymerization are a result from the initiators and termination method used. There are many types of initiators used in modern free radical polymerizations, and below are examples of some well-known ones. For example,azobisisobutyronitrile
Azobisisobutyronitrile (abbreviated AIBN) is an organic compound with the formula CH3)2C(CN)sub>2N2. This white powder is soluble in alcohols and common organic solvents but is insoluble in water. It is often used as a foamer in plastics and rubb ...
or AIBN forms radicals that can be used as the end groups for new starting polymer chains with styrene to form polystyrene. Once the polymer chain has formed and the reaction is terminated, the end group opposite from the initiator is a result of the terminating agent or the chain transfer agent used.

End groups in graft polymers
Graft copolymers are generated by attaching chains of one monomer to the main chain of another polymer; a branched block copolymer is formed. Furthermore, end groups play an important role in the process of initiation, propagation and termination of graft polymers. Graft polymers can be achieved by either "grafting from" or "grafting to"; these different methods are able to produce a vast array of different polymer structures, which can be tailored to the application in question. The "grafting from" approach involves, for example, generation of radicals along a polymer chain, which can then be reacted with monomers to grow a new polymer from the backbone of another. In "grafting from," the initiation sites on the backbone of the first polymer can be part of the backbone structure originally or generated in situ. The "grafting to" approach involves the reaction of functionalized monomers to a polymer backbone. In graft polymers, end groups play an important role, for example, in the "grafting to" technique the generation of the reactive functionalized monomers occurs at the end group, which is then tethered to the polymer chain. There are various methods to synthesize graft polymers some of the more common includeredox reaction
Redox (reduction–oxidation, , ) is a type of chemical reaction in which the oxidation states of substrate change. Oxidation is the loss of electrons or an increase in the oxidation state, while reduction is the gain of electrons or a d ...
to produce free radicals
In chemistry, a radical, also known as a free radical, is an atom, molecule, or ion that has at least one unpaired valence electron.
With some exceptions, these unpaired electrons make radicals highly chemically reactive. Many radicals spont ...
, by free radical polymerization
In polymer chemistry, free-radical polymerization (FRP) is a method of polymerization by which a polymer forms by the successive addition of free-radical building blocks (repeat units). Free radicals can be formed by a number of different mechanis ...
techniques avoiding chain termination
Chain termination is any chemical reaction that ceases the formation of reactive intermediates in a chain propagation step in the course of a polymerization, effectively bringing it to a halt.
Mechanisms of termination
In polymer chemistry, ...
(ATRP, RAFT
A raft is any flat structure for support or transportation over water. It is usually of basic design, characterized by the absence of a hull. Rafts are usually kept afloat by using any combination of buoyant materials such as wood, sealed barrel ...
, nitroxide mediated, for example) and step-growth polymerization
Step-growth polymerization refers to a type of polymerization mechanism in which bi-functional or multifunctional monomers react to form first dimers, then trimers, longer oligomers and eventually long chain polymers. Many naturally occurring a ...
. A schematic of "grafting from" and "grafting to" is illustrated in the figure below.
 The "grafting from" technique involves the generation of radicals along the polymer backbone from an abstraction of a halogen, from either the backbone or a functional group along the backbone. Monomers are reacted with the radicals along the backbone and subsequently generate polymers which are grafted from the backbone of the first polymer. The schematic for "grafting to" shows an example using anionic polymerizations, the polymer containing the carbonyl functionalities gets attacked by the activated polymer chain and generates a polymer attached to the associated carbon along with an alcohol group, in this example. These examples show us the potential of fine tuning end groups of polymer chains to target certain copolymer structures.
The "grafting from" technique involves the generation of radicals along the polymer backbone from an abstraction of a halogen, from either the backbone or a functional group along the backbone. Monomers are reacted with the radicals along the backbone and subsequently generate polymers which are grafted from the backbone of the first polymer. The schematic for "grafting to" shows an example using anionic polymerizations, the polymer containing the carbonyl functionalities gets attacked by the activated polymer chain and generates a polymer attached to the associated carbon along with an alcohol group, in this example. These examples show us the potential of fine tuning end groups of polymer chains to target certain copolymer structures.
Analysis of polymers using end groups
Because of the importance of end groups, there have been many analytical techniques developed for the identification of the groups. The three main methods for analyzing the identity of the end group are byNMR
Nuclear magnetic resonance (NMR) is a physical phenomenon in which nuclei in a strong constant magnetic field are perturbed by a weak oscillating magnetic field (in the near field) and respond by producing an electromagnetic signal with ...
, mass spectrometry
Mass spectrometry (MS) is an analytical technique that is used to measure the mass-to-charge ratio of ions. The results are presented as a ''mass spectrum'', a plot of intensity as a function of the mass-to-charge ratio. Mass spectrometry is use ...
(MS) or vibrational spectroscopy ( IR or Raman). Each technique has its advantages and disadvantages, which are details below.
NMR spectroscopy
The advantage ofNMR
Nuclear magnetic resonance (NMR) is a physical phenomenon in which nuclei in a strong constant magnetic field are perturbed by a weak oscillating magnetic field (in the near field) and respond by producing an electromagnetic signal with ...
for end groups is that it allows for not only the identification of the end group units, but also allows for the quantification of the number-average length of the polymer. End-group analysis with NMR
Nuclear magnetic resonance (NMR) is a physical phenomenon in which nuclei in a strong constant magnetic field are perturbed by a weak oscillating magnetic field (in the near field) and respond by producing an electromagnetic signal with ...
requires that the polymer be soluble in organic or aqueous solvents. Additionally, the signal on the end-group must be visible as a distinct spectral frequency, i.e. it must not overlap with other signals. As molecular weight increases, the width of the spectral peaks also increase. As a result of this, methods which rely on resolution of the end-group signal are mostly used for polymers of low molecular weight (roughly less than 20,000 g/mol number-average molecular weight). By using the information obtained from the integration of a 1H NMR spectrum, the degree of polymerization
The degree of polymerization, or DP, is the number of monomeric units in a macromolecule or polymer or oligomer molecule.
For a homopolymer, there is only one type of monomeric unit and the ''number-average'' degree of polymerization is given by ...
(Xn) can be calculated. With knowledge of the identity of the end groups/repeat unit and the number of protons contained on each, the Xn can then be calculated. For this example above, once the 1H NMR has been integrated and the values have been normalized to 1, the degree of polymerization is calculated by simply dividing the normalized value for the repeat unit by the number of protons continued in the repeat unit. For this case, Xn = n = 100/2, and therefore Xn = 50, or there are 50 repeat units in this monomer.

Mass spectrometry
Mass spectrometry (MS) is helpful for the determination of the molecular weight of the polymer, structure of the polymer, etc. Although chemists utilize many kinds of MS, the two that are used most typically are matrix-assisted laser desorption ionization/time of flight (MALDI-TOF) and electrospray ionization-mass spectroscopy (ESI-MS). One of the biggest disadvantages of this technique is that much like NMR spectroscopy the polymers have to be soluble in some organic solvent. An advantage of using MALDI is that it provides the simpler data to interpret for end group identification compared with ESI, but a disadvantage is that the ionization can be rather hard and as a result some end groups do not remain intact for analysis. Because of the harsh ionization in MALDI, one of the biggest advantages of using ESI is for its "softer" ionization methods. The disadvantage of using ESI is that the data obtained can be very complex due to the mechanism of the ionization and thus can be difficult to interpret.Vibrational spectroscopy
The vibrational spectroscopy methods used to analyze the end groups of a polymer areinfrared
Infrared (IR), sometimes called infrared light, is electromagnetic radiation (EMR) with wavelengths longer than those of visible light. It is therefore invisible to the human eye. IR is generally understood to encompass wavelengths from around ...
(IR) and Raman spectroscopy
Raman spectroscopy () (named after Indian physicist C. V. Raman) is a spectroscopic technique typically used to determine vibrational modes of molecules, although rotational and other low-frequency modes of systems may also be observed. Raman sp ...
. These methods are useful in fact that the polymers do not need to be soluble in a solvent and spectra can be obtained simply from solid material. A disadvantage of the technique is that only qualitative data is typically obtained on the identification end groups.
End group removal
Controlled radical polymerization
Living free radical polymerization is a type of living polymerization where the active polymer chain end is a free radical. Several methods exist. IUPAC recommends to use the term "reversible-deactivation radical polymerization" instead of "livi ...
, namely reversible addition−fragmentation chain-transfer polymerization
Reversible addition−fragmentation chain-transfer or RAFT polymerization is one of several kinds of reversible-deactivation radical polymerization. It makes use of a chain-transfer agent in the form of a thiocarbonylthio compound (or similar, fr ...
(RAFT), is a common method for the polymerization of acrylate
Acrylates (IUPAC: prop-2-enoates) are the salts, esters, and conjugate bases of acrylic acid. The acrylate ion is the anion C H2=CHC OO−. Often, acrylate refers to esters of acrylic acid, the most common member being methyl acrylate. These acry ...
s, methacrylate Methacrylates are derivatives of methacrylic acid.
* Methyl methacrylate
* Ethyl methacrylate
* Butyl methacrylate
* Hydroxyethyl methacrylate
* Glycidyl methacrylate
Glycidyl methacrylate (GMA) is an ester of methacrylic acid and glycidol. Co ...
s and acrylamide
Acrylamide (or acrylic amide) is an organic compound with the chemical formula CH2=CHC(O)NH2. It is a white odorless solid, soluble in water and several organic solvents. From the chemistry perspective, acrylamide is a vinyl-substituted primary ...
s. Usually, a thiocarbonate Thiocarbonate describes a family of anions with the general chemical formula (''x'' = 0, 1, or 2):
*for ''x'' = 2 it is monothiocarbonate ion
*for ''x'' = 1 it is dithiocarbonate ion
*for ''x'' = 0 it is trithiocarbonate ion
Like the carbonate d ...
is used in combination with an effective initiator for RAFT. The thiocarbonate moiety can be functionalized at the R-group for end group analysis. The end group is a result of the propagation of chain-transfer agents during the free-radical polymerization process. The end groups can subsequently be modified by the reaction of the thiocarbonylthio compounds with nucleophiles and ionic reducing agents.
 The method for removal of thiocarbonyl containing end groups includes reacting the polymers containing the end-groups with en excess of radicals which add to the reactive C=S bond of the end group forming an intermediate
The method for removal of thiocarbonyl containing end groups includes reacting the polymers containing the end-groups with en excess of radicals which add to the reactive C=S bond of the end group forming an intermediate radical
Radical may refer to:
Politics and ideology Politics
*Radical politics, the political intent of fundamental societal change
*Radicalism (historical), the Radical Movement that began in late 18th century Britain and spread to continental Europe and ...
(shown below). The remaining radical on the polymer chain can be hydrogenated
Hydrogenation is a chemical reaction between molecular hydrogen (H2) and another compound or element, usually in the presence of a catalyst such as nickel, palladium or platinum. The process is commonly employed to reduce or saturate organic co ...
by what is referred to as a trapping group and terminate; this results in a polymer that is free of the end groups at the α and ω positions.
 Another method of end group removal for the thiocarbonyl containing end-groups of RAFT polymers is the addition of heat to the polymer; this is referred to as
Another method of end group removal for the thiocarbonyl containing end-groups of RAFT polymers is the addition of heat to the polymer; this is referred to as thermolysis
Thermal decomposition, or thermolysis, is a chemical decomposition caused by heat. The decomposition temperature of a substance is the temperature at which the substance chemically decomposes. The reaction is usually endothermic as heat is req ...
. One method of monitoring thermolysis of RAFT polymers is by thermogravimetric analysis resulting in a weight-loss of the end group. An advantage of this technique is that no additional chemicals are required to remove the end group; however, it is required that the polymer be thermally stable to high temperature and therefore may not be effective for some polymers. Depending on the polymers sensitivity to ultraviolet radiation (UV) it has been reported in recent years that decomposition of end-groups can be effective, but preliminary data suggest that decomposition by UV leads to a change in the distribution of molecular weights of the polymer.
Surface modification using RAFT
Surface modification has gained a lot of interest in recent years for a variety of applications. An example of the application of free radical polymerizations to forming new architectures is throughRAFT
A raft is any flat structure for support or transportation over water. It is usually of basic design, characterized by the absence of a hull. Rafts are usually kept afloat by using any combination of buoyant materials such as wood, sealed barrel ...
polymerizations which result in dithioester end groups. These dithioesters can be reduced to the thiol which can be immobilized on a metal surface; this is important for applications in electronics, sensing and catalysis. The schematic below demonstrates the immobilization of copolymers onto a gold surface as reported for poly(sodium 4-styrenesulfonate) by the McCormick group at the University of Southern Mississippi.
References
{{Reflist Polymer chemistry