Disjoining Pressure on:
[Wikipedia]
[Google]
[Amazon]
In
(Google books)
/ref> : where (in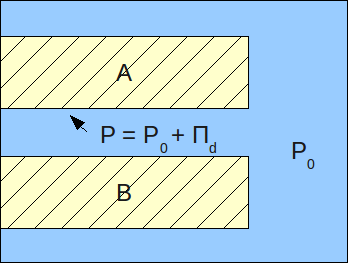 Using the concept of the disjoining pressure, the pressure in a film can be viewed as:
:
where:
* - pressure in a film (Pa)
* - pressure in the bulk of the same phase as that of the film (Pa)
Disjoining pressure is interpreted as a sum of several interactions:
Using the concept of the disjoining pressure, the pressure in a film can be viewed as:
:
where:
* - pressure in a film (Pa)
* - pressure in the bulk of the same phase as that of the film (Pa)
Disjoining pressure is interpreted as a sum of several interactions:
(Google books)
/ref> : where: * - Hamaker constant (J) * - liquid film thickness (m) For a solid-liquid-vapor system where the solid surface is structured, the disjoining pressure is affected by the solid surface profile, , and the
surface chemistry
Surface science is the study of physical and chemical phenomena that occur at the interface of two phases, including solid–liquid interfaces, solid–gas interfaces, solid–vacuum interfaces, and liquid–gas interfaces. It includes the fiel ...
, disjoining pressure (symbol ) according to an IUPAC
The International Union of Pure and Applied Chemistry (IUPAC ) is an international federation of National Adhering Organizations working for the advancement of the chemical sciences, especially by developing nomenclature and terminology. It is ...
definition arises from an attractive interaction between two surfaces. For two flat and parallel surfaces, the value of the disjoining pressure
Pressure (symbol: ''p'' or ''P'') is the force applied perpendicular to the surface of an object per unit area over which that force is distributed. Gauge pressure (also spelled ''gage'' pressure)The preferred spelling varies by country and e ...
(i.e., the force
In physics, a force is an influence that can change the motion of an object. A force can cause an object with mass to change its velocity (e.g. moving from a state of rest), i.e., to accelerate. Force can also be described intuitively as a p ...
per unit area) can be calculated as the derivative
In mathematics, the derivative of a function of a real variable measures the sensitivity to change of the function value (output value) with respect to a change in its argument (input value). Derivatives are a fundamental tool of calculus. F ...
of the Gibbs energy
In thermodynamics, the Gibbs free energy (or Gibbs energy; symbol G) is a thermodynamic potential that can be used to calculate the maximum amount of work (physics), work that may be performed by a closed system, thermodynamically closed system a ...
of interaction per unit area in respect to distance (in the direction normal Normal(s) or The Normal(s) may refer to:
Film and television
* ''Normal'' (2003 film), starring Jessica Lange and Tom Wilkinson
* ''Normal'' (2007 film), starring Carrie-Anne Moss, Kevin Zegers, Callum Keith Rennie, and Andrew Airlie
* ''Norma ...
to that of the interacting surfaces). There is also a related concept of disjoining force, which can be viewed as disjoining pressure times the surface area of the interacting surfaces.
The concept of disjoining pressure was introduced by Derjaguin (1936) as the difference between the pressure in a region of a phase
Phase or phases may refer to:
Science
*State of matter, or phase, one of the distinct forms in which matter can exist
*Phase (matter), a region of space throughout which all physical properties are essentially uniform
* Phase space, a mathematic ...
adjacent to a surface confining it, and the pressure in the bulk of this phase.
Description
Disjoining pressure can be expressed as:Hans-Jürgen Butt, Karlheinz Graf, Michael Kappl,"Physics and chemistry of interfaces", John Wiley & Sons Canada, Ltd., 1 edition, 2003, page 9(Google books)
/ref> : where (in
SI units
The International System of Units, known by the international abbreviation SI in all languages and sometimes Pleonasm#Acronyms and initialisms, pleonastically as the SI system, is the modern form of the metric system and the world's most wid ...
):
* - disjoining pressure (N/m2)
* - the surface area of the interacting surfaces (m2)
* - total Gibbs energy
In thermodynamics, the Gibbs free energy (or Gibbs energy; symbol G) is a thermodynamic potential that can be used to calculate the maximum amount of work (physics), work that may be performed by a closed system, thermodynamically closed system a ...
of the interaction of the two surfaces (J)
* - distance (m)
* indices and signify that the temperature, volume, and the surface area remain constant in the derivative.
 Using the concept of the disjoining pressure, the pressure in a film can be viewed as:
:
where:
* - pressure in a film (Pa)
* - pressure in the bulk of the same phase as that of the film (Pa)
Disjoining pressure is interpreted as a sum of several interactions:
Using the concept of the disjoining pressure, the pressure in a film can be viewed as:
:
where:
* - pressure in a film (Pa)
* - pressure in the bulk of the same phase as that of the film (Pa)
Disjoining pressure is interpreted as a sum of several interactions: dispersion forces
London dispersion forces (LDF, also known as dispersion forces, London forces, instantaneous dipole–induced dipole forces, fluctuating induced dipole bonds or loosely as van der Waals forces) are a type of intermolecular force acting between at ...
, electrostatic forces between charged surfaces, interactions due to layers of neutral molecules adsorbed
Adsorption is the adhesion of atoms, ions or molecules from a gas, liquid or dissolved solid to a surface. This process creates a film of the ''adsorbate'' on the surface of the ''adsorbent''. This process differs from absorption, in which a ...
on the two surfaces, and the structural effects of the solvent.
Classic theory predicts that the disjoining pressure of a thin liquid film on a flat surface as follows,Jacob N. Israelachvili,"Intermolecular and Surface Forces", Academic Press, Revised Third edition, 2011, page 267-26(Google books)
/ref> : where: * - Hamaker constant (J) * - liquid film thickness (m) For a solid-liquid-vapor system where the solid surface is structured, the disjoining pressure is affected by the solid surface profile, , and the
meniscus
Meniscus may refer to:
*Meniscus (anatomy), crescent-shaped fibrocartilaginous structure that partly divides a joint cavity
*Meniscus (liquid)
The meniscus (plural: ''menisci'', from the Greek for "crescent") is the curve in the upper surface ...
shape,
:
where:
* - solid-liquid potential (J/m6)
The meniscus shape can be by minimization of total system free energy as follows
:
where:
* - total system free energy including surface excess energy and free energy due to solid-liquid interactions (J/m2)
* - meniscus shape (m)
* - slope of meniscus shape (1)
In the theory of liquid drops and films, the disjoining pressure can be shown to be related to the equilibrium liquid-solid contact angle
The contact angle is the angle, conventionally measured through the liquid, where a liquid–vapor interface meets a solid surface. It quantifies the wettability of a solid surface by a liquid via the Young equation. A given system of solid, liq ...
through the relation
:
where is the liquid-vapor surface tension
Surface tension is the tendency of liquid surfaces at rest to shrink into the minimum surface area possible. Surface tension is what allows objects with a higher density than water such as razor blades and insects (e.g. water striders) to f ...
and is the precursor film thickness.
See also
* Capillary condensation * Capillary pressure *Hamaker constant
The Hamaker constant ''A'' can be defined for a van der Waals (vdW) body–body interaction:
:A=\pi^2C\rho_1\rho_2,
where \rho_1 and \rho_2 are the number densities of the two interacting kinds of particles, and ''C'' is the London coefficient in ...
*Thin-film equation
In fluid mechanics, the thin-film equation is a partial differential equation that approximately predicts the time evolution of the thickness of a liquid film that lies on a surface. The equation is derived via lubrication theory which is based o ...
References