Allotropes Of Carbon on:
[Wikipedia]
[Google]
[Amazon]


 Carbon nanobuds are a newly discovered allotrope of
Carbon nanobuds are a newly discovered allotrope of
 Glassy carbon or vitreous carbon is a class of non-graphitizing
Glassy carbon or vitreous carbon is a class of non-graphitizing
 bcc-carbon: At ultrahigh pressures of above 1000 GPa, diamond is predicted to transform into a body-centred cubic structure. This phase has importance in astrophysics and deep interiors of planets like
bcc-carbon: At ultrahigh pressures of above 1000 GPa, diamond is predicted to transform into a body-centred cubic structure. This phase has importance in astrophysics and deep interiors of planets like  * The Laves graph or ''K''4 crystal is a theoretically predicted three-dimensional crystalline metastable carbon structure in which each carbon atom is bonded to three others, at 120° angles (like graphite), but where the bond planes of adjacent layers lie at an angle of 70.5°, rather than coinciding.
* M-carbon: Monoclinic C-centered carbon is thought to have been first created in 1963 by compressing graphite at room temperature. Its structure was theorized in 2006,
then in 2009 it was related to those experimental observations.
Many structural candidates, including bct-carbon, were proposed to be equally compatible with experimental data available at the time, until in 2012 it was shown theoretically that this structure is kinetically the most likely to form from graphite.
High-resolution data appeared shortly after, demonstrating that among all structure candidates only M-carbon is compatible with experiment.
* Metallic carbon: Theoretical studies have shown that there are regions in the phase diagram, at extremely high pressures, where carbon has metallic character. Laser shock experiments and theory indicate that above 600 GPa liquid carbon is metallic.
* Novamene: A combination of both hexagonal diamond and sp2 hexagons as in graphene.
*
* The Laves graph or ''K''4 crystal is a theoretically predicted three-dimensional crystalline metastable carbon structure in which each carbon atom is bonded to three others, at 120° angles (like graphite), but where the bond planes of adjacent layers lie at an angle of 70.5°, rather than coinciding.
* M-carbon: Monoclinic C-centered carbon is thought to have been first created in 1963 by compressing graphite at room temperature. Its structure was theorized in 2006,
then in 2009 it was related to those experimental observations.
Many structural candidates, including bct-carbon, were proposed to be equally compatible with experimental data available at the time, until in 2012 it was shown theoretically that this structure is kinetically the most likely to form from graphite.
High-resolution data appeared shortly after, demonstrating that among all structure candidates only M-carbon is compatible with experiment.
* Metallic carbon: Theoretical studies have shown that there are regions in the phase diagram, at extremely high pressures, where carbon has metallic character. Laser shock experiments and theory indicate that above 600 GPa liquid carbon is metallic.
* Novamene: A combination of both hexagonal diamond and sp2 hexagons as in graphene.
*
 The system of carbon allotropes spans an astounding range of extremes, considering that they are all merely structural formations of the same element.
Between diamond and graphite:
* Diamond crystallizes in the
The system of carbon allotropes spans an astounding range of extremes, considering that they are all merely structural formations of the same element.
Between diamond and graphite:
* Diamond crystallizes in the

Carbon
Carbon () is a chemical element with the symbol C and atomic number 6. It is nonmetallic and tetravalent—its atom making four electrons available to form covalent chemical bonds. It belongs to group 14 of the periodic table. Carbon makes ...
is capable of forming many allotropes (structurally different forms of the same element) due to its valency
Valence or valency may refer to:
Science
* Valence (chemistry), a measure of an element's combining power with other atoms
* Degree (graph theory), also called the valency of a vertex in graph theory
* Valency (linguistics), aspect of verbs re ...
. Well-known forms of carbon include diamond
Diamond is a solid form of the element carbon with its atoms arranged in a crystal structure called diamond cubic. Another solid form of carbon known as graphite is the chemically stable form of carbon at room temperature and pressure, ...
and graphite. In recent decades, many more allotropes have been discovered and researched, including ball shapes such as buckminsterfullerene
Buckminsterfullerene is a type of fullerene with the formula C60. It has a cage-like fused-ring structure (truncated icosahedron) made of twenty hexagons and twelve pentagons, and resembles a soccer ball. Each of its 60 carbon atoms is bonded ...
and sheets such as graphene
Graphene () is an allotrope of carbon consisting of a Single-layer materials, single layer of atoms arranged in a hexagonal lattice nanostructure.
. Larger-scale structures of carbon include carbon nano tube, nanotubes, nanobuds and nanoribbons. Other unusual forms of carbon exist at very high temperatures or extreme pressures. Around 500 hypothetical 3‑periodic allotropes of carbon are known at the present time, according to the Samara Carbon Allotrope Database (SACADA).
Diamond
Diamond is a well-known allotrope of carbon. Thehardness
In materials science, hardness (antonym: softness) is a measure of the resistance to localized plastic deformation induced by either mechanical indentation or abrasion (mechanical), abrasion. In general, different materials differ in their hardn ...
, extremely high refractive index, and high dispersion of light make diamond useful for industrial applications and for jewelry. Diamond is the hardest known natural mineral
In geology and mineralogy, a mineral or mineral species is, broadly speaking, a solid chemical compound with a fairly well-defined chemical composition and a specific crystal structure that occurs naturally in pure form.John P. Rafferty, ed. ...
. This makes it an excellent abrasive and makes it hold polish and luster extremely well. No known naturally occurring substance can cut or scratch diamond, except another diamond. In diamond form, carbon is one of the costliest elements.
The crystal structure of diamond is a face-centred cubic lattice having eight atoms per unit cell to form a diamond cubic structure. Each carbon atom is covalently bonded to four other carbons in a tetrahedral geometry. These tetrahedrons together form a 3-dimensional network of six-membered carbon rings in the chair conformation, allowing for zero bond angle strain. The bonding occurs through sp3 hybridized orbitals to give a C-C bond length of 154 pm. This network of unstrained covalent bonds makes diamond extremely strong. Diamond is thermodynamically less stable than graphite at pressures below .
The dominant industrial use of diamond is cutting, drilling ( drill bits), grinding (diamond edged cutters), and polishing. Most uses of diamonds in these technologies do not require large diamonds, and most diamonds that are not gem-quality can find an industrial use. Diamonds are embedded in drill tips and saw blades, or ground into a powder for use in grinding and polishing applications (due to its extraordinary hardness). Specialized applications include use in laboratories as containment for high pressure experiments (see diamond anvil), high-performance bearings, and specialized windows of technical apparatuses.
The market for industrial-grade diamonds operates much differently from its gem-grade counterpart. Industrial diamonds are valued mostly for their hardness and heat conductivity, making many of the gemological characteristics of diamond, including clarity and color, mostly irrelevant. This helps explain why 80% of mined diamonds (equal to about 100 million carats or 20 tonne
The tonne ( or ; symbol: t) is a unit of mass equal to 1000 kilograms. It is a non-SI unit accepted for use with SI. It is also referred to as a metric ton to distinguish it from the non-metric units of the short ton ( United State ...
s annually) are unsuitable for use as gemstones and known as '' bort'', are destined for industrial use. In addition to mined diamonds, synthetic diamonds found industrial applications almost immediately after their invention in the 1950s; another 400 million carats (80 tonnes) of synthetic diamonds are produced annually for industrial use, which is nearly four times the mass of natural diamonds mined over the same period.
With the continuing advances being made in the production of synthetic diamond, future applications are beginning to become feasible. Garnering much excitement is the possible use of diamond as a semiconductor suitable to build microchips from, or the use of diamond as a heat sink
A heat sink (also commonly spelled heatsink) is a passive heat exchanger that transfers the heat generated by an electronic or a mechanical device to a fluid medium, often air or a liquid coolant, where it is dissipated away from the device, t ...
in electronics
The field of electronics is a branch of physics and electrical engineering that deals with the emission, behaviour and effects of electrons using electronic devices. Electronics uses active devices to control electron flow by amplification ...
. Significant research efforts in Japan, Europe
Europe is a large peninsula conventionally considered a continent in its own right because of its great physical size and the weight of its history and traditions. Europe is also considered a subcontinent of Eurasia and it is located enti ...
, and the United States
The United States of America (U.S.A. or USA), commonly known as the United States (U.S. or US) or America, is a country primarily located in North America. It consists of 50 U.S. state, states, a Washington, D.C., federal district, five ma ...
are under way to capitalize on the potential offered by diamond's unique material properties, combined with increased quality and quantity of supply starting to become available from synthetic diamond manufacturers.
Graphite
Graphite, named by Abraham Gottlob Werner in 1789, from the Greek γράφειν (, "to draw/write", for its use in pencils) is one of the most common allotropes of carbon. Unlike diamond, graphite is an electrical conductor. Thus, it can be used in, for instance, electrical arc lamp electrodes. Likewise, under standard conditions, graphite is the most stable form of carbon. Therefore, it is used in thermochemistry as the standard state for defining the heat of formation of carbon compounds. Graphite conducts electricity, due to delocalization of thepi bond
In chemistry, pi bonds (π bonds) are covalent chemical bonds, in each of which two lobes of an orbital on one atom overlap with two lobes of an orbital on another atom, and in which this overlap occurs laterally. Each of these atomic orbita ...
electron
The electron (, or in nuclear reactions) is a subatomic particle with a negative one elementary electric charge. Electrons belong to the first generation of the lepton particle family,
and are generally thought to be elementary partic ...
s above and below the planes of the carbon atoms. These electrons are free to move, so are able to conduct electricity. However, the electricity is only conducted along the plane of the layers. In diamond, all four outer electrons of each carbon atom are 'localized' between the atoms in covalent bonding. The movement of electrons is restricted and diamond does not conduct an electric current. In graphite, each carbon atom uses only 3 of its 4 outer energy level electrons in covalently bonding to three other carbon atoms in a plane. Each carbon atom contributes one electron to a delocalized system of electrons that is also a part of the chemical bonding. The delocalized electrons are free to move throughout the plane. For this reason, graphite conducts electricity along the planes of carbon atoms, but does not conduct electricity in a direction at right angles to the plane.
Graphite powder is used as a dry lubricant. Although it might be thought that this industrially important property is due entirely to the loose interlamellar coupling between sheets in the structure, in fact in a vacuum
A vacuum is a space devoid of matter. The word is derived from the Latin adjective ''vacuus'' for "vacant" or " void". An approximation to such vacuum is a region with a gaseous pressure much less than atmospheric pressure. Physicists often di ...
environment (such as in technologies for use in space
Space is the boundless three-dimensional extent in which objects and events have relative position and direction. In classical physics, physical space is often conceived in three linear dimensions, although modern physicists usually con ...
), graphite was found to be a very poor lubricant. This fact led to the discovery that graphite's lubricity is due to adsorbed air and water between the layers, unlike other layered dry lubricants such as molybdenum disulfide. Recent studies suggest that an effect called superlubricity can also account for this effect.
When a large number of crystallographic defects (physical) bind these planes together, graphite loses its lubrication properties and becomes pyrolytic carbon, a useful material in blood-contacting implants such as prosthetic heart valves.
Graphite is the most stable allotrope of carbon. Contrary to popular belief, high-purity graphite does not readily burn, even at elevated temperatures. For this reason, it is used in nuclear reactor
A nuclear reactor is a device used to initiate and control a fission nuclear chain reaction or nuclear fusion reactions. Nuclear reactors are used at nuclear power plants for electricity generation and in nuclear marine propulsion. Heat from nu ...
s and for high-temperature crucibles for melting metals. At very high temperatures and pressures (roughly 2000 °C and 5 GPa), it can be transformed into diamond.
Natural and crystalline graphites are not often used in pure form as structural materials due to their shear-planes, brittleness and inconsistent mechanical properties.
In its pure glassy (isotropic) synthetic forms, pyrolytic graphite and carbon fiber
Carbon fiber-reinforced polymers (American English), carbon-fibre-reinforced polymers (Commonwealth English), carbon-fiber-reinforced plastics, carbon-fiber reinforced-thermoplastic (CFRP, CRP, CFRTP), also known as carbon fiber, carbon compo ...
graphite are extremely strong, heat-resistant (to 3000 °C) materials, used in reentry shields for missile nosecones, solid rocket engines, high temperature reactors, brake
A brake is a mechanical device that inhibits motion by absorbing energy from a moving system. It is used for slowing or stopping a moving vehicle, wheel, axle, or to prevent its motion, most often accomplished by means of friction.
Backgroun ...
shoes and electric motor
An electric motor is an electrical machine that converts electrical energy into mechanical energy. Most electric motors operate through the interaction between the motor's magnetic field and electric current in a wire winding to generate forc ...
brushes.
Intumescent or expandable graphites are used in fire seals, fitted around the perimeter of a fire door. During a fire the graphite intumesces (expands and chars) to resist fire penetration and prevent the spread of fumes. A typical ''start expansion temperature'' (SET) is between 150 and 300 °C.
Density: Graphite's specific gravity is 2.3, which makes it lighter than diamond.
Chemical activity: it is slightly more reactive than diamond. This is because the reactants are able to penetrate between the hexagonal layers of carbon atoms in graphite. It is unaffected by ordinary solvents, dilute acids, or fused alkalis. However, chromic acid oxidizes it to carbon dioxide.
Graphene
A single layer of graphite is calledgraphene
Graphene () is an allotrope of carbon consisting of a Single-layer materials, single layer of atoms arranged in a hexagonal lattice nanostructure.
and has extraordinary electrical, thermal, and physical properties. It can be produced by epitaxy on an insulating or conducting substrate or by mechanical exfoliation (repeated peeling) from graphite. Its applications may include replacing silicon
Silicon is a chemical element with the symbol Si and atomic number 14. It is a hard, brittle crystalline solid with a blue-grey metallic luster, and is a tetravalent metalloid and semiconductor. It is a member of group 14 in the periodic ...
in high-performance electronic devices. With two layers stacked, bilayer graphene results with different properties.
Lonsdaleite (hexagonal diamond)
Lonsdaleite is an allotrope sometimes called " hexagonal diamond", formed from graphite present inmeteor
A meteoroid () is a small rocky or metallic body in outer space.
Meteoroids are defined as objects significantly smaller than asteroids, ranging in size from grains to objects up to a meter wide. Objects smaller than this are classified as mic ...
ites upon their impact on the earth. The great heat and pressure of the impact transforms the graphite into a denser form similar to diamond, but retaining graphite's hexagonal crystal lattice. "Hexagonal diamond" has also been synthesized in the laboratory, by compressing and heating graphite either in a static press or using explosives. It can also be produced by the thermal decomposition of a polymer, poly(hydridocarbyne)
Poly(hydridocarbyne) (PHC) is one of a class of carbon-based random network polymers primarily composed of tetrahedrally hybridized carbon atoms, each having one hydride substituent, exhibiting the generic formula C. PHC is made from bromoform, ...
, at atmospheric pressure, under inert gas atmosphere (e.g. argon, nitrogen), starting at temperature .
Graphenylene
Graphenylene is a single layer carbon material with biphenylene-like subunits as basis in its hexagonal lattice structure. It is also known as biphenylene-carbon.Carbophene
Carbophene is a 2 dimensional covalent organic framework. 4-6 carbophene has been synthesized from 1-3-5 trihydroxybenzene. It consists of 4-carbon and 6-carbon rings in 1:1 ratio. The angles between the three σ-bonds of the orbitals are approximately 120°, 90°, and 150°.AA'-graphite
AA'-graphite
AA'-graphite is an allotrope of carbon similar to graphite, but where the layers are positioned differently to each other as compared to the order in graphite.
AA’ stacking of graphene planes is another crystalline form of graphite (orthorhomb ...
is an allotrope of carbon similar to graphite, but where the layers are positioned differently to each other as compared to the order in graphite.
Diamane
Diamane is a 2D form of diamond. It can be made via high pressures, but without that pressure, the material reverts to graphene. Another technique is to add hydrogen atoms but those bonds are weak. Using fluorine (xenon-difluoride) instead brings the layers closer together, strengthening the bonds. This is called f-diamane.Amorphous carbon
Amorphous carbon is the name used forcarbon
Carbon () is a chemical element with the symbol C and atomic number 6. It is nonmetallic and tetravalent—its atom making four electrons available to form covalent chemical bonds. It belongs to group 14 of the periodic table. Carbon makes ...
that does not have any crystal
A crystal or crystalline solid is a solid material whose constituents (such as atoms, molecules, or ions) are arranged in a highly ordered microscopic structure, forming a crystal lattice that extends in all directions. In addition, macr ...
line structure. As with all glassy Glassy is an adjective meaning, of or resembling glass. Glassy may also mean:
* Glassy, amorphous metal
* ''Glassy'', a 2021 single album, or the title song by Jo Yu-ri
* Glassy phase, amorphous solid
* Glassy water, amorphous ice
* Glassy carbo ...
materials, some short-range order can be observed, but there is no long-range pattern of atomic positions. While entirely amorphous carbon can be produced, most amorphous carbon actually contains microscopic crystals of graphite-like, or even diamond
Diamond is a solid form of the element carbon with its atoms arranged in a crystal structure called diamond cubic. Another solid form of carbon known as graphite is the chemically stable form of carbon at room temperature and pressure, ...
-like carbon.
Coal
Coal is a combustible black or brownish-black sedimentary rock, formed as stratum, rock strata called coal seams. Coal is mostly carbon with variable amounts of other Chemical element, elements, chiefly hydrogen, sulfur, oxygen, and nitrogen ...
and soot
Soot ( ) is a mass of impure carbon particles resulting from the incomplete combustion of hydrocarbons. It is more properly restricted to the product of the gas-phase combustion process but is commonly extended to include the residual pyrolyse ...
or carbon black are informally called amorphous carbon. However, they are products of pyrolysis (the process of decomposing a substance by the action of heat), which does not produce true amorphous carbon under normal conditions.
Nanocarbons
Buckminsterfullerenes
The ''buckminsterfullerenes'', or usually just ''fullerenes'' or ''buckyballs'' for short, were discovered in 1985 by a team of scientists from Rice University and the University of Sussex, three of whom were awarded the 1996 Nobel Prize in Chemistry. They are named for the resemblance to the geodesic structures devised by Richard Buckminster "Bucky" Fuller. Fullerenes are positively curved molecules of varying sizes composed entirely of carbon, which take the form of a hollow sphere, ellipsoid, or tube. As of the early twenty-first century, the chemical and physical properties of fullerenes are still under heavy study, in both pure and applied research labs. In April 2003, fullerenes were under study for potential medicinal use — binding specific antibiotics to the structure to target resistant bacteria and even target certain cancer cells such as melanoma.Carbon nanotubes
Carbon nanotubes, also called buckytubes, are cylindricalcarbon
Carbon () is a chemical element with the symbol C and atomic number 6. It is nonmetallic and tetravalent—its atom making four electrons available to form covalent chemical bonds. It belongs to group 14 of the periodic table. Carbon makes ...
molecules
A molecule is a group of two or more atoms held together by attractive forces known as chemical bonds; depending on context, the term may or may not include ions which satisfy this criterion. In quantum physics, organic chemistry, and bioc ...
with novel properties that make them potentially useful in a wide variety of applications (e.g., nano-electronics, optics
Optics is the branch of physics that studies the behaviour and properties of light, including its interactions with matter and the construction of instruments that use or detect it. Optics usually describes the behaviour of visible, ultra ...
, materials
Material is a substance or mixture of substances that constitutes an object. Materials can be pure or impure, living or non-living matter. Materials can be classified on the basis of their physical and chemical properties, or on their geologica ...
applications, etc.). They exhibit extraordinary strength, unique electrical properties, and are efficient conductors of heat
In thermodynamics, heat is defined as the form of energy crossing the boundary of a thermodynamic system by virtue of a temperature difference across the boundary. A thermodynamic system does not ''contain'' heat. Nevertheless, the term is ...
. Non-carbon nanotube
A non-carbon nanotube is a cylindrical molecule often composed of metal oxides, or group III-Nitrides and morphologically similar to a carbon nanotube. Non-carbon nanotubes have been observed to occur naturally in some mineral deposits.
A few year ...
s have also been synthesized.
Carbon nanotubes are a members of the fullerene structural family, which also includes buckyball
Buckminsterfullerene is a type of fullerene with the formula C60. It has a cage-like fused-ring structure (truncated icosahedron) made of twenty hexagons and twelve pentagons, and resembles a soccer ball. Each of its 60 carbon atoms is bonded t ...
s. Whereas buckyballs are spherical in shape, a nanotube is cylindrical, with at least one end typically capped with a hemisphere of the buckyball structure. Their name is derived from their size, since the diameter of a nanotube is on the order of a few nanometers (approximately 50,000 times smaller than the width of a human hair), while they can be up to several centimeters in length. There are two main types of nanotubes: single-walled nanotubes
A scanning tunneling microscopy image of a single-walled carbon nanotube
Rotating single-walled zigzag carbon nanotube
A carbon nanotube (CNT) is a tube made of carbon with diameters typically measured in nanometers.
''Single-wall carbon nan ...
(SWNTs) and multi-walled nanotubes (MWNTs).
Carbon nanobuds
 Carbon nanobuds are a newly discovered allotrope of
Carbon nanobuds are a newly discovered allotrope of carbon
Carbon () is a chemical element with the symbol C and atomic number 6. It is nonmetallic and tetravalent—its atom making four electrons available to form covalent chemical bonds. It belongs to group 14 of the periodic table. Carbon makes ...
in which fullerene like "buds" are covalently attached to the outer sidewalls of the carbon nanotubes
A scanning tunneling microscopy image of a single-walled carbon nanotube
Rotating single-walled zigzag carbon nanotube
A carbon nanotube (CNT) is a tube made of carbon with diameters typically measured in nanometers.
''Single-wall carbon nan ...
. This hybrid material has useful properties of both fullerenes and carbon nanotubes. For instance, they have been found to be exceptionally good field emitters.
Schwarzites
Schwarzites are negatively curved carbon surfaces originally proposed by decorating triply periodic minimal surfaces with carbon atoms. Thegeometric topology
In mathematics, geometric topology is the study of manifolds and maps between them, particularly embeddings of one manifold into another.
History
Geometric topology as an area distinct from algebraic topology may be said to have originat ...
of the structure is determined by the presence of ring defects, such as heptagons and octagons, to graphene
Graphene () is an allotrope of carbon consisting of a Single-layer materials, single layer of atoms arranged in a hexagonal lattice nanostructure.
's hexagonal lattice.
(Negative curvature bends surfaces outwards like a saddle rather than bending inwards like a sphere.)
Recent work has proposed zeolite-templated carbons (ZTCs) may be schwarzites. The name, ZTC, derives from their origin inside the pores of zeolites, crystalline silicon dioxide minerals. A vapor of carbon-containing molecules is injected into the zeolite, where the carbon gathers on the pores' walls, creating the negative curve. Dissolving the zeolite leaves the carbon. A team generated structures by decorating the pores of a zeolite with carbon through a Monte Carlo method
Monte Carlo methods, or Monte Carlo experiments, are a broad class of computational algorithms that rely on repeated random sampling to obtain numerical results. The underlying concept is to use randomness to solve problems that might be deter ...
. Some of the resulting models resemble schwarzite-like structures.
Glassy carbon
 Glassy carbon or vitreous carbon is a class of non-graphitizing
Glassy carbon or vitreous carbon is a class of non-graphitizing carbon
Carbon () is a chemical element with the symbol C and atomic number 6. It is nonmetallic and tetravalent—its atom making four electrons available to form covalent chemical bonds. It belongs to group 14 of the periodic table. Carbon makes ...
widely used as an electrode material in electrochemistry
Electrochemistry is the branch of physical chemistry concerned with the relationship between electrical potential difference, as a measurable and quantitative phenomenon, and identifiable chemical change, with the potential difference as an outc ...
, as well as for high-temperature crucibles and as a component of some prosthetic devices.
It was first produced by Bernard Redfern in the mid-1950s at the laboratories of The Carborundum Company, Manchester, UK. He had set out to develop a polymer matrix to mirror a diamond structure and discovered a resole (phenolic) resin that would, with special preparation, set without a catalyst. Using this resin the first glassy carbon was produced.
The preparation of glassy carbon involves subjecting the organic precursors to a series of heat treatments at temperatures up to 3000 °C. Unlike many non-graphitizing carbons, they are impermeable to gases and are chemically extremely inert, especially those prepared at very high temperatures. It has been demonstrated that the rates of oxidation of certain glassy carbons in oxygen, carbon dioxide or water vapor are lower than those of any other carbon. They are also highly resistant to attack by acids. Thus, while normal graphite is reduced to a powder by a mixture of concentrated sulfuric and nitric acids at room temperature, glassy carbon is unaffected by such treatment, even after several months.
Atomic and diatomic carbon
Under certain conditions, carbon can be found in its atomic form. It can be formed by vaporizing graphite, by passing large electric currents to form a carbon arc under very low pressures. It is extremely reactive, but it is an intermediate product used in the creation of carbenes. Diatomic carbon can also be found under certain conditions. It is often detected via spectroscopy in extraterrestrial bodies, includingcomet
A comet is an icy, small Solar System body that, when passing close to the Sun, warms and begins to release gases, a process that is called outgassing. This produces a visible atmosphere or Coma (cometary), coma, and sometimes also a Comet ta ...
s and certain star
A star is an astronomical object comprising a luminous spheroid of plasma held together by its gravity. The nearest star to Earth is the Sun. Many other stars are visible to the naked eye at night, but their immense distances from Earth make ...
s.
Carbon nanofoam
Carbon nanofoam is the fifth known allotrope of carbon, discovered in 1997 byAndrei V. Rode
Andrei, Andrey or Andrej (in Cyrillic script: Андрэй , Андрей or Андреј) is a form of Andreas/Ἀνδρέας in Slavic languages and Romanian. People with the name include:
*Andrei of Polotsk (–1399), Lithuanian nobleman
*And ...
and co-workers at the Australian National University
The Australian National University (ANU) is a public research university located in Canberra, the capital of Australia. Its main campus in Acton encompasses seven teaching and research colleges, in addition to several national academies and ...
in Canberra. It consists of a low-density cluster-assembly of carbon atoms strung together in a loose three-dimensional web.
Each cluster is about 6 nanometers wide and consists of about 4000 carbon atom
Every atom is composed of a nucleus and one or more electrons bound to the nucleus. The nucleus is made of one or more protons and a number of neutrons. Only the most common variety of hydrogen has no neutrons.
Every solid, liquid, gas ...
s linked in graphite-like sheets that are given negative curvature by the inclusion of heptagons among the regular hexagon
In geometry, a hexagon (from Greek , , meaning "six", and , , meaning "corner, angle") is a six-sided polygon. The total of the internal angles of any simple (non-self-intersecting) hexagon is 720°.
Regular hexagon
A ''regular hexagon'' h ...
al pattern. This is the opposite of what happens in the case of buckminsterfullerene
Buckminsterfullerene is a type of fullerene with the formula C60. It has a cage-like fused-ring structure (truncated icosahedron) made of twenty hexagons and twelve pentagons, and resembles a soccer ball. Each of its 60 carbon atoms is bonded ...
s, in which carbon sheets are given positive curvature by the inclusion of pentagon
In geometry, a pentagon (from the Greek language, Greek πέντε ''pente'' meaning ''five'' and γωνία ''gonia'' meaning ''angle'') is any five-sided polygon or 5-gon. The sum of the internal angles in a simple polygon, simple pentagon is ...
s.
The large-scale structure of carbon nanofoam is similar to that of an aerogel, but with 1% of the density of previously produced carbon aerogels – only a few times the density of air at sea level
Mean sea level (MSL, often shortened to sea level) is an average surface level of one or more among Earth's coastal bodies of water from which heights such as elevation may be measured. The global MSL is a type of vertical datuma standardise ...
. Unlike carbon aerogels, carbon nanofoam is a poor electrical conductor.
Carbide-derived carbon
Carbide-derived carbon (CDC) is a family of carbon materials with different surface geometries and carbon ordering that are produced via selective removal of metals from metal carbide precursors, such as TiC, SiC, , , etc. This synthesis is accomplished using chlorine treatment, hydrothermal synthesis, or high-temperature selective metal desorption under vacuum. Depending on the synthesis method, carbide precursor, and reaction parameters, multiple carbon allotropes can be achieved, including endohedral particles composed of predominantly amorphous carbon, carbon nanotubes, epitaxial graphene, nanocrystalline diamond, onion-like carbon, and graphitic ribbons, barrels, and horns. These structures exhibit high porosity and specific surface areas, with highly tunable pore diameters, making them promising materials for supercapacitor-based energy storage, water filtration and capacitive desalinization, catalyst support, and cytokine removal.Linear acetylenic carbon
A one-dimensional carbon polymer with the structure —(C≡C)n—.Cyclocarbons
Cyclo 8arbon (C18) was synthesised in 2019.Other possible allotropes
Many other allotropes have been hypothesized but have yet to be synthesized. *Uranus
Uranus is the seventh planet from the Sun. Its name is a reference to the Greek god of the sky, Uranus (Caelus), who, according to Greek mythology, was the great-grandfather of Ares (Mars), grandfather of Zeus (Jupiter) and father of Cronu ...
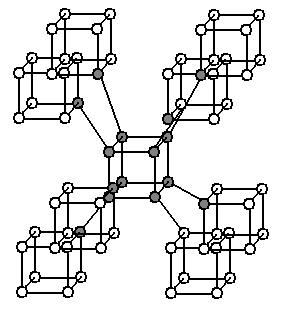
and Neptune. Various structures have been proposed. Superdense and superhard material resembling this phase was synthesized and published in 1979 and reported to have the Im space group with eight atoms per primitive unit cell (16 atoms per conventional unit cell). Claims were made that the so-called C structure had been synthesized, having eight-carbon cubes similar to cubane in the Imm space group, with eight atoms per primitive unit cell, or 16 atoms per conventional unit cell (also called supercubane, see illustration to the right). But a paper in 1988 claimed that a better theory was that the structure was the same as that of an allotrope of silicon
Silicon is a chemical element with the symbol Si and atomic number 14. It is a hard, brittle crystalline solid with a blue-grey metallic luster, and is a tetravalent metalloid and semiconductor. It is a member of group 14 in the periodic ...
called Si-III or γ-silicon, the so-called BC8 structure with space group Ia and 8 atoms per primitive unit cell (16 atoms per conventional unit cell). In 2008 it was reported that the cubane-like structure had been identified. A paper in 2012 considered four proposed structures, the supercubane structure, the BC8 structure, a structure with clusters of four carbon atoms in tetrahedra in space group I3m having four atoms per primitive unit cell (eight per conventional unit cell), and a structure the authors called "carbon sodalite". They found in favor of this carbon sodalite structure, with a calculated density of 2.927 g/cm, shown in the upper left of the illustration under the abstract. This structure has just six atoms per primitive unit cell (twelve per conventional unit cell). The carbon atoms are in the same locations as the silicon and aluminum atoms of the mineral sodalite. The space group, I3m, is the same as the fully expanded form of sodalite would have if sodalite had just silicon or just aluminum.
* bct-carbon: Body-centered tetragonal carbon was proposed by theorists in 2010.
* Chaoite is a mineral believed to have been formed in meteorite impacts. It has been described as slightly harder than graphite with a reflection color of grey to white. However, the existence of carbyne phases is disputed – see the article on chaoite for details.
* D-carbon: D-carbon was proposed by theorists in 2018.
D-carbon is an orthorhombic sp3 carbon allotrope (6 atoms per cell). Total-energy calculations demonstrate that D-carbon is energetically more favorable than the previously proposed T6 structure (with 6 atoms per cell) as well as many others.
* Haeckelites: Ordered arrangements of pentagons, hexagons, and heptagons which can either be flat or tubular.
Phagraphene Phagraphene 'fæ’græfiːn''is a proposed graphene allotropes of carbon, allotrope composed of 5-6-7 carbon rings. Phagraphene was proposed in 2015 based on systematic evolutionary structure searching. Theoretical calculations showed that phag ...
: Graphene-like allotrope with distorted Dirac cones.
* Prismane C8 is a theoretically predicted metastable carbon allotrope comprising an atomic cluster
may refer to:
Science and technology Astronomy
* Cluster (spacecraft), constellation of four European Space Agency spacecraft
* Asteroid cluster, a small asteroid family
* Cluster II (spacecraft), a European Space Agency mission to study th ...
of eight carbon atoms, with the shape of an elongated triangular bipyramid—a six-atom triangular prism with two more atoms above and below its bases.
* Protomene: A hexagonal crystal structure with a fully relaxed primitive cell involving 48 atoms. Out of these, 12 atoms have the potential to switch hybridization between sp2 and sp3, forming dimers.
* Q-carbon: Ferromagnetic carbon was discovered in 2015.
* T-carbon: Every carbon atom in diamond is replaced with a carbon tetrahedron (hence 'T-carbon'). This was proposed by theorists in 1985.
* There is evidence that white dwarf stars have a core of crystallized carbon and oxygen nuclei. The largest of these found in the universe so far, BPM 37093, is located away in the constellation Centaurus. A news release from the Harvard-Smithsonian Center for Astrophysics described the -wide stellar core as a ''diamond'', and it was named as ''Lucy'', after the Beatles' song "Lucy in the Sky With Diamonds"; however, it is more likely an exotic form of carbon. Penta-graphene is a predicted carbon allotrope that utilizes the Cairo pentagonal tiling.
* U carbon is predicted to consist of corrugated layers tiled with six- or 12-atom rings, linked by covalent bonds. Notably, it can be harder than steel, as conductive as stainless steel, highly reflective and ferromagnetic, behaving as a permanent magnet
A magnet is a material or object that produces a magnetic field. This magnetic field is invisible but is responsible for the most notable property of a magnet: a force that pulls on other ferromagnetic materials, such as iron, steel, nic ...
at temperatures up to 125 °C.
* Zayedene: A combination of linear sp carbon chains and sp3 bulk carbon. The structure of these crystalline carbon allotropes consists of sp chains inserted in cylindrical cavities periodically arranged in hexagonal diamond (lonsdaleite).
Variability of carbon
 The system of carbon allotropes spans an astounding range of extremes, considering that they are all merely structural formations of the same element.
Between diamond and graphite:
* Diamond crystallizes in the
The system of carbon allotropes spans an astounding range of extremes, considering that they are all merely structural formations of the same element.
Between diamond and graphite:
* Diamond crystallizes in the cubic system
In crystallography, the cubic (or isometric) crystal system is a crystal system where the unit cell is in the shape of a cube. This is one of the most common and simplest shapes found in crystals and minerals.
There are three main varieties of ...
but graphite crystallizes in the hexagonal system
In crystallography, the hexagonal crystal family is one of the six crystal families, which includes two crystal systems (hexagonal and trigonal) and two lattice systems (hexagonal and rhombohedral). While commonly confused, the trigonal crystal ...
.
* Diamond is clear and transparent, but graphite is black and opaque.
* Diamond is the hardest mineral known (10 on the Mohs scale), but graphite is one of the softest (1–2 on Mohs scale).
* Diamond is the ultimate abrasive, but graphite is soft and is a very good lubricant.
* Diamond is an excellent electrical insulator, but graphite is an excellent conductor.
* Diamond is an excellent thermal conductor, but some forms of graphite are used for thermal insulation (for example heat shields and firebreaks).
* At standard temperature and pressure, graphite is the thermodynamically stable form. Thus diamonds do not exist forever. The conversion from diamond to graphite, however, has a very high activation energy and is therefore extremely slow.
Despite the hardness of diamonds, the chemical bonds that hold the carbon atoms in diamonds together are actually weaker than those that hold together graphite. The difference is that in diamond, the bonds form an inflexible three-dimensional lattice. In graphite, the atoms are tightly bonded into sheets, but the sheets can slide easily over each other, making graphite soft.
See also
*Superdense carbon allotropes Superdense carbon allotropes are proposed configurations of carbon atoms that result in a stable material with a higher density than diamond.
Few hypothetical carbon allotropes denser than diamond are known. All these allotropes can be divided at ...
References
External links
* * * {{DEFAULTSORT:Carbon Allotropes Carbon