|
Umbelliferone
Umbelliferone, also known as 7-hydroxycoumarin, hydrangine, skimmetine, and ''beta''-umbelliferone, is a natural product of the coumarin family. It absorbs ultraviolet light strongly at several wavelengths. There are some indications that this chemical is antimutagenic, it is used in sunscreens. Umbelliferone has been reported to have antioxidant properties. It is a yellowish-white crystalline solid that has a slight solubility in hot water, but high solubility in ethanol. Natural occurrences and name Umbelliferone's name is from the umbelliferae family of plants, and the plant family in turn was named for their umbrella-shaped inflorescences, each called an umbel. Umbelliferone occurs in many familiar plants from the Apiaceae (Umbelliferae) family such as carrot, coriander and garden angelica, as well as in plants from other families, such as the mouse-ear hawkweed (''Hieracium pilosella'', Asteraceae) or the bigleaf hydrangea ('' Hydrangea macrophylla'', Hydrangea ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Coumarin
Coumarin () or 2''H''-chromen-2-one is an aromatic organic chemical compound with formula . Its molecule can be described as a benzene molecule with two adjacent hydrogen atoms replaced by a lactone-like chain , forming a second six-membered heterocycle that shares two carbons with the benzene ring. It can be placed in the benzopyrone chemical class and considered as a lactone. Coumarin is a colorless crystalline solid with a sweet odor resembling the scent of vanilla and a bitter taste. It is found in many plants, where it may serve as a chemical defense against predators. By inhibiting synthesis of vitamin K, a related compound is used as the prescription drug warfarin – an anticoagulant – to inhibit formation of blood clots, deep vein thrombosis, and pulmonary embolism. Etymology Coumarin is derived from ''coumarou'', the French word for the tonka bean. The word ''tonka'' for the tonka bean is taken from the Galibi (Carib) tongue spoken by natives of ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Umbellic Acid
Umbellic acid (2,4-dihydroxycinnamic acid) is a hydroxycinnamic acid. It is an isomer of caffeic acid. It is a precursor in the umbelliferone biosynthesis pathway. Umbelliferone is a phenylpropanoid and as such is synthesized from L-phenylalanine, which in turn is produced via the shikimate pathway. Phenylalanine is lysated into cinnamic acid, followed by hydroxylation by cinnamate 4-hydroxylase to yield 4-coumaric acid. The 4-coumaric acid is again hydroxylated by cinnamate/coumarate 2-hydroxylase to yield 2,4-dihydroxy-cinnamic acid followed by a bond rotation of the unsaturated bond adjacent to the carboxylic acid group. Finally an intramolecular attack from the hydroxyl group of C2' to the carboxylic acid group closes the ring and forms the lactone umbelliferone. : \xrightarrow \xrightarrow \xrightarrow \longrightarrow The enzyme 4-hydroxycinnamate decarboxylase, induced in bacteria species such as ''Klebsiella oxytoca ''Kleb ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Justicia Pectoralis
''Justicia pectoralis'' is an herb in the family Acanthaceae. This water-willow is widely known as tilo in Latin America and in Cuba. In Haiti it is called ''chapantye'' and ''zeb chapantyè'' on Dominica and Martinique. Other folk names are freshcut, chambá ''carpintero'' ("carpenter"), ''té criollo'' ("'' Criollo'' tea"), curia, death-angel, masha-hari, or "piri piri". This species was described by Nikolaus Joseph von Jacquin in 1760, who provided additional data in 1763. A well-marked variety, var. ''stenophylla'', was described by Emery Clarence Leonard in 1958. Uses Traditional uses Across its range it is used in folk medicine as a relaxant and general tonic. Additionally it is often used in Ayahuasca, a tea containing the ''Banisteriopsis caapi'' vine. Other uses As regards other applications, it is noted for its pleasant smell and as a source of coumarin, which it produces in plenty, and which in combination with umbelliferone is responsible for many of its notable pr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Asafoetida
Asafoetida (; also spelled asafetida) is the dried latex (gum oleoresin) exuded from the rhizome or tap root of several species of '' Ferula'', perennial herbs growing tall. They are part of the celery family, Umbelliferae. Asafoetida is thought to be in the same genus as silphium, a North African plant now believed to be extinct, and was used as a cheaper substitute for that historically important herb from classical antiquity. The species are native to the deserts of Iran and mountains of Afghanistan where substantial amounts are grown. Asafoetida has a pungent smell, as reflected in its name, lending it the trivial name of "stinking gum". The odor dissipates upon cooking; in cooked dishes, it delivers a smooth flavour reminiscent of leeks or other onion relatives. Asafoetida is also known colloquially as "devil's dung" in English (and similar expressions in many other languages). Etymology and other names The English name is derived from ''asa'', a latinised form o ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrangea Macrophylla
''Hydrangea macrophylla'' is a species of flowering plant in the family Hydrangeaceae, native to Japan. It is a deciduous shrub growing to tall by broad with large heads of pink or blue flowers in summer and autumn. Common names include bigleaf hydrangea, French hydrangea, lacecap hydrangea, mophead hydrangea, penny mac and hortensia. It is widely cultivated in many parts of the world in many climates. It is not to be confused with '' H. aspera'' 'Macrophylla'. Description The term ''macrophylla'' means large- or long-leaved. The opposite leaves can grow to in length. They are simple, membranous, orbicular to elliptic and acuminate. They are generally serrated. The inflorescence of ''Hydrangea macrophylla'' is a corymb, with all flowers placed in a plane or hemisphere, or even a whole sphere in cultivated forms. Two distinct types of flowers can be identified: central, non-ornamental, pentamerous ones, and peripheral, ornamental, tetramerous ones. The latter have sterile ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phenylpropanoid
The phenylpropanoids are a diverse family of organic compounds that are synthesized by plants from the amino acids phenylalanine and tyrosine. Their name is derived from the six-carbon, aromatic phenyl group and the three-carbon propene tail of coumaric acid, which is the central intermediate in phenylpropanoid biosynthesis. From 4-coumaroyl-CoA emanates the biosynthesis of myriad natural products including lignols (precursors to lignin and lignocellulose), flavonoids, isoflavonoids, coumarins, aurones, stilbenes, catechin, and phenylpropanoids. The coumaroyl component is produced from cinnamic acid. Phenylpropanoids are found throughout the plant kingdom, where they serve as essential components of a number of structural polymers, provide protection from ultraviolet light, defend against herbivores and pathogens, and also mediate plant-pollinator interactions as floral pigments and scent compounds. Hydroxycinnamic acids Phenylalanine is first converted to cinnamic a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Carboxylic Acid
In organic chemistry, a carboxylic acid is an organic acid that contains a carboxyl group () attached to an R-group. The general formula of a carboxylic acid is or , with R referring to the alkyl, alkenyl, aryl, or other group. Carboxylic acids occur widely. Important examples include the amino acids and fatty acids. Deprotonation of a carboxylic acid gives a carboxylate anion. Examples and nomenclature Carboxylic acids are commonly identified by their trivial names. They at oftentimes have the suffix ''-ic acid''. IUPAC-recommended names also exist; in this system, carboxylic acids have an ''-oic acid'' suffix. For example, butyric acid (C3H7CO2H) is butanoic acid by IUPAC guidelines. For nomenclature of complex molecules containing a carboxylic acid, the carboxyl can be considered position one of the parent chain even if there are other substituents, such as 3-chloropropanoic acid. Alternately, it can be named as a "carboxy" or "carboxylic acid" substituent on ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
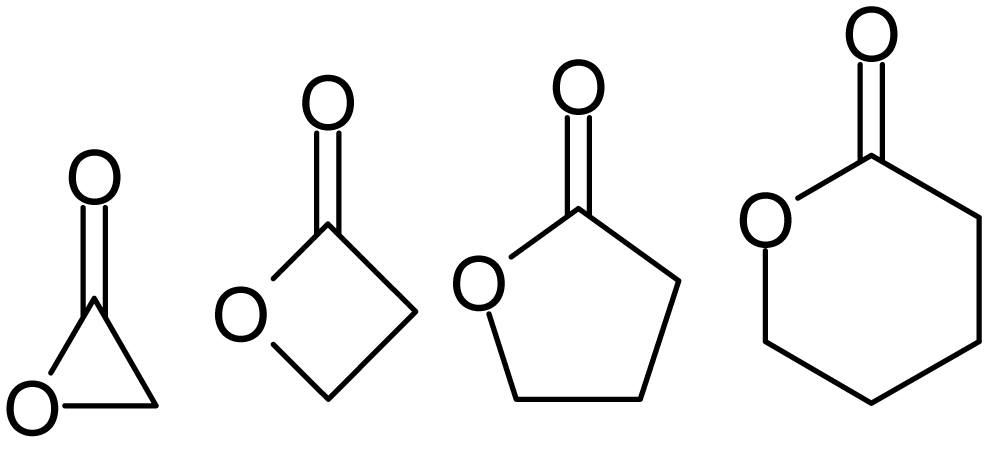
Lactone
Lactones are cyclic carboxylic esters, containing a 1-oxacycloalkan-2-one structure (), or analogues having unsaturation or heteroatoms replacing one or more carbon atoms of the ring. Lactones are formed by intramolecular esterification of the corresponding hydroxycarboxylic acids, which takes place spontaneously when the ring that is formed is five- or six-membered. Lactones with three- or four-membered rings (α-lactones and β-lactones) are very reactive, making their isolation difficult. Special methods are normally required for the laboratory synthesis of small-ring lactones as well as those that contain rings larger than six-membered. Nomenclature Lactones are usually named according to the precursor acid molecule (''aceto'' = 2 carbon atoms, ''propio'' = 3, ''butyro'' = 4, ''valero'' = 5, ''capro'' = 6, etc.), with a ''-lactone'' suffix and a Greek letter prefix that specifies the number of carbon atoms in the heterocycle — that is, the distance between the relevant -OH ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydroxylation
In chemistry, hydroxylation can refer to: *(i) most commonly, hydroxylation describes a chemical process that introduces a hydroxyl group () into an organic compound. *(ii) the ''degree of hydroxylation'' refers to the number of OH groups in a molecule. The ''pattern of hydroxylation'' refers to the location of hydroxy groups on a molecule or material. Hydroxylation reactions Synthetic hydroxylations Installing hydroxyl groups into organic compounds can be effected by various metal catalysts. Many such catalysts are biomimetic, i.e. they are inspired by or intended to mimic enzymes such as cytochrome P450. Whereas many hydroxylations insert O atoms into bonds, some reactions ''add'' OH groups to unsaturated substrates. The Sharpless dihydroxylation is such a reaction: it converts alkenes into diols. The hydroxy groups are provided by hydrogen peroxide, which adds across the double bond of alkenes. Biological hydroxylation In biochemistry, hydroxylation reactions are often ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cinnamic Acid
Cinnamic acid is an organic compound with the formula C6H5-CH=CH- COOH. It is a white crystalline compound that is slightly soluble in water, and freely soluble in many organic solvents. Classified as an unsaturated carboxylic acid, it occurs naturally in a number of plants. It exists as both a ''cis'' and a ''trans'' isomer, although the latter is more common. Occurrence and production Biosynthesis Cinnamic acid is a central intermediate in the biosynthesis of a myriad of natural products including lignols (precursors to lignin and lignocellulose), flavonoids, isoflavonoids, coumarins, aurones, stilbenes, catechin, and phenylpropanoids. Its biosynthesis involves the action of the enzyme phenylalanine ammonia-lyase (PAL) on phenylalanine. Natural occurrence It is obtained from oil of cinnamon, or from balsams such as storax. It is also found in shea butter. Cinnamic acid has a honey-like odor; it and its more volatile ethyl ester ( ethyl cinnamate) are flavor componen ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phenylalanine Ammonia-lyase
The enzyme phenylalanine ammonia lyase (EC 4.3.1.24) catalyzes the conversion of L-phenylalanine to ammonia and ''trans''-cinnamic acid.: :L-phenylalanine = ''trans''-cinnamate + NH3 Phenylalanine ammonia lyase (PAL) is the first and committed step in the phenyl propanoid pathway and is therefore involved in the biosynthesis of the polyphenol compounds such as flavonoids, phenylpropanoids, and lignin in plants. Phenylalanine ammonia lyase is found widely in plants, as well as some bacteria, yeast, and fungi, with isoenzymes existing within many different species. It has a molecular mass in the range of 270–330 kDa. The activity of PAL is induced dramatically in response to various stimuli such as tissue wounding, pathogenic attack, light, low temperatures, and hormones. PAL has recently been studied for possible therapeutic benefits in humans afflicted with phenylketonuria. It has also been used in the generation of L-phenylalanine as precursor of the sweeten ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |