|
Triazacyclononane
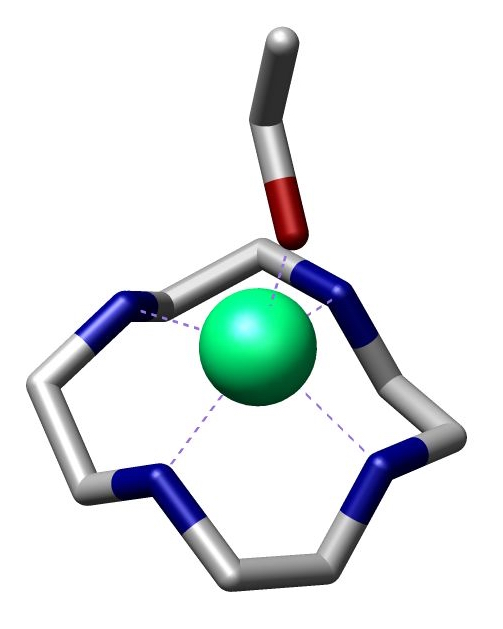
1,4,7-Triazacyclononane, known as "TACN" which is pronounced "tack-en," is an aza-crown ether with the formula (C2H4NH)3. TACN is derived, formally speaking, from cyclononane by replacing three equidistant CH2 groups with NH groups. TACN is one of the oligomers derived from aziridine, C2H4NH. Other members of the series include piperazine, C4H8(NH)2, and the cyclic tetramer 1,4,7,10-tetraazacyclododecane. Synthesis The ligand is prepared from diethylene triamine as follows by macrocyclization using ethyleneglycol ditosylate. :H2NCH2CH2NHCH2CH2NH2 + 3 TsCl → Ts(H)NCH2CH2N(Ts)CH2CHH2N(H)Ts + 3 HCl :Ts(H)NCH2CH2N(Ts)CH2CH2N(H)Ts + 2 NaOEt → Ts(Na)NCH2CH2N(Ts)CH2CH2N(Na)Ts :Ts(Na)NCHH2CH2N(Ts)CH2CH2N(Na)Ts + TsOCH2CH2OTs + → CH2CH2N(Ts)sub>3 + 2 NaOTs : CH2CH2N(Ts)sub>3 + 3 H2O → H2CH2NHsub>3 + 3 HOTs Coordination chemistry TACN is a popular tridentate ligand. It is threefold symmetric and binds to one face of an octahedron of metalloids and transition ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Trimethyltriazacyclononane
1,4,7-Trimethyl-1,4,7-triazacyclononane is the aza-crown ether with the formula (CH2CH2NCH3)3. This colorless liquid is the N-methylated derivative of triazacyclononane (TACN), a face-capping tridentate ligand that is popular in coordination chemistry A coordination complex consists of a central atom or ion, which is usually metallic and is called the ''coordination centre'', and a surrounding array of bound molecules or ions, that are in turn known as ''ligands'' or complexing agents. Many .... Although TACN is known for forming 2:1 "sandwich" complexes with many metal ions, corresponding 2:1 complexes of Me3TACN are only known for Ag+, Na+, and K+. This effect is mainly due to the greater bulk of Me3TACN, which requires ions with a larger ionic radius to accommodate two ligands. Several related derivatives have been prepared with diverse substituents on nitrogen. References {{DEFAULTSORT:Trimethyl-1,4,7-triazacyclononane, 1,4,7- Polyamines Chelating agents Mac ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Aza-crown Ether
In organic chemistry, an aza-crown ether is an aza analogue of a crown ether (cyclic polyether). That is, it has a nitrogen atom (amine linkage, or ) in place of each oxygen atom (ether linkage, ) around the ring. While the parent crown ethers have the formulae , the parent ''aza''-crown ethers have the formulae , where n = 3, 4, 5, 6. Well-studied aza crowns include triazacyclononane (n = 3), cyclen (n = 4), and hexaaza-18-crown-6 (n = 6). File:Me3TACN.png, 1,4,7-Trimethyl-1,4,7-triazacyclononane, a tridentate ligand used in coordination chemistry. File:Cyclam.png, Cyclam is a tetraaza crown ether with alternating and linkers between amine centers. File:Plerixafor.svg, Plerixafor, a derivative of cyclam, is used to treat lymphoma and multiple myeloma. File:Cryptand.svg, 2.2.2-Cryptand is an aza-crown of the mixed ether-amine variety. Synthesis The synthesis of aza crown ethers are subject to the challenges associated with the preparation of macrocycles. The 18-membered ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Aziridine
Aziridine is an organic compound consisting of the three-membered heterocycle . It is a colorless, toxic, volatile liquid that is of significant practical interest. Aziridine was discovered in 1888 by the chemist Siegmund Gabriel. Its derivatives, also referred to as aziridines, are of broader interest in medicinal chemistry. Structure The bond angles in aziridine are approximately 60°, considerably less than the normal hydrocarbon bond angle of 109.5°, which results in angle strain as in the comparable cyclopropane and ethylene oxide molecules. A banana bond model explains bonding in such compounds. Aziridine is less basic than acyclic aliphatic amines, with a pKa of 7.9 for the conjugate acid, due to increased s character of the nitrogen free electron pair. Angle strain in aziridine also increases the barrier to nitrogen inversion. This barrier height permits the isolation of separate ''invertomers'', for example the ''cis'' and ''trans'' invertomers of ''N''-chloro- ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Piperazine
Piperazine () is an organic compound that consists of a six-membered ring containing two nitrogen atoms at opposite positions in the ring. Piperazine exists as small alkaline deliquescent crystals with a saline taste. The piperazines are a broad class of chemical compounds, many with important pharmacological properties, which contain a core piperazine functional group. Origin and naming Piperazines were originally named because of their chemical similarity with piperidine, part of the structure of piperine in the black pepper plant (''Piper nigrum''). The -az- infix added to "piperazine" refers to the extra nitrogen atom, compared to piperidine. It is important to note, however, that piperazines are ''not'' derived from plants in the '' Piper'' genus. Chemistry Piperazine is freely soluble in water and ethylene glycol, but insoluble in diethyl ether. It is a weak base with two pKb of 5.35 and 9.73 at 25 °C.; the pH of a 10% aqueous solution of piperazine is 10.8–11.8. Pi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cyclen
Cyclen (1,4,7,10-tetraazacyclododecane) is a aza-crown ether with the formula (CH2CH2NH)4. It is a white solid. Synthesis Some syntheses exploit the Thorpe-Ingold effect to facilitate ring-formation. Illustrative is the reaction of the deprotonated tosylamides with ditosylates: :TsN(CH2CH2NTsNa)2 + TsN(CH2CH2OTs)2 → (TsNCH2CH2)4 The resulting macrocycle can be deprotected with strong acid. Base gives the tetramine. High dilution conditions result in a low reaction rate penalty and this disadvantage is removed in an alternative procedure starting from triethylenetetraamine and dithiooxamide to a bisamidine – also a bis(imidazoline) – followed by reduction and ring expansion with DIBAL. : In one study cyclen is covalently bonded through a propylene molecular spacer to adenine and chelated with zinc diperchlorate. This complex is able to selectively bind uracil and uridine in a 1:2 ratio both through the adenine part and cyclen part of the molecule as evi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Diethylene Triamine
Diethylenetriamine (abbreviated Dien or DETA) and also known as 2,2’-Iminodi(ethylamine)) is an organic compound with the formula HN(CH2CH2NH2)2. This colourless hygroscopic liquid is soluble in water and polar organic solvents, but not simple hydrocarbons. Diethylenetriamine is structural analogue of diethylene glycol. Its chemical properties resemble those for ethylene diamine, and it has similar uses. It is a weak base and its aqueous solution is alkaline. DETA is a byproduct of the production of ethylenediamine from ethylene dichloride. Reactions and uses Diethylenetriamine is a common curing agent for epoxy resins in epoxy adhesives and other thermosets. It is N-alkylated upon reaction with epoxide groups forming crosslinks. In coordination chemistry, it serves as a tridentate ligand forming complexes such as Co(dien)(NO2)3. Like some related amines, it is used in oil industry for the extraction of acid gas. Like ethylenediamine, DETA can also be used to sensitize nitr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chemische Berichte
''Chemische Berichte'' (usually abbreviated as ''Ber.'' or ''Chem. Ber.'') was a German-language scientific journal of all disciplines of chemistry founded in 1868. It was one of the oldest scientific journals in chemistry, until it merged with ''Recueil des Travaux Chimiques des Pays-Bas'' to form ''Chemische Berichte/Recueil'' in 1997. ''Chemische Berichte/Recueil'' was then merged with other European journals in 1998 to form ''European Journal of Inorganic Chemistry''. History Founded in 1868 as ''Berichte der Deutschen Chemischen Gesellschaft'' (, CODEN BDCGAS), it operated under this title until 1928 (Vol. 61). The journal was then split into: * ''Berichte der Deutschen Chemischen Gesellschaft, A: Vereins-Nachrichten'' (, CODEN BDCAAS), and * ''Berichte der Deutschen Chemischen Gesellschaft, B: Abhandlungen'' (, CODEN BDCBAD). Vol. 78 and 79 (1945–1946) were omitted and not published due to World War II. The journal was renamed ''Chemische Berichte'' (, CODEN CHBEAM) in 19 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Epoxidation
In organic chemistry, an epoxide is a cyclic ether () with a three-atom ring. This ring approximates an equilateral triangle, which makes it strained, and hence highly reactive, more so than other ethers. They are produced on a large scale for many applications. In general, low molecular weight epoxides are colourless and nonpolar, and often volatile. Nomenclature A compound containing the epoxide functional group can be called an epoxy, epoxide, oxirane, and ethoxyline. Simple epoxides are often referred to as oxides. Thus, the epoxide of ethylene (C2H4) is ethylene oxide (C2H4O). Many compounds have trivial names; for instance, ethylene oxide is called "oxirane". Some names emphasize the presence of the epoxide functional group, as in the compound ''1,2-epoxyheptane'', which can also be called ''1,2-heptene oxide''. A polymer formed from epoxide precursors is called an '' epoxy'', but such materials do not contain epoxide groups (or contain only a few residual epoxy gr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Alkene
In organic chemistry, an alkene is a hydrocarbon containing a carbon–carbon double bond. Alkene is often used as synonym of olefin, that is, any hydrocarbon containing one or more double bonds.H. Stephen Stoker (2015): General, Organic, and Biological Chemistry'. 1232 pages. Two general types of monoalkenes are distinguished: terminal and internal. Also called α-olefins, terminal alkenes are more useful. However, the International Union of Pure and Applied Chemistry (IUPAC) recommends using the name "alkene" only for acyclic hydrocarbons with just one double bond; alkadiene, alkatriene, etc., or polyene for acyclic hydrocarbons with two or more double bonds; cycloalkene, cycloalkadiene, etc. for cyclic ones; and "olefin" for the general class – cyclic or acyclic, with one or more double bonds. Acyclic alkenes, with only one double bond and no other functional groups (also known as mono-enes) form a homologous series of hydrocarbons with the general formula with '' ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Styrene
Styrene () is an organic compound with the chemical formula C6H5CH=CH2. This derivative of benzene is a colorless oily liquid, although aged samples can appear yellowish. The compound evaporates easily and has a sweet smell, although high concentrations have a less pleasant odor. Styrene is the precursor to polystyrene and several copolymers. Approximately 25 million tonnes of styrene were produced in 2010, increasing to around 35 million tonnes by 2018. Natural occurrence Styrene is named after storax balsam (often commercially sold as ''styrax''), the resin of Liquidambar trees of the Altingiaceae plant family. Styrene occurs naturally in small quantities in some plants and foods (cinnamon, coffee beans, balsam tree (other), balsam trees and peanuts) and is also found in coal tar. History In 1839, the German apothecary Eduard Simon isolated a volatile liquid from the resin (called ''storax'' or ''styrax'' (Latin)) of the Liquidambar styraciflua, American sweetgu ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Methanol
Methanol (also called methyl alcohol and wood spirit, amongst other names) is an organic chemical and the simplest aliphatic alcohol, with the formula C H3 O H (a methyl group linked to a hydroxyl group, often abbreviated as MeOH). It is a light, volatile, colourless, flammable liquid with a distinctive alcoholic odour similar to that of ethanol (potable alcohol). A polar solvent, methanol acquired the name wood alcohol because it was once produced chiefly by the destructive distillation of wood. Today, methanol is mainly produced industrially by hydrogenation of carbon monoxide. Methanol consists of a methyl group linked to a polar hydroxyl group. With more than 20 million tons produced annually, it is used as a precursor to other commodity chemicals, including formaldehyde, acetic acid, methyl tert-butyl ether, methyl benzoate, anisole, peroxyacids, as well as a host of more specialised chemicals. Occurrence Small amounts of methanol are present in normal, healthy hu ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ethyleneamines
Ethyleneamines are a class of amine compounds containing ethylene (-CH2CH2-) linkages between amine groups. These compounds are generally colorless, low-viscosity liquids with a fishy amine odor. They are primarily used as building block chemicals and in epoxy resin curing agent chemistry. Production There are two main routes for the production of ethyleneamines, the reaction between ethylene dichloride and ammonia, and the reductive amination of monethanolamine. World capacity of ethyleneamines for the year 2001 was estimated to be 385,000 tonnes/year, with the majority comprising the ethylene dichloride route. In the ethylene dichloride route, the initial product of this reaction is ethylenediamine. In the presence of excess ethylene dichloride, the initial ethyleneamine is extended by one ethylene unit. The terminal alkyl chloride reacts with ammonia to give the amine, and the polyamine chain can be extended further in this fashion. Addition of a polyamine to the initial react ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
ML3.png)
6) (BIGNOU01).png)