|
Transition Metal Carboxylate Complex
Transition metal carboxylate complexes are coordination complexes with carboxylate (RCO2−) ligands. Reflecting the diversity of carboxylic acids, the inventory of metal carboxylates is large. Many are useful commercially, and many have attracted intense scholarly scrutiny. Carboxylates exhibit a variety of coordination modes, most common are κ1- (O-monodentate), κ2 (O,O-bidentate), and bridging. Acetate and related monocarboxylates Structure and bonding Carboxylates bind to single metals by one or both oxygen atoms, respectively κ1- and κ2-. In terms of electron counting, κ1- and κ2- are "X", a pseudohalide, and "L-X ligands", i.e. a combination of a Lewis base (L) and a pseudohalide (X). Carboxylates are classified as hard ligands, in HSAB theory. File:BasicFeacetate.png, Basic ferric acetate File:Ag2(OAc)2.png, Silver acetate File:BasicZnAcetate.png, Basic zinc acetate File:Mo2(OAc)4.svg, Molybdenum(II) acetate, illustrating the Mo-Mo quadruple bond. File:DasCuban ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nickel(II)-acetate-tetrahydrate-3D-balls
Nickel is a chemical element with symbol Ni and atomic number 28. It is a silvery-white lustrous metal with a slight golden tinge. Nickel is a hard and ductile transition metal. Pure nickel is chemically reactive but large pieces are slow to react with air under standard conditions because a passivation layer of nickel oxide forms on the surface that prevents further corrosion. Even so, pure native nickel is found in Earth's crust only in tiny amounts, usually in ultramafic rocks, and in the interiors of larger nickel–iron meteorites that were not exposed to oxygen when outside Earth's atmosphere. Meteoric nickel is found in combination with iron, a reflection of the origin of those elements as major end products of supernova nucleosynthesis. An iron–nickel mixture is thought to compose Earth's outer and inner cores. Use of nickel (as natural meteoric nickel–iron alloy) has been traced as far back as 3500 BCE. Nickel was first isolated and classified as an e ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Platinum(II) Acetate
Platinum(II) acetate is a purple-colored coordination complex. The complex adopts an unusual structure consisting of a square array of Pt atoms. Preparation Several syntheses of platinum(II) acetate have been reported. Geoffrey Wilkinson ''et al.'' reported a synthesis from sodium hexahydroxyplatinate, nitric acid, and acetic acid. This intermediate solution was reducted with formic acid. The procedure is not highly reproducible. Alternatively, the complex can be prepared by the reaction of silver acetate with platinum(II) chloride. Structure According to X-ray crystallography X-ray crystallography is the experimental science determining the atomic and molecular structure of a crystal, in which the crystalline structure causes a beam of incident X-rays to diffract into many specific directions. By measuring the angles ..., the complex is tetrameric, in contrast to the trimeric palladium analog. The four platinum atoms form a square cluster, with eight bridging acetate liga ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Metal Organic Framework
A metal (from Greek μέταλλον ''métallon'', "mine, quarry, metal") is a material that, when freshly prepared, polished, or fractured, shows a lustrous appearance, and conducts electricity and heat relatively well. Metals are typically ductile (can be drawn into wires) and malleable (they can be hammered into thin sheets). These properties are the result of the ''metallic bond'' between the atoms or molecules of the metal. A metal may be a chemical element such as iron; an alloy such as stainless steel; or a molecular compound such as polymeric sulfur nitride. In physics, a metal is generally regarded as any substance capable of conducting electricity at a temperature of absolute zero. Many elements and compounds that are not normally classified as metals become metallic under high pressures. For example, the nonmetal iodine gradually becomes a metal at a pressure of between 40 and 170 thousand times atmospheric pressure. Equally, some materials regarded as metals ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Zinc Diacetate
Zinc acetate is a Salt (chemistry), salt with the chemical formula, formula Zn(CH3CO2)2, which commonly occurs as the Hydrate, dihydrate Zn(CH3CO2)2·2H2O. Both the hydrate and the anhydrous forms are colorless solids that are used as dietary supplements. When used as a food additive, it has the E number E650. Uses Zinc acetate is a component of some medicines, e.g., Zinc and the common cold, lozenges for treating the common cold. Zinc acetate can also be used as a dietary supplement. As an oral daily supplement it is used to inhibit the body's absorption of copper as part of the treatment for Wilson's disease. Zinc acetate is also sold as an astringent in the form of an ointment, a topical lotion, or combined with an antibiotic such as erythromycin for the topical treatment of acne. It is commonly sold as a topical anti-itch ointment. Zinc acetate is used as the catalyst for the industrial production of vinyl acetate from acetylene: Approximately 1/3 of the worlds production u ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Acid Anhydride
An acid anhydride is a type of chemical compound derived by the removal of water molecules from an acid. In organic chemistry, organic acid anhydrides contain the functional group R(CO)O(CO)R'. Organic acid anhydrides often form when one equivalent of water is removed from two equivalents of an organic acid in a dehydration reaction. In inorganic chemistry, an acid anhydride refers to an acidic oxide, an oxide that reacts with water to form an oxyacid (an inorganic acid that contains oxygen or carbonic acid), or with a base to form a salt. Nomenclature The nomenclature of organic acid anhydrides is derived from the names of the constituent carboxylic acids which underwent dehydration to form the compound. In symmetrical acid anhydrides, where only one constituent carboxylic acid was used to form the compound (such as the dehydration of propanoic acid, 2CH3CH2COOH → CH3CH2C(O)OC(O)CH2CH3 + H2O), only the prefix of the original carboxylic acid is used and the suffix "anhydride" ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Concerted Metalation Deprotonation
Concerted metalation-deprotonation (CMD) is a mechanistic pathway through which transition-metal catalyzed C–H activation reactions can take place. In a CMD pathway, the C–H bond of the substrate is cleaved and the new C–Metal bond forms through a single transition state. This process does not go through a metal species that is bound to the cleaved hydrogen atom. Instead, a carboxylate or carbonate base deprotonates the substrate. The first proposal of a concerted metalation deprotonation pathway was by S. Winstein and T. G. Traylor in 1955 for the acetolysis of diphenylmercury. It was found to be the lowest energy transition state in a number of computational studies, was experimentally confirmed through NMR experiments, and has been hypothesized to occur in mechanistic studies. While there are a number of different possible mechanisms for C–H activation, a CMD pathway is common for high valent, late transition metals like PdII, RhIII, IrIII, and RuII. The C–H bonds that ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dimolybdenum Tetraacetate
Molybdenum(II) acetate is a coordination compound with the formula Mo2(O2CCH3)4. It is a yellow, diamagnetic, air-stable solid that is slightly soluble in organic solvents. Molybdenum(II) acetate is an iconic example of a compound with a metal-metal quadruple bond.Girolami, G. S.; Rauchfuss, T. B. and Angelici, R. J., "Synthesis and Technique in Inorganic Chemistry third edition", University Science Books: Mill Valley, CA, 1999, Structure and bonding It adopts the same Chinese lantern structure as related acetate dimers such as rhodium(II) acetate, copper(II) acetate, and chromium(II) acetate. Each Mo(II) center in Mo2(O2CCH3)4 has four d valence electrons. These eight d-electrons form one σ, two π bonds, and one δ bond, creating a bonding electron configuration of σ2π4δ2. Each of these bonds are formed by the overlapping of pairs of d orbitals.Blaudeau, J. P.; Pitzer, R. M. “ Ab Initio Studies of Ligand Effects on the Metal-Metal Bond in Dimolybdenum Complexes” J.P ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Octachlorodimolybdate
Potassium octachlorodimolybdate (systematically named potassium bis(tetrachloromolybdate)(''Mo''–''Mo'')(4−)) is an inorganic compound with the chemical formula . It is known as a red-coloured, microcrystalline solid. The anion is of historic interest as one of the earliest illustrations of a quadruple bonding. The salt is usually obtained as the pink-coloured dihydrate. The compound is prepared in two steps from molybdenum hexacarbonyl: : : The reaction of the acetate with HCl was first described as providing trimolybdenum compounds, but subsequent crystallographic analysis confirmed that the salt contains the anion, with D4h symmetry, in which the two Mo atoms are linked by a quadruple bond. Each Mo atom is bounded with four ligands by a single bond. Each group is an regular square pyramid, with Mo atom at the apex, and four Cl atoms at the vertices of the square base of the pyramid A pyramid (from el, πυραμίς ') is a structure whose outer surfaces are ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
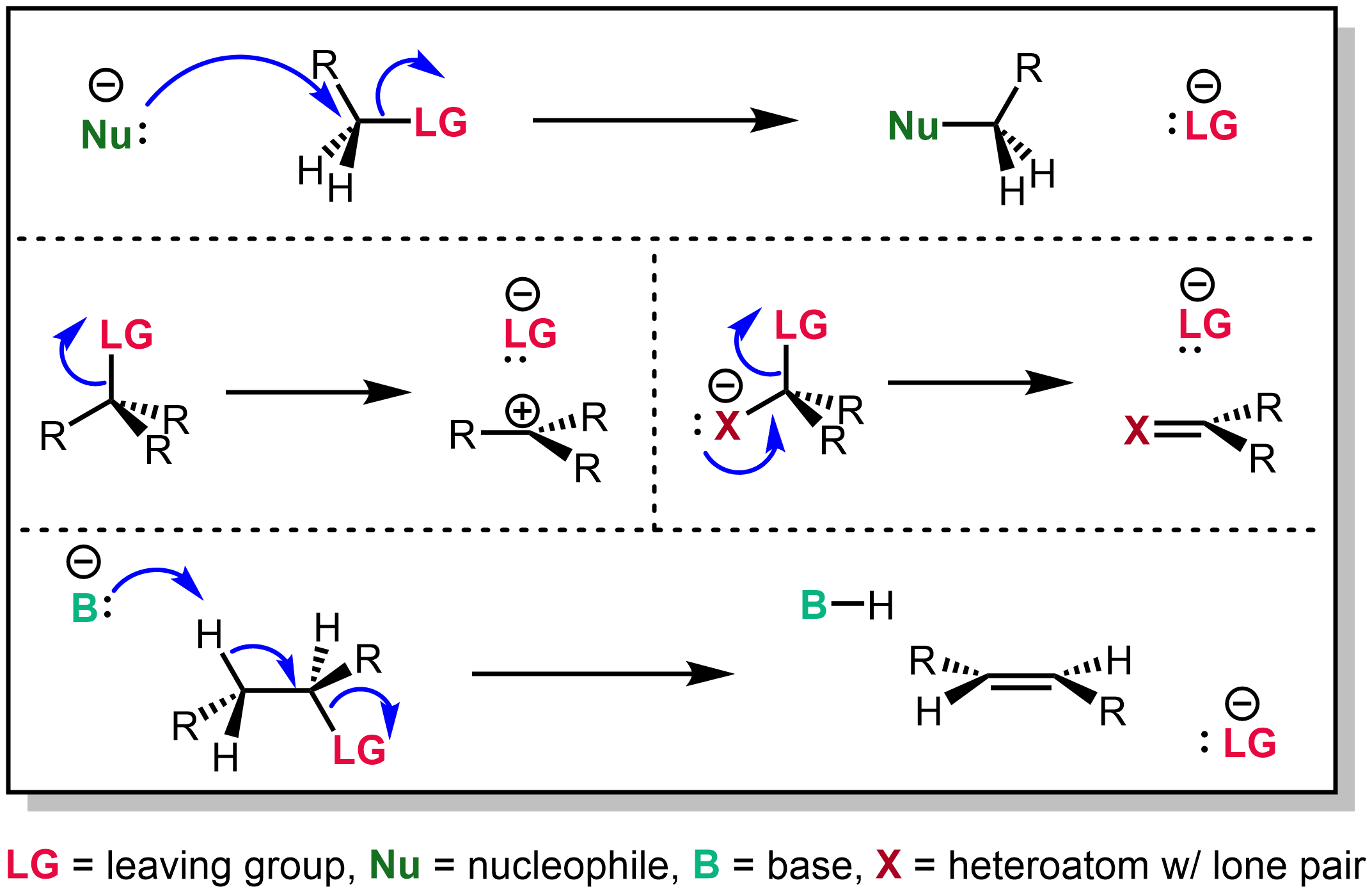
Leaving Group
In chemistry, a leaving group is defined by the IUPAC as an atom or group of atoms that detaches from the main or residual part of a substrate during a reaction or elementary step of a reaction. However, in common usage, the term is often limited to a fragment that departs with a pair of electrons in heterolytic bond cleavage. In this usage, a leaving group is a less formal but more commonly used synonym of the term '' nucleofuge''. In this context, leaving groups are generally anions or neutral species, departing from a neutral or cationic substrates, respectively, though in rare cases, cations leaving from a dicationic substrate are also known. A species' ability to serve as a leaving group depends on its ability to stabilize the additional electron density that results from bond heterolysis. Common anionic leaving groups are halides such as Cl−, Br−, and I−, and sulfonate esters such as tosylate (TsO−), while water (H2O), alcohols (HOR), and amines (R3N) are common neutr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Salt Metathesis Reaction
A salt metathesis reaction, sometimes called a double displacement reaction, is a chemical process involving the exchange of bonds between two reacting chemical species which results in the creation of products with similar or identical bonding affiliations. This reaction is represented by the general scheme: :AB + CD -> AD + CB The bond between the reacting species can be either ionic or covalent. Classically, these reactions result in the precipitation of one product. In older literature, the term double decomposition is frequently encountered. The term double decomposition is more specifically used when at least one of the substances does not dissolve in the solvent, as the ligand or ion exchange takes place in the solid state of the reactant. For example: :AX(aq) + BY(s) → AY(aq) + BX(s). Types of reactions Counterion exchange Salt metathesis is a common technique for exchanging counterions. The choice of reactants is guided by a solubility chart or lattice energy. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Oxidative Addition
Oxidative addition and reductive elimination are two important and related classes of reactions in organometallic chemistry. Oxidative addition is a process that increases both the oxidation state and coordination number of a metal centre. Oxidative addition is often a step in catalytic cycles, in conjunction with its reverse reaction, reductive elimination. Role in transition metal chemistry For transition metals, oxidative reaction results in the decrease in the d''n'' to a configuration with fewer electrons, often 2e fewer. Oxidative addition is favored for metals that are (i) basic and/or (ii) easily oxidized. Metals with a relatively low oxidation state often satisfy one of these requirements, but even high oxidation state metals undergo oxidative addition, as illustrated by the oxidation of Pt(II) with chlorine: : tCl4sup>2− + Cl2 → tCl6sup>2− In classical organometallic chemistry, the formal oxidation state of the metal and the electron count of the complex both in ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ethylhexanoate
2-Ethylhexanoic acid is the organic compound with the formula CH3(CH2)3CH(C2H5)CO2H. It is a carboxylic acid that is widely used to prepare lipophilic metal derivatives that are soluble in nonpolar organic solvents. 2-Ethylhexanoic acid is a colorless viscous oil. It is supplied as a racemic mixture. Production 2-Ethylhexanoic acid is produced industrially from propylene, which is hydroformylated to give butyraldehyde. Aldol condensation of the aldehyde gives 2-ethylhexenal, which is hydrogenated to 2-ethylhexanal. Oxidation of this aldehyde gives the carboxylic acid. Metal ethylhexanoates 2-Ethylhexanoic acid forms compounds with metal cations that have stoichiometry as metal acetates. These ethylhexanoate complexes are used in organic and industrial chemical synthesis. They function as catalysts in polymerizations as well as for oxidation reactions as "oil drying agents." They are highly soluble in nonpolar solvents. These metal complexes are often described as sal ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |



-3D-balls.png)

2.jpg)