|
Threohydrobupropion
Threohydrobupropion (developmental code names BW 494, BW A494U) is a substituted amphetamine derivativeŌĆöspecifically a ╬▓-hydroxyamphetamineŌĆöand a major active metabolite of the antidepressant drug bupropion (Wellbutrin). Bupropion is a norepinephrineŌĆōdopamine reuptake inhibitor and nicotinic acetylcholine receptor negative allosteric modulator, with its metabolites contributing substantially to its activities. Threohydrobupropion exists as two isomers, (1''R'',2''R'')-threohydrobupropion and (1''S'',2''S'')-threohydrobupropion. Other metabolites of bupropion include hydroxybupropion and erythrohydrobupropion. Information on the pharmacological actions of threohydrobupropion is scarce. In any case, it is about 20% as pharmacologically potent as bupropion and in the range of 20 to 50% as potent as bupropion in mouse models of depression. Moreover, threohydrobupropion has been reported to weakly inhibit the reuptake of norepinephrine, dopamine, and serotonin with rat o ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Erythrohydrobupropion
Erythrohydrobupropion (developmental code names BW 287, BW 17U) is a substituted amphetamine derivativeŌĆöspecifically a ╬▓-hydroxyamphetamineŌĆöand a minor active metabolite of the antidepressant drug bupropion (Wellbutrin). Bupropion is a norepinephrineŌĆōdopamine reuptake inhibitor and nicotinic acetylcholine receptor negative allosteric modulator, with its metabolites contributing substantially to its activities. Erythrohydrobupropion exists as two isomers, (1''R'',2''S'')-erythrohydrobupropion and (1''S'',2''R'')-erythrohydrobupropion. Other metabolites of bupropion include hydroxybupropion and threohydrobupropion. Information on the pharmacological actions of erythrohydrobupropion is scarce. In any case, it is about 20% as pharmacologically potent as bupropion and in the range of 20 to 50% as potent as bupropion in mouse models of depression. It circulates at similar concentrations as bupropion during bupropion therapy. Conversely, two other metabolites, hydroxybupropio ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
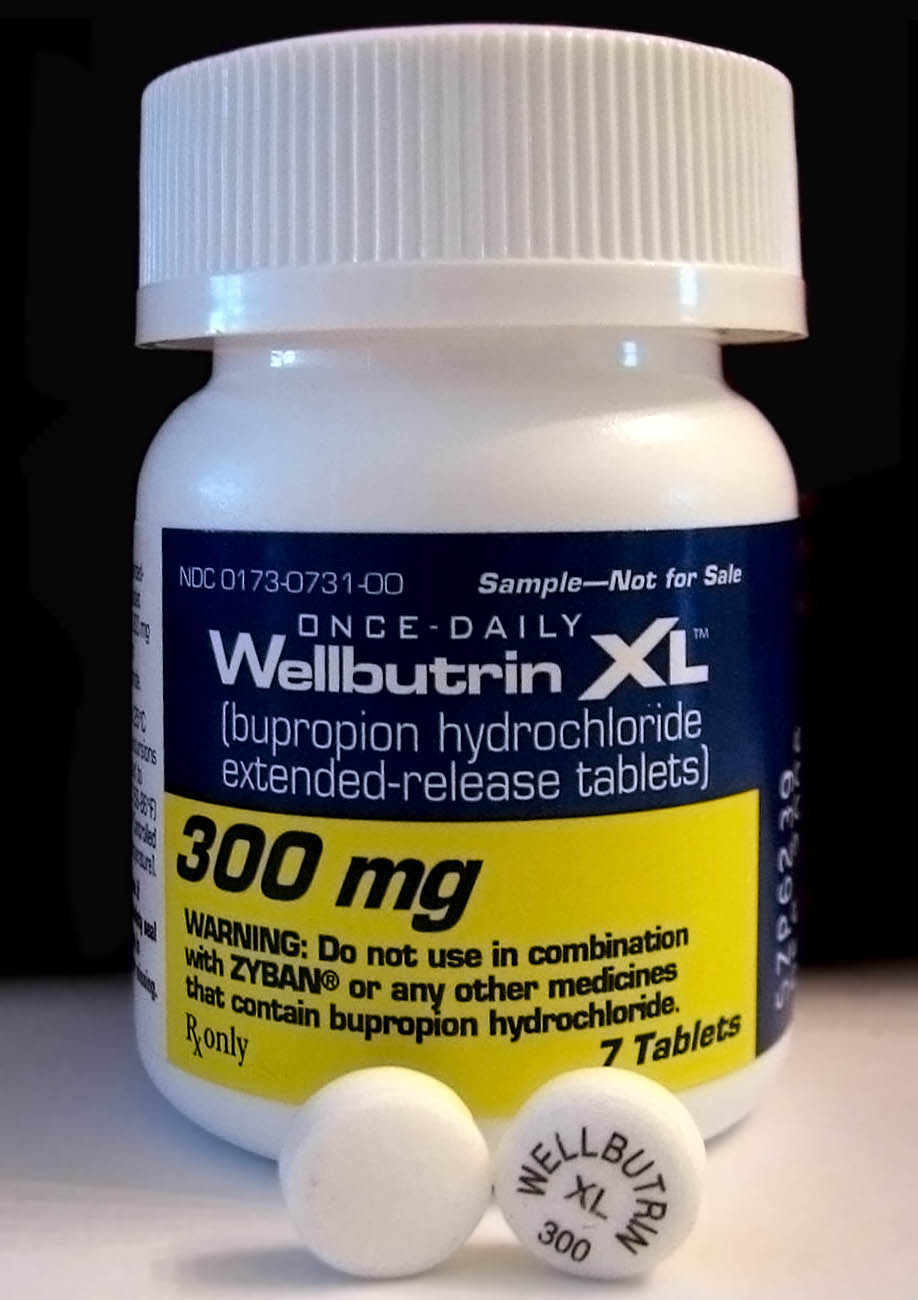
Bupropion
Bupropion, sold under the brand names Wellbutrin and Zyban among others, is an atypical antidepressant primarily used to treat major depressive disorder and to support smoking cessation. It is also popular as an add-on medication in the cases of "incomplete response" to the first-line selective serotonin reuptake inhibitor (SSRI) antidepressant. Bupropion has several features that distinguish it from other antidepressants: it does not usually cause sexual dysfunction; it is not associated with weight gain and sleepiness, and it is more effective than SSRIs at improving symptoms of hypersomnia and fatigue. Bupropion does, however, carry a much higher risk of seizure than many other antidepressants and extreme caution must be taken in patients with a history of seizure disorder. Common adverse effects of bupropion with the greatest difference from placebo are dry mouth, nausea, constipation, insomnia, anxiety, tremor, and excessive sweating. Raised blood pressure is notable. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydroxybupropion
Hydroxybupropion (code name BW 306U), or 6-hydroxybupropion, is the major active metabolite of the antidepressant and smoking cessation drug bupropion. It is formed from bupropion by the liver enzyme CYP2B6 during first-pass metabolism. With oral bupropion treatment, hydroxybupropion is present in plasma at area under the curve concentrations that are as many as 16ŌĆō20 times greater than those of bupropion itself, demonstrating extensive conversion of bupropion into hydroxybupropion in humans. As such, hydroxybupropion is likely to play a very important role in the effects of oral bupropion, which could accurately be thought of as functioning largely as a prodrug to hydroxybupropion. Other metabolites of bupropion besides hydroxybupropion include threohydrobupropion and erythrohydrobupropion. Pharmacology Pharmacodynamics Compared to bupropion, hydroxybupropion is similar in its potency as a norepinephrine reuptake inhibitor ( IC50 = 1.7 ╬╝M), but is substantially weaker a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ketone
In organic chemistry, a ketone is a functional group with the structure RŌĆōC(=O)ŌĆōR', where R and R' can be a variety of carbon-containing substituents. Ketones contain a carbonyl group ŌĆōC(=O)ŌĆō (which contains a carbon-oxygen double bond C=O). The simplest ketone is acetone (where R and R' is methyl), with the formula . Many ketones are of great importance in biology and in industry. Examples include many sugars (ketoses), many steroids (e.g., testosterone), and the solvent acetone. Nomenclature and etymology The word ''ketone'' is derived from ''Aketon'', an old German word for ''acetone''. According to the rules of IUPAC nomenclature, ketone names are derived by changing the suffix ''-ane'' of the parent alkane to ''-anone''. Typically, the position of the carbonyl group is denoted by a number, but traditional nonsystematic names are still generally used for the most important ketones, for example acetone and benzophenone. These nonsystematic names are considered reta ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Redox
Redox (reductionŌĆōoxidation, , ) is a type of chemical reaction in which the oxidation states of substrate (chemistry), substrate change. Oxidation is the loss of Electron, electrons or an increase in the oxidation state, while reduction is the gain of electrons or a decrease in the oxidation state. There are two classes of redox reactions: * ''Electron-transfer'' ŌĆō Only one (usually) electron flows from the reducing agent to the oxidant. This type of redox reaction is often discussed in terms of redox couples and electrode potentials. * ''Atom transfer'' ŌĆō An atom transfers from one substrate to another. For example, in the rusting of iron, the oxidation state of iron atoms increases as the iron converts to an oxide, and simultaneously the oxidation state of oxygen decreases as it accepts electrons released by the iron. Although oxidation reactions are commonly associated with the formation of oxides, other chemical species can serve the same function. In hydrogen ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Plasma Protein Binding
Plasma protein binding refers to the degree to which medications attach to proteins within the blood. A drug's efficiency may be affected by the degree to which it binds. The less bound a drug is, the more efficiently it can traverse or diffuse through cell membranes. Common blood proteins that drugs bind to are human serum albumin, lipoprotein, glycoprotein, and ╬▒, ╬▓ŌĆÜ and ╬│ globulins. Binding (drug distribution) A drug in blood exists in two forms: bound and unbound. Depending on a specific drug's affinity for plasma proteins, a proportion of the drug may become bound to the proteins, with the remainder being unbound. If the protein binding is reversible, then a chemical equilibrium will exist between the bound and unbound states, such that: :Protein + drug Ōćī Protein-drug complex Notably, it is the unbound fraction which exhibits pharmacologic effects. It is also the fraction that may be metabolized and/or excreted. For example, the "fraction bound" of the anticoagu ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Alpha-3 Beta-4 Nicotinic Receptor
The alpha-3 beta-4 nicotinic receptor, also known as the ╬▒3╬▓4 receptor and the ganglion-type nicotinic receptor,Pharmacology, (Rang, Dale, Ritter & Moore, , 5th ed., Churchill Livingstone 2003) p. 138. is a type of nicotinic acetylcholine receptor, consisting of ╬▒3 and ╬▓4 subunits. It is located in the autonomic ganglia and adrenal medulla, where activation yields post- and/or presynaptic excitation, mainly by increased Na+ and K+ permeability. As with other nicotinic acetylcholine receptors, the ╬▒3╬▓4 receptor is pentameric ╬▒3)m(╬▓4)n where m + n = 5 The exact subunit stoichiometry is not known and it is possible that more than one functional ╬▒3╬▓4 receptor assembles in vivo with varying subunit stoichiometries. Ligands which inhibit the ╬▒3╬▓4 receptor have been shown to modulate drug-seeking behavior, making ╬▒3╬▓4 a promising target for the development of novel antiaddictive agents. Ligands Agonists * Acetylcholine (endogenous neurotransmitter that binds non-s ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Monoamine Reuptake Inhibition
A monoamine reuptake inhibitor (MRI) is a drug that acts as a reuptake inhibitor of one or more of the three major monoamine neurotransmitters serotonin, norepinephrine, and dopamine by blocking the action of one or more of the respective monoamine transporters (MATs), which include the serotonin transporter (SERT), norepinephrine transporter (NET), and dopamine transporter (DAT). This in turn results in an increase in the synaptic concentrations of one or more of these neurotransmitters and therefore an increase in monoaminergic neurotransmission. Uses The majority of currently approved antidepressants act predominantly or exclusively as MRIs, including the selective serotonin reuptake inhibitors (SSRIs), serotoninŌĆōnorepinephrine reuptake inhibitors (SNRIs), and almost all of the tricyclic antidepressants (TCAs). Many psychostimulants used either in the treatment of or as appetite suppressants in the treatment of obesity also behave as MRIs, although notably amphetamine (and me ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Serotonin
Serotonin () or 5-hydroxytryptamine (5-HT) is a monoamine neurotransmitter. Its biological function is complex and multifaceted, modulating mood, cognition, reward, learning, memory, and numerous physiological processes such as vomiting and vasoconstriction. Approximately 90% of the serotonin that the body produces is in the intestinal tract. Biochemically, the indoleamine molecule derives from the amino acid tryptophan, via the (rate-limiting) Tryptophan hydroxylase, hydroxylation of the 5 position on the ring (forming the intermediate 5-Hydroxytryptophan, 5-hydroxytryptophan), and then Aromatic L-amino acid decarboxylase, decarboxylation to produce serotonin. Serotonin is primarily found in the enteric nervous system located in the human gastrointestinal tract, gastrointestinal tract (GI tract). However, it is also produced in the central nervous system (CNS), specifically in the raphe nuclei located in the brainstem, Merkel cells located in the skin, Neuroendocrine cell#Pulmo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dopamine
Dopamine (DA, a contraction of 3,4-dihydroxyphenethylamine) is a neuromodulatory molecule that plays several important roles in cells. It is an organic chemical of the catecholamine and phenethylamine families. Dopamine constitutes about 80% of the catecholamine content in the brain. It is an amine synthesized by removing a carboxyl group from a molecule of its precursor chemical, L-DOPA, which is synthesized in the brain and kidneys. Dopamine is also synthesized in plants and most animals. In the brain, dopamine functions as a neurotransmitterŌĆöa chemical released by neurons (nerve cells) to send signals to other nerve cells. Neurotransmitters are synthesized in specific regions of the brain, but affect many regions systemically. The brain includes several distinct dopamine pathways, one of which plays a major role in the motivational component of reward-motivated behavior. The anticipation of most types of rewards increases the level of dopamine in the brain, and ma ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Norepinephrine
Norepinephrine (NE), also called noradrenaline (NA) or noradrenalin, is an organic chemical in the catecholamine family that functions in the brain and body as both a hormone and neurotransmitter. The name "noradrenaline" (from Latin '' ad'', "near", and ''ren'', "kidney") is more commonly used in the United Kingdom, whereas "norepinephrine" (from Ancient Greek ß╝ÉŽĆß┐É╠ü (''ep├Ł''), "upon", and ╬Į╬ĄŽåŽüŽīŽé (''nephr├│s''), "kidney") is usually preferred in the United States. "Norepinephrine" is also the international nonproprietary name given to the drug. Regardless of which name is used for the substance itself, parts of the body that produce or are affected by it are referred to as noradrenergic. The general function of norepinephrine is to mobilize the brain and body for action. Norepinephrine release is lowest during sleep, rises during wakefulness, and reaches much higher levels during situations of stress or danger, in the so-called fight-or-flight response. In th ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Reuptake
Reuptake is the reabsorption of a neurotransmitter by a neurotransmitter transporter located along the plasma membrane of an axon terminal (i.e., the pre-synaptic neuron at a synapse) or glial cell after it has performed its function of transmitting a neural impulse. Reuptake is necessary for normal synaptic physiology because it allows for the recycling of neurotransmitters and regulates the level of neurotransmitter present in the synapse, thereby controlling how long a signal resulting from neurotransmitter release lasts. Because neurotransmitters are too large and hydrophilic to diffuse through the membrane, specific transport proteins are necessary for the reabsorption of neurotransmitters. Much research, both biochemical and structural, has been performed to obtain clues about the mechanism of reuptake. Protein structure The first primary sequence of a reuptake protein was published in 1990. The technique for protein sequence determination relied upon the purification ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |

_with_tube_model.png)
