|
Thiocarbamoyl Chloride
Dimethylthiocarbamoyl chloride is an organosulfur compound with the formula (CH3)2NC(S)Cl. A yellow solid, it is often encountered as a yellow syrup. It is a key reagent in the synthesis of arylthiols via the Newman-Kwart rearrangement. Synthesis and reactions Representative of other thiocarbamoyl chlorides, dimethylthiocarbamoyl chloride is electrophilic, serving as a source of R2NC(S)+.R. J. Cremlyn “An Introduction to Organosulfur Chemistry” John Wiley and Sons: Chichester (1996). It is analogous to dimethylcarbamoyl chloride (R2NC(O)Cl). Dimethylthiocarbamoyl chloride is prepared by chlorination of the related tetramethylthiuram disulfide: : e2NC(S)sub>2S2 + 3 Cl2 → 2 Me2NC(S)Cl + 2 SCl2 Dimethylthiocarbamoyl chloride reacts with dithiocarbamates (R2NCS{{su, b=2, p=−) to give thiuram sulfides 2NC(S)sub>2S. With methanethiolate, it gives methyl dimethyldithiocarbamate Methyl dimethyldithiocarbamate is the organosulfur compound with the formula (CH ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Organosulfur Compound
Organosulfur compounds are organic compounds that contain sulfur. They are often associated with foul odors, but many of the sweetest compounds known are organosulfur derivatives, e.g., saccharin. Nature abounds with organosulfur compounds—sulfur is vital for life. Of the 20 common amino acids, two ( cysteine and methionine) are organosulfur compounds, and the antibiotics penicillin and sulfa drugs both contain sulfur. While sulfur-containing antibiotics save many lives, sulfur mustard is a deadly chemical warfare agent. Fossil fuels, coal, petroleum, and natural gas, which are derived from ancient organisms, necessarily contain organosulfur compounds, the removal of which is a major focus of oil refineries. Sulfur shares the chalcogen group with oxygen, selenium, and tellurium, and it is expected that organosulfur compounds have similarities with carbon–oxygen, carbon–selenium, and carbon–tellurium compounds. A classical chemical test for the detection of sulfur co ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
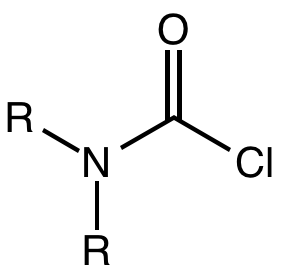
Carbamoyl Chloride
A carbamoyl chloride is the functional group with the formula R2NC(O)Cl. The parent carbamoyl chloride, H2NCOCl is unstable, but many N-substituted analogues are known. Most examples are moisture sensitive, colourless, and soluble in nonpolar organic solvents. An example is dimethylcarbamoyl chloride (m.p. −90 °C and b.p. 93 °C). Carbamoyl chlorides are used to prepare a number of pesticides, e.g. carbofuran and aldicarb. Production and examples Carbamoyl chlorides are prepared by the reaction of an amine with phosgene: :2 R2NH + COCl2 → R2NCOCl + 2NH2l They also arise by the addition of hydrogen chloride to isocyanates: :RNCO + HCl → RNHCOCl In this way, carbamonyl chlorides can be prepared with N-H functionality. Reactions In a reaction that is typically avoided, hydrolysis of carbamoyl chlorides gives carbamic acids: :R2NCOCl + H2O → R2NC(O)OH + HCl Owing to the influence of the amino group, these compounds are less hydrolytically sensitive than the usual acid chlo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tetramethylthiuram Disulfide
Thiram is the simplest thiuram disulfide and the oxidized dimer of dimethyldithiocarbamate. It is used as a fungicide, ectoparasiticide to prevent fungal diseases in seed and crops and similarly as an animal repellent to protect fruit trees and ornamentals from damage by rabbits, rodents and deer. It is effective against Stem gall of coriander, damping off, smut of millet, neck rot of onion, etc. Thiram has been used in the treatment of human scabies, as a sun screen and as a bactericide applied directly to the skin or incorporated into soap. Thiram is also used as a sulfur source and secondary accelerator the sulfur vulcanization Sulfur vulcanization is a chemical process for converting natural rubber or related polymers into materials of varying hardness, elasticity, and mechanical durability by heating them with sulfur or sulfur-containing compounds. Sulfur forms cros ... of rubbers. Usage Thiram was traditionally used in apple and wine farming. Since 2010 most thiram ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dithiocarbamate
In organic chemistry, a dithiocarbamate is a functional group with the general formula and structure . It is the analog of a carbamate in which both oxygen atoms are replaced by sulfur atoms (when only 1 oxygen is replaced the result is thiocarbamate). A common example is sodium diethyldithiocarbamate. Dithiocarbamates and their derivatives are widely used in the vulcanization of rubber. Formation Many primary and secondary amines react with carbon disulfide and sodium hydroxide to form dithiocarbamate salts: :R2NH + CS2 + NaOH → R2NCS2−Na+ + H2O Ammonia reacts with CS2 similarly: :2 NH3 + CS2 → H2NCS2−NH4+ Dithiocarbamate salts are pale colored solids that are soluble in water and polar organic solvents. Reactions Dithiocarbamates are readily S-alkylated. Thus, methyl dimethyldithiocarbamate can be prepared by methylation of the dithiocarbamate: :(CH3)2NCS2Na + (CH3O)2SO2 → (CH3)2NC(S)SCH3 + Na H3OSO3 Oxidation of dithiocarbamates gives th ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Methyl Dimethyldithiocarbamate
Methyl dimethyldithiocarbamate is the organosulfur compound with the formula (CH3)2NC(S)SCH3. It is the one of simplest dithiocarbamic esters. It is a white volatile solid that is poorly soluble in water but soluble in many organic solvents. It was once used as a pesticide. Methyl dimethyldithiocarbamate can be prepared by methylation of salts of dimethyldithiocarbamate: :(CH3)2NCS2Na + (CH3O)2SO2 → (CH3)2NC(S)SCH3 + Na H3OSO3 It can also be prepared by the reaction of a tetramethylthiuram disulfide Thiuram disulfides are a class of organosulfur compounds with the formula (R2NCSS)2. Many examples are known, but popular ones include R = Me and Et. They are disulfides obtained by oxidation of the dithiocarbamates. These compounds are used i ... with methyl Grignard reagents:{{cite journal, title=Reaction of Grignard Reagents with Tetramethylthiuram Disulfide ielding dithiocarbamatesauthor=John R. Grunwell, journal=J. Org. Chem., year=1970, volume=35, issue=5, pa ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |