|
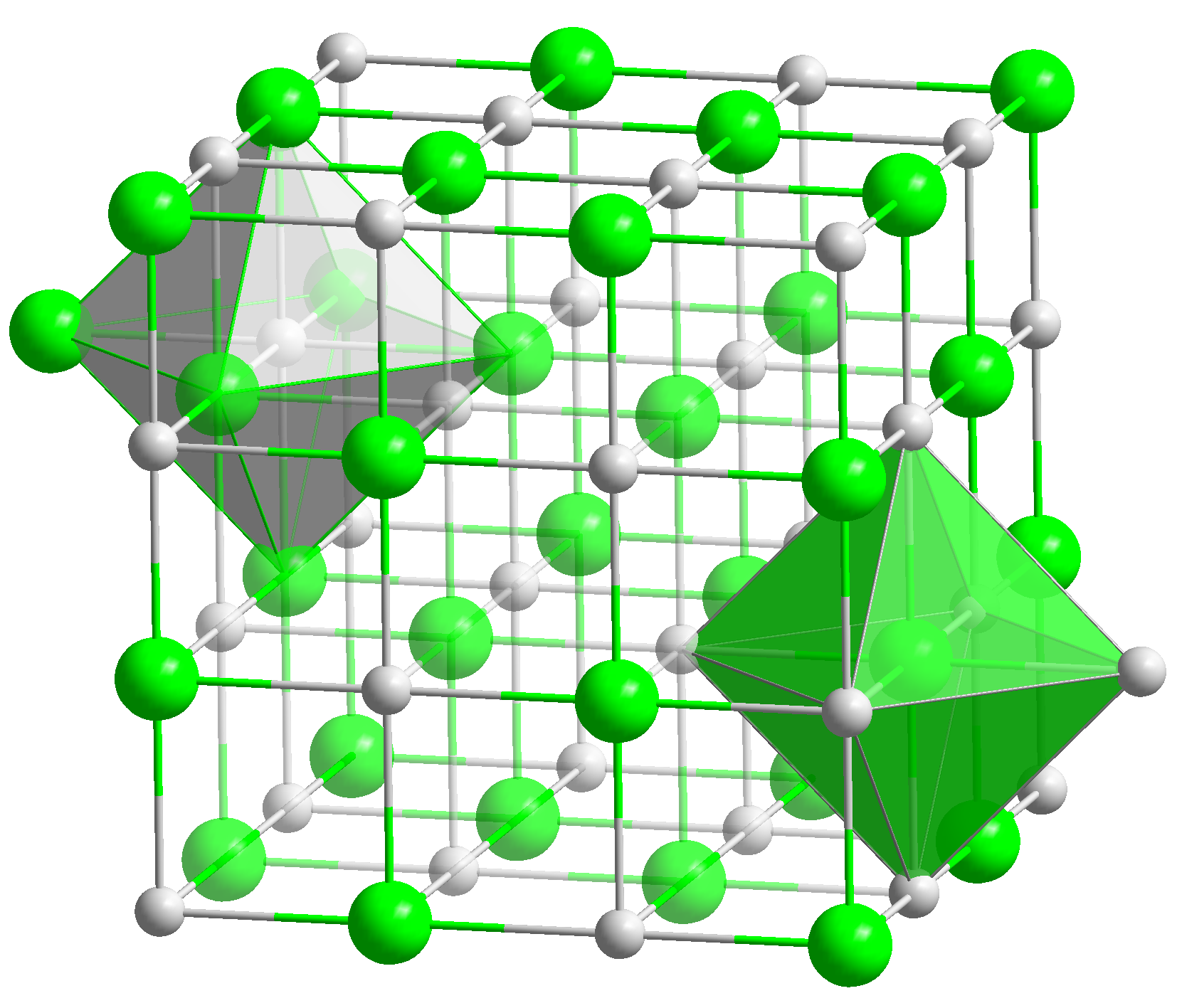
Sodium Zincate
Sodium zincate refers to anionic zinc oxides or hydroxides, depending on conditions. In the applications of these materials, the exact formula is not necessarily important and it is likely that aqueous zincate solutions consist of mixtures. Hydroxyzincates Solutions of sodium zincate may be prepared by dissolving zinc, zinc hydroxide, or zinc oxide Zinc oxide is an inorganic compound with the formula . It is a white powder that is insoluble in water. ZnO is used as an additive in numerous materials and products including cosmetics, food supplements, rubbers, plastics, ceramics, glass, cement ... in an aqueous solution of sodium hydroxide. Simplified equations for these complex processes are: :ZnO + H2O + 2 NaOH → Na2Zn(OH)4 :Zn + 2 H2O + 2 NaOH → Na2Zn(OH)4 + H2 From such solutions, one can crystallize salts of containing the anions Zn(OH)42−, Zn2(OH)62−, and Zn(OH)64−. Na2Zn(OH)4 consists of tetrahedral zincate ion and octahedral sodium cations. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Zincate
In chemistry the term zincate may refer to several substances containing the element zinc: * usually the anion ZnO22−, more properly called tetrahydroxozincate or salts thereof, such as sodium zincate . * the polymeric anion n(OH)3−and its salts, for example NaZn(OH)3· H2O. * an oxide containing zinc and a less electronegative element e.g. Na2ZnO2. In the health supplement industry zincate may also mean a commercially available zinc supplement, typically formulated as zinc sulfate. Solutions prepared from dissolving zinc hydroxide or zinc oxide in a strong alkali like sodium hydroxide, which contains various zincate anions, are used in the metal plating industry, in processes such as immersion zinc plating and electroplating (electrogalvanization). Any of these techniques may be called zincate process. Inorganic compound nomenclature In the naming of inorganic compounds, "-zincate" is a suffix that indicates that a polyatomic anion contains a central zinc atom. Examples ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Zinc
Zinc is a chemical element with the symbol Zn and atomic number 30. Zinc is a slightly brittle metal at room temperature and has a shiny-greyish appearance when oxidation is removed. It is the first element in group 12 (IIB) of the periodic table. In some respects, zinc is chemically similar to magnesium: both elements exhibit only one normal oxidation state (+2), and the Zn2+ and Mg2+ ions are of similar size.The elements are from different metal groups. See periodic table. Zinc is the 24th most abundant element in Earth's crust and has five stable isotopes. The most common zinc ore is sphalerite (zinc blende), a zinc sulfide mineral. The largest workable lodes are in Australia, Asia, and the United States. Zinc is refined by froth flotation of the ore, roasting, and final extraction using electricity ( electrowinning). Zinc is an essential trace element for humans, animals, plants and for microorganisms and is necessary for prenatal and postnatal development. It ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Zinc Hydroxide
Zinc hydroxide Zn( OH)2 is an inorganic chemical compound. It also occurs naturally as 3 rare minerals: wülfingite (orthorhombic), ashoverite and sweetite (both tetragonal). Like the hydroxides of other metals, such as lead, aluminium, beryllium, tin and chromium, Zinc hydroxide (and Zinc oxide), is amphoteric. Thus it will dissolve readily in a dilute solution of a strong acid, such as HCl, and also in a solution of an alkali such as sodium hydroxide. Preparation It can be prepared by adding sodium hydroxide solution, but not in excess, to a solution of any zinc salt. A white precipitate will be seen: :Zn2+ + 2 OH− → Zn(OH)2. Zn2+ is known to form hexa-aqua ions at high water concentrations and tetra-aqua ions at low concentrations of water Sze, Yu-Keung, and Donald E. Irish. "Vibrational spectral studies of ion-ion and ion-solvent interactions. I. Zinc nitrate in water." Journal of Solution Chemistry 7.6 (1978): 395-415. and, thus, this reaction may be better writt ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Zinc Oxide
Zinc oxide is an inorganic compound with the formula . It is a white powder that is insoluble in water. ZnO is used as an additive in numerous materials and products including cosmetics, food supplements, rubbers, plastics, ceramics, glass, cement, lubricants, paints, ointments, adhesives, sealants, pigments, foods, batteries, ferrites, fire retardants, and first-aid tapes. Although it occurs naturally as the mineral zincite, most zinc oxide is produced synthetically. ZnO is a wide-band gap semiconductor of the II-VI semiconductor group. The native doping of the semiconductor due to oxygen vacancies or zinc interstitials is n-type. Other favorable properties include good transparency, high electron mobility, wide band gap, and strong room-temperature luminescence. Those properties make ZnO valuable for a variety of emerging applications: transparent electrodes in liquid crystal displays, energy-saving or heat-protecting windows, and electronics as thin-film transistors and lig ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sodium Hydroxide
Sodium hydroxide, also known as lye and caustic soda, is an inorganic compound with the formula NaOH. It is a white solid ionic compound consisting of sodium cations and hydroxide anions . Sodium hydroxide is a highly caustic base and alkali that decomposes proteins at ordinary ambient temperatures and may cause severe chemical burns. It is highly soluble in water, and readily absorbs moisture and carbon dioxide from the air. It forms a series of hydrates . The monohydrate crystallizes from water solutions between 12.3 and 61.8 °C. The commercially available "sodium hydroxide" is often this monohydrate, and published data may refer to it instead of the anhydrous compound. As one of the simplest hydroxides, sodium hydroxide is frequently used alongside neutral water and acidic hydrochloric acid to demonstrate the pH scale to chemistry students. Sodium hydroxide is used in many industries: in the manufacture of pulp and paper, textiles, drinking water, soaps and deterge ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sodium Compounds
Sodium atoms have 11 electrons, one more than the stable configuration of the noble gas neon. As a result, sodium usually forms ionic compounds involving the Na+ cation. Sodium is a reactive alkali metal and is much more stable in ionic compounds. It can also form intermetallic compounds and organosodium compounds. Sodium compounds are often soluble in water. Metallic sodium Metallic sodium is generally less reactive than potassium and more reactive than lithium. Sodium metal is highly reducing, with the standard reduction potential for the Na+/Na couple being −2.71 volts, though potassium and lithium have even more negative potentials. The thermal, fluidic, chemical, and nuclear properties of molten sodium metal have caused it to be one of the main coolants of choice for the fast breeder reactor. Such nuclear reactors are seen as a crucial step for the production of clean energy. Salts and oxides Sodium compounds are of immense commercial importance, being particularly centra ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |