|
Salination
Soil salinity is the salt content in the soil; the process of increasing the salt content is known as salinization. Salts occur naturally within soils and water. Salination can be caused by natural processes such as mineral weathering or by the gradual withdrawal of an ocean. It can also come about through artificial processes such as irrigation and road salt. Natural occurrence Salts are a natural component in soils and water. The ions responsible for salination are: Na+, K+, Ca2+, Mg2+ and Cl−. Over long periods of time, as soil minerals weather and release salts, these salts are flushed or leached out of the soil by drainage water in areas with sufficient precipitation. In addition to mineral weathering, salts are also deposited via dust and precipitation. Salts may accumulate in dry regions, leading to naturally saline soils. This is the case, for example, in large parts of Australia. Human practices can increase the salinity of soils by the addition of salts i ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Soil
Soil, also commonly referred to as earth or dirt Dirt is an unclean matter, especially when in contact with a person's clothes, skin, or possessions. In such cases, they are said to become dirty. Common types of dirt include: * Debris: scattered pieces of waste or remains * Dust: a gener ..., is a mixture of organic matter, minerals, gases, liquids, and organisms that together support life. Some scientific definitions distinguish ''dirt'' from ''soil'' by restricting the former term specifically to displaced soil. Soil consists of a solid phase of minerals and organic matter (the soil matrix), as well as a Porosity, porous phase that holds Soil gas, gases (the soil atmosphere) and water (the soil solution). Accordingly, soil is a three-state of matter, state system of solids, liquids, and gases. Soil is a product of several factors: the influence of climate, terrain, relief (elevation, orientation, and slope of terrain), organisms, and the soil's parent materials (ori ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
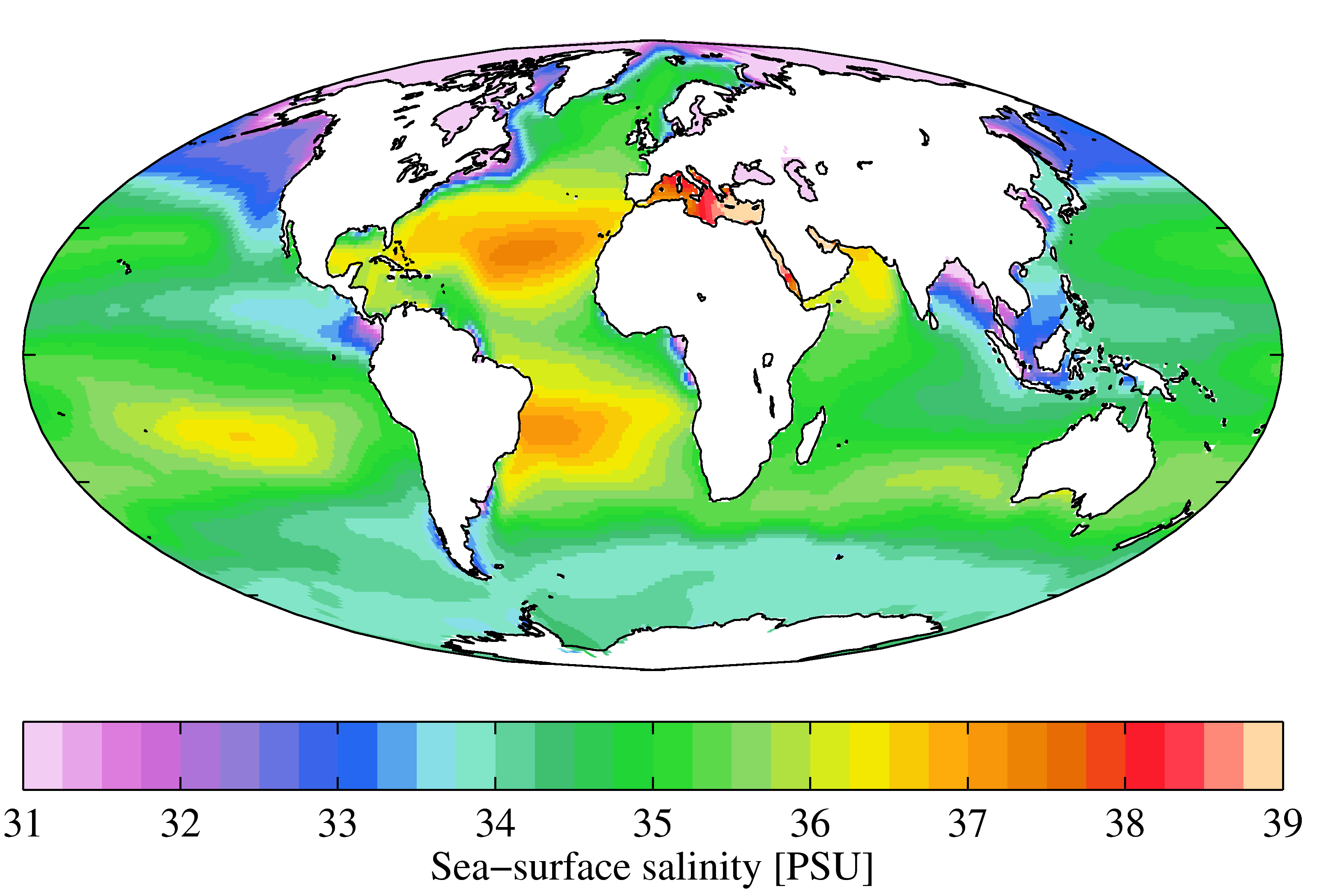
Salinity
Salinity () is the saltiness or amount of salt (chemistry), salt dissolved in a body of water, called saline water (see also soil salinity). It is usually measured in g/L or g/kg (grams of salt per liter/kilogram of water; the latter is dimensionless and equal to ‰). Salinity is an important factor in determining many aspects of the chemistry of natural waters and of biological processes within it, and is a state function, thermodynamic state variable that, along with temperature and pressure, governs physical characteristics like the density and heat capacity of the water. A contour line of constant salinity is called an ''isohaline'', or sometimes ''isohale''. Definitions Salinity in rivers, lakes, and the ocean is conceptually simple, but technically challenging to define and measure precisely. Conceptually the salinity is the quantity of dissolved salt content of the water. Salts are compounds like sodium chloride, magnesium sulfate, potassium nitrate, and sodium bicarbo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Gypsum
Gypsum is a soft sulfate mineral composed of calcium sulfate dihydrate, with the chemical formula . It is widely mined and is used as a fertilizer and as the main constituent in many forms of plaster, blackboard or sidewalk chalk, and drywall. Alabaster, a fine-grained white or lightly tinted variety of gypsum, has been used for sculpture by many cultures including Ancient Egypt, Mesopotamia, Ancient Rome, the Byzantine Empire, and the Nottingham alabasters of Medieval England. Gypsum also crystallizes as translucent crystals of selenite. It forms as an evaporite mineral and as a hydration product of anhydrite. The Mohs scale of mineral hardness defines gypsum as hardness value 2 based on scratch hardness comparison. Etymology and history The word '' gypsum'' is derived from the Greek word (), "plaster". Because the quarries of the Montmartre district of Paris have long furnished burnt gypsum (calcined gypsum) used for various purposes, this dehydrated gypsum becam ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sodic Soil
Soil salinity is the salt content in the soil; the process of increasing the salt content is known as salinization. Salts occur naturally within soils and water. Salination can be caused by natural processes such as mineral weathering or by the gradual withdrawal of an ocean. It can also come about through artificial processes such as irrigation and road salt. Natural occurrence Salts are a natural component in soils and water. The ions responsible for salination are: Na+, K+, Ca2+, Mg2+ and Cl−. Over long periods of time, as soil minerals weather and release salts, these salts are flushed or leached out of the soil by drainage water in areas with sufficient precipitation. In addition to mineral weathering, salts are also deposited via dust and precipitation. Salts may accumulate in dry regions, leading to naturally saline soils. This is the case, for example, in large parts of Australia. Human practices can increase the salinity of soils by the addition of salts in ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Arable Land
Arable land (from the la, arabilis, "able to be ploughed") is any land capable of being ploughed and used to grow crops.''Oxford English Dictionary'', "arable, ''adj''. and ''n.''" Oxford University Press (Oxford), 2013. Alternatively, for the purposes of agricultural statistics, the term often has a more precise definition: A more concise definition appearing in the Eurostat glossary similarly refers to actual rather than potential uses: "land worked (ploughed or tilled) regularly, generally under a system of crop rotation". In Britain, arable land has traditionally been contrasted with pasturable land such as heaths, which could be used for sheep-rearing but not as farmland. Arable land area According to the Food and Agriculture Organization of the United Nations, in 2013, the world's arable land amounted to 1.407 billion hectares, out of a total of 4.924 billion hectares of land used for agriculture. Arable land (hectares per person) Non-arable lan ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ground Water
Groundwater is the water present beneath Earth's surface in rock and soil pore spaces and in the fractures of rock formations. About 30 percent of all readily available freshwater in the world is groundwater. A unit of rock or an unconsolidated deposit is called an aquifer when it can yield a usable quantity of water. The depth at which soil pore spaces or fractures and voids in rock become completely saturated with water is called the water table. Groundwater is recharged from the surface; it may discharge from the surface naturally at springs and seeps, and can form oases or wetlands. Groundwater is also often withdrawn for agricultural, municipal, and industrial use by constructing and operating extraction wells. The study of the distribution and movement of groundwater is hydrogeology, also called groundwater hydrology. Typically, groundwater is thought of as water flowing through shallow aquifers, but, in the technical sense, it can also contain soil moisture, permafro ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Potassium
Potassium is the chemical element with the symbol K (from Neo-Latin ''kalium'') and atomic number19. Potassium is a silvery-white metal that is soft enough to be cut with a knife with little force. Potassium metal reacts rapidly with atmospheric oxygen to form flaky white potassium peroxide in only seconds of exposure. It was first isolated from potash, the ashes of plants, from which its name derives. In the periodic table, potassium is one of the alkali metals, all of which have a single valence electron in the outer electron shell, that is easily removed to create an ion with a positive charge – a cation, that combines with anions to form salts. Potassium in nature occurs only in ionic salts. Elemental potassium reacts vigorously with water, generating sufficient heat to ignite hydrogen emitted in the reaction, and burning with a lilac- colored flame. It is found dissolved in sea water (which is 0.04% potassium by weight), and occurs in many minerals such as orthocl ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dryland Salinity
Dryland salinity is a natural process for soil, just like other processes such as wind erosion. Salinity degrades land by an increase in soil salt concentration in the environment, watercourse or soil in unirrigated landscapes, being in excess of normal soil salt concentrations in dryland regions. Overview Salinity refers to the movement and concentration of salt in the landscape and its associated detriment to land and water resources; dryland salinity refers to salinity in unirrigated landscapes. Salinity processes extend from local to regional scales and are driven by imbalances in the water budget that result, primarily, from agriculturally driven landscape change. There are two types of salinity: Types of salinity There are two types of salinity. Primary salinity (natural salinity) and secondary salinity (induced salinity). (Nrm.qld.gov.au, 2013) Primary salinity naturally occurs in arid and saline environments such as salt lakes, marshes, pans and salt flats. Natural accum ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sodium
Sodium is a chemical element with the symbol Na (from Latin ''natrium'') and atomic number 11. It is a soft, silvery-white, highly reactive metal. Sodium is an alkali metal, being in group 1 of the periodic table. Its only stable isotope is 23Na. The free metal does not occur in nature, and must be prepared from compounds. Sodium is the sixth most abundant element in the Earth's crust and exists in numerous minerals such as feldspars, sodalite, and halite (NaCl). Many salts of sodium are highly water-soluble: sodium ions have been leached by the action of water from the Earth's minerals over eons, and thus sodium and chlorine are the most common dissolved elements by weight in the oceans. Sodium was first isolated by Humphry Davy in 1807 by the electrolysis of sodium hydroxide. Among many other useful sodium compounds, sodium hydroxide (lye) is used in soap manufacture, and sodium chloride (edible salt) is a de-icing agent and a nutrient for animals inc ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Alkaline Soil
Alkali, or Alkaline, soils are clay soils with high pH (greater than 8.5), a poor soil structure and a low infiltration capacity. Often they have a hard calcareous layer at 0.5 to 1 metre depth. Alkali soils owe their unfavorable physico-chemical properties mainly to the dominating presence of sodium carbonate, which causes the soil to swellManaging irrigation water quality, Oregon State University, USA Retrieved on 2012-10-04. and difficult to clarify/settle. They derive their name from the group of elements, to which sodium belon ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Alkali Soil
Alkali, or Alkaline, soils are clay soils with high pH (greater than 8.5), a poor soil structure and a low infiltration capacity. Often they have a hard calcareous layer at 0.5 to 1 metre depth. Alkali soils owe their unfavorable physico-chemical properties mainly to the dominating presence of sodium carbonate, which causes the soil to swellManaging irrigation water quality, Oregon State University, USA Retrieved on 2012-10-04. and difficult to clarify/settle. They derive their name from the group of elements, to which |
Ion Toxicity
An ion () is an atom or molecule with a net electrical charge. The charge of an electron is considered to be negative by convention and this charge is equal and opposite to the charge of a proton, which is considered to be positive by convention. The net charge of an ion is not zero because its total number of electrons is unequal to its total number of protons. A cation is a positively charged ion with fewer electrons than protons while an anion is a negatively charged ion with more electrons than protons. Opposite electric charges are pulled towards one another by electrostatic force, so cations and anions attract each other and readily form ionic compounds. Ions consisting of only a single atom are termed atomic or monatomic ions, while two or more atoms form molecular ions or polyatomic ions. In the case of physical ionization in a fluid (gas or liquid), "ion pairs" are created by spontaneous molecule collisions, where each generated pair consists of a free electron and ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |