|
Resiniferatoxin
Resiniferatoxin (RTX) is a naturally occurring chemical found in resin spurge ('' Euphorbia resinifera''), a cactus-like plant commonly found in Morocco, and in '' Euphorbia poissonii'' found in northern Nigeria.''Euphorbia poissonii'' in BoDD – Botanical Dermatology Database It is a potent functional analog of , the active ingredient in . Biological activity |
Euphorbia Resinifera
''Euphorbia resinifera'', the resin spurge, is a species of spurge native to Morocco, where it occurs on the slopes of the Atlas Mountains. The dried latex of the plant was used in ancient medicine. It contains resiniferatoxin, an extremely potent capsaicin analog tested as an analgesic since 1997. Growth It is a shrub growing to tall, forming multi-stemmed cushion-shaped clumps up to wide. The stems are erect, succulent, superficially like a cactus, four-angled, with short but sharp pairs of spines on the angles, spaced about apart up the stem. Geographical distribution ''Euphorbia resinifera'' is a species of spurge native to Morocco, where it occurs on the slopes of the Atlas Mountains. It is similar to its relative '' Euphorbia echinus'', which occurs on the Moroccan coast and the Canary Islands. Due to its origin it is also called the African spurge. Chemical constituents ''Euphorbia resinifera'' contains a milky fluid or latex, which in its dried form is called E ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
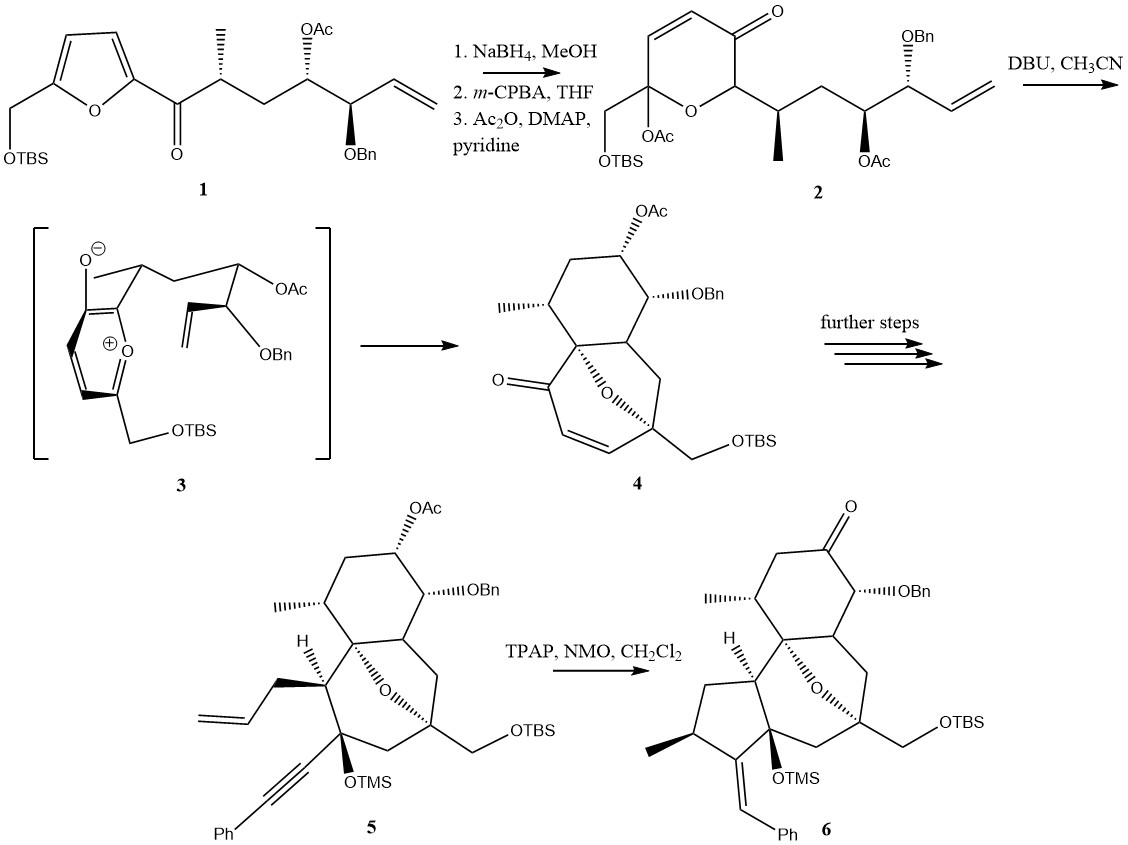
RTX Partial Synthesis
RTX may refer to: Science and technology * Radiation therapy, is therapy using ionizing radiation, generally as part of cancer treatment to control or kill malignant cells * Resiniferatoxin, a natural, high-potency ligand that activates the capsaicin (VR1, TRPV1) receptor * Rituximab, a monoclonal antibody used in the treatment of leukemia, lymphomas, transplant rejection and autoimmune disorders * RTX2010, a radiation-hardened microprocessor which has been used in numerous spacecraft * RTX toxin (repeats in toxin), a type of toxin secreted by Gram-negative bacteria Computing * Ray tracing (graphics), sometimes abbreviated RTX * Nvidia GeForce RTX, a series of Nvidia graphic cards * .rtx (ring tone XML file extension), of the Ring Tone Transfer Language * RTX (operating system), a real-time operating system extension for Microsoft Windows * Keil RTX, a real-time operating system (RTOS) for ARM devices Other uses * RTX, a band formed by Jennifer Herrema * RTX (event), a video ga ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Capsaicin
Capsaicin (8-methyl-''N''-vanillyl-6-nonenamide) ( or ) is an active component of chili peppers, which are plants belonging to the genus ''Capsicum''. It is a chemical irritant for mammals, including humans, and produces a sensation of burning in any tissue with which it comes into contact. Capsaicin and several related alkaloids are called capsaicinoids and are produced as secondary metabolites by chili peppers, probably as deterrents against certain mammals and fungi.What Made Chili Peppers So Spicy? Talk of the Nation, 15 August 2008. Pure capsaicin is a , colorless, highly pungent, [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Scoville Heat Units
The Scoville scale is a measurement of the pungency (spiciness or "heat") of chili peppers, as recorded in Scoville heat units (SHU), based on the concentration of capsaicinoids, among which capsaicin is the predominant component. The scale is named after its creator, American pharmacist Wilbur Scoville, whose 1912 method is known as the Scoville organoleptic test. The Scoville organoleptic test is a subjective assessment derived from the capsaicinoid sensitivity by people experienced with eating hot chilis. An alternative method, high-performance liquid chromatography (HPLC), can be used to analytically quantify the capsaicinoid content as an indicator of pungency. As of 2011, the subjective organoleptic test has been largely superseded by analytical methods such as HPLC. Scoville organoleptic test In the Scoville organoleptic test, an exact weight of dried pepper is dissolved in alcohol to extract the heat components (capsaicinoids), then diluted in a solution of sugar ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chemical Burn
A chemical burn occurs when living tissue is exposed to a corrosive substance (such as a strong acid, base or oxidizer) or a cytotoxic agent (such as mustard gas, lewisite or arsine). Chemical burns follow standard burn classification and may cause extensive tissue damage. The main types of irritant and/or corrosive products are: acids, bases, oxidizers / reducing agents, solvents, and alkylants. Additionally, chemical burns can be caused by some types of cytotoxic chemical weapons, e.g., vesicants such as mustard gas and Lewisite, or urticants such as phosgene oxime. Chemical burns may: * need no source of heat * occur immediately on contact * not be immediately evident or noticeable * be extremely painful * diffuse into tissue and damage cellular structures under skin without immediately apparent damage to skin surface Presentation The exact symptoms of a chemical burn depend on the chemical involved. Symptoms include itching, bleaching or darkening of skin, burning sens ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
University Of Tokyo
, abbreviated as or UTokyo, is a public research university located in Bunkyō, Tokyo, Japan. Established in 1877, the university was the first Imperial University and is currently a Top Type university of the Top Global University Project by the Japanese government. UTokyo has 10 faculties, 15 graduate schools and enrolls about 30,000 students, about 4,200 of whom are international students. In particular, the number of privately funded international students, who account for more than 80%, has increased 1.75 times in the 10 years since 2010, and the university is focusing on supporting international students. Its five campuses are in Hongō, Komaba, Kashiwa, Shirokane and Nakano. It is considered to be the most selective and prestigious university in Japan. As of 2021, University of Tokyo's alumni, faculty members and researchers include seventeen prime ministers, 18 Nobel Prize laureates, four Pritzker Prize laureates, five astronauts, and a Fields Medalist. Hi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Masayuki Inoue
Masayuki (written: , ,, , , , , , , , , , , , , , or ) is a masculine Japanese given name. Notable people with the name include: *, Japanese animator and director *, Japanese actor *, Japanese baseball player and manager *, Japanese politician *, Japanese samurai *, Japanese pianist and composer *, Japanese karateka *, Japanese ''daimyō'' *, Japanese handball player *, Japanese manga artist *, Japanese astronomer *, Japanese volleyball player *, Japanese voice actor *, Japanese speed skater *, Japanese physician *Masayuki Kawamura (golfer) (born 1967), Japanese golfer *, Japanese seismologist *, Japanese swimmer *, Japanese animator and anime director *, Japanese professional wrestler and mixed martial artist *, Japanese footballer *, Japanese sport wrestler *, Japanese gymnast *, Japanese volleyball player *, Japanese footballer *, Japanese anime director *, Japanese artist *, Japanese sumo wrestler *, Japanese actor *, Japanese film producer *, Japanese sculptor *, Japanese foot ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tetrapropylammonium Perruthenate
Tetrapropylammonium perruthenate (TPAP or TPAPR) is the chemical compound described by the formula N(C3H7)4RuO4. Sometimes known as the Ley–Griffith reagent, this ruthenium compound is used as a reagent in organic synthesis. This salt consists of the tetrapropylammonium cation and the perruthenate anion, . Uses Ruthenium tetroxide is a highly aggressive oxidant, but TPAP, which is its one-electron reduced derivative, is a mild oxidizing agent for the conversion of primary alcohols to aldehydes (the Ley oxidation). Secondary alcohols are similarly oxidized to ketones. It can also be used to oxidize primary alcohols all the way to the carboxylic acid with a higher catalyst loading, larger amount of the cooxidant, and addition of two equivalents of water. In this situation, the aldehyde reacts with water to form the geminal diol hydrate, which is then oxidized again. The oxidation generates water that can be removed by adding molecular sieves. TPAP is expensive, but it ca ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
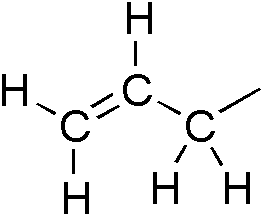
Allylic
In organic chemistry, an allyl group is a substituent with the structural formula , where R is the rest of the molecule. It consists of a methylene bridge () attached to a vinyl group (). The name is derived from the scientific name for garlic, . In 1844, Theodor Wertheim isolated an allyl derivative from garlic oil and named it "". The term allyl applies to many compounds related to , some of which are of practical or of everyday importance, for example, allyl chloride. Allylation is any chemical reaction that adds an allyl group to a substrate. Nomenclature A site adjacent to the unsaturated carbon atom is called the allylic position or allylic site. A group attached at this site is sometimes described as allylic. Thus, "has an allylic hydroxyl group". Allylic C−H bonds are about 15% weaker than the C−H bonds in ordinary sp3 carbon centers and are thus more reactive. Benzylic and allylic are related in terms of structure, bond strength, and reactivity. Other ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Acetonitrile
Acetonitrile, often abbreviated MeCN (methyl cyanide), is the chemical compound with the formula and structure . This colourless liquid is the simplest organic nitrile (hydrogen cyanide is a simpler nitrile, but the cyanide anion is not classed as organic). It is produced mainly as a byproduct of acrylonitrile manufacture. It is used as a polar aprotic solvent in organic synthesis and in the purification of butadiene. The skeleton is linear with a short distance of 1.16 Å. Acetonitrile was first prepared in 1847 by the French chemist Jean-Baptiste Dumas. Applications Acetonitrile is used mainly as a solvent in the purification of butadiene in refineries. Specifically, acetonitrile is fed into the top of a distillation column filled with hydrocarbons including butadiene, and as the acetonitrile falls down through the column, it absorbs the butadiene which is then sent from the bottom of the tower to a second separating tower. Heat is then employed in the separatin ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
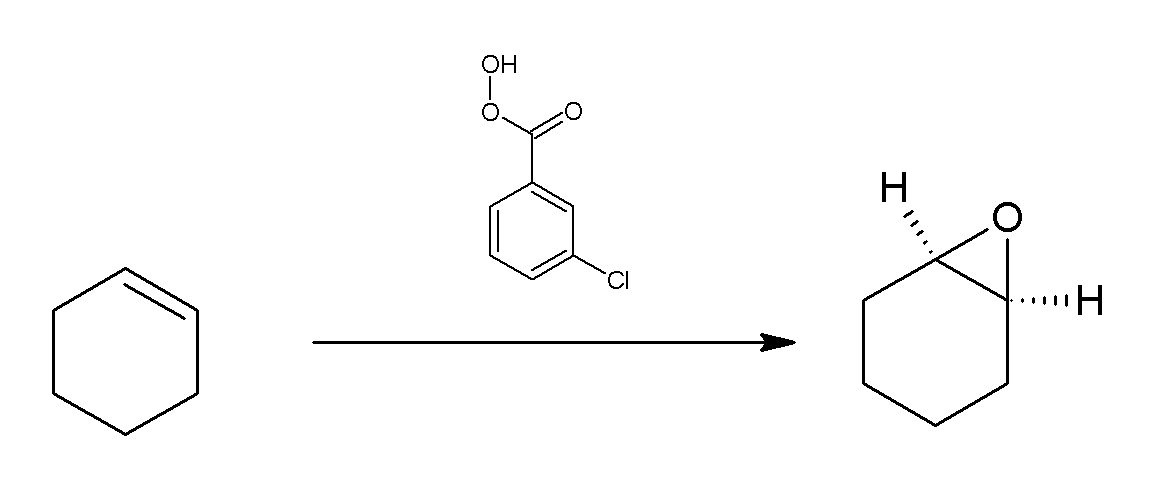
M-CPBA
''meta''-Chloroperoxybenzoic acid (mCPBA or ''m''CPBA) is a peroxycarboxylic acid. A white solid, it is used widely as an oxidant in organic synthesis. mCPBA is often preferred to other peroxy acids because of its relative ease of handling. mCPBA is a strong oxidizing agent that may cause fire upon contact with flammable material. Preparation and purification mCPBA can be prepared by reacting m-Chlorobenzoyl chloride with a basic solution of hydrogen peroxide, followed by acidification. It is sold commercially as a shelf-stable mixture that is less than 72% mCPBA, with the balance made up of ''m''-chlorobenzoic acid (10%) and water. The peroxyacid can be purified by washing the commercial material with a sodium hydroxide and potassium phosphate solution buffered at pH = 7.5. Peroxyacids are generally slightly less acidic than their carboxylic acid counterparts, so one can extract the acid impurity by careful control of pH. The purified material is reasonably stable against d ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |