|
Photonic Molecule
Photonic molecules are a theoretical natural form of matter which can also be made artificially in which photons bind together to form "molecules". They were first predicted in 2007. Photonic molecules are formed when individual (massless) photons "interact with each other so strongly that they act as though they have mass". In an alternative definition (which is not equivalent), photons confined to two or more coupled optical cavities also reproduce the physics of interacting atomic energy levels, and have been termed as photonic molecules. Researchers drew analogies between the phenomenon and the fictional "lightsaber" from ''Star Wars''. Construction Gaseous rubidium atoms were pumped into a vacuum chamber. The cloud was cooled using lasers to just a few degrees above absolute zero. Using weak laser pulses, small numbers of photons were fired into the cloud. As the photons entered the cloud, their energy excited atoms along their path, causing them to lose speed. Inside the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Photon
A photon () is an elementary particle that is a quantum of the electromagnetic field, including electromagnetic radiation such as light and radio waves, and the force carrier for the electromagnetic force. Photons are massless, so they always move at the speed of light in vacuum, (or about ). The photon belongs to the class of bosons. As with other elementary particles, photons are best explained by quantum mechanics and exhibit wave–particle duality, their behavior featuring properties of both waves and particles. The modern photon concept originated during the first two decades of the 20th century with the work of Albert Einstein, who built upon the research of Max Planck. While trying to explain how matter and electromagnetic radiation could be in thermal equilibrium with one another, Planck proposed that the energy stored within a material object should be regarded as composed of an integer number of discrete, equal-sized parts. To explain the photoelectric effect, Eins ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Bonding Molecular Orbital
In theoretical chemistry, the bonding orbital is used in molecular orbital (MO) theory to describe the attractive interactions between the atomic orbitals of two or more atoms in a molecule. In MO theory, electrons are portrayed to move in waves. When more than one of these waves come close together, the in-phase combination of these waves produces an interaction that leads to a species that is greatly stabilized. The result of the waves’ constructive interference causes the density of the electrons to be found within the binding region, creating a stable bond between the two species. Diatomic molecules In the classic example of the H2 MO, the two separate H atoms have identical atomic orbitals. When creating the molecule dihydrogen, the individual valence orbitals, 1''s'', either: merge in phase to get bonding orbitals, where the electron density is in between the nuclei of the atoms; or, merge out of phase to get antibonding orbitals, where the electron density is everywh ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
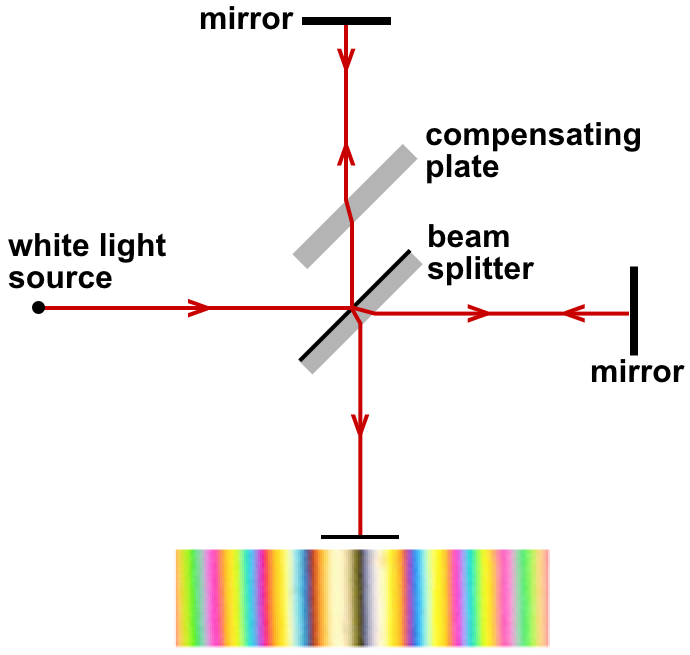
Luminiferous Aether
Luminiferous aether or ether ("luminiferous", meaning "light-bearing") was the postulated medium for the propagation of light. It was invoked to explain the ability of the apparently wave-based light to propagate through empty space (a vacuum), something that waves should not be able to do. The assumption of a spatial plenum of luminiferous aether, rather than a spatial vacuum, provided the theoretical medium that was required by wave theories of light. The aether hypothesis was the topic of considerable debate throughout its history, as it required the existence of an invisible and infinite material with no interaction with physical objects. As the nature of light was explored, especially in the 19th century, the physical qualities required of an aether became increasingly contradictory. By the late 1800s, the existence of the aether was being questioned, although there was no physical theory to replace it. The negative outcome of the Michelson–Morley experiment (1887) suggeste ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Photoluminescence
Photoluminescence (abbreviated as PL) is light emission from any form of matter after the absorption of photons (electromagnetic radiation). It is one of many forms of luminescence (light emission) and is initiated by photoexcitation (i.e. photons that excite electrons to a higher energy level in an atom), hence the prefix ''photo-''. Following excitation, various relaxation processes typically occur in which other photons are re-radiated. Time periods between absorption and emission may vary: ranging from short femtosecond-regime for emission involving free-carrier plasma in inorganic semiconductorsHayes, G.R.; Deveaud, B. (2002). "Is Luminescence from Quantum Wells Due to Excitons?". ''Physica Status Solidi A'' 190 (3): 637–640doi:10.1002/1521-396X(200204)190:33.0.CO;2-7/ref> up to milliseconds for phosphoresence processes in molecular systems; and under special circumstances delay of emission may even span to minutes or hours. Observation of photoluminescence at a certain ene ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Polarization (waves)
Polarization (also polarisation) is a property applying to transverse waves that specifies the geometrical orientation of the oscillations. In a transverse wave, the direction of the oscillation is perpendicular to the direction of motion of the wave. A simple example of a polarized transverse wave is vibrations traveling along a taut string ''(see image)''; for example, in a musical instrument like a guitar string. Depending on how the string is plucked, the vibrations can be in a vertical direction, horizontal direction, or at any angle perpendicular to the string. In contrast, in longitudinal waves, such as sound waves in a liquid or gas, the displacement of the particles in the oscillation is always in the direction of propagation, so these waves do not exhibit polarization. Transverse waves that exhibit polarization include electromagnetic waves such as light and radio waves, gravitational waves, and transverse sound waves (shear waves) in solids. An electromagnetic wa ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
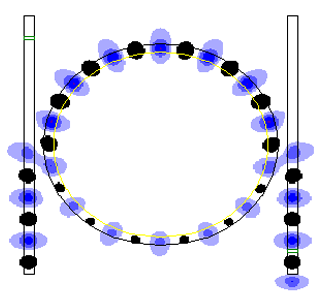
Optical Ring Resonators
An optical ring resonator is a set of waveguides in which at least one is a closed loop coupled to some sort of light input and output. (These can be, but are not limited to being, waveguides.) The concepts behind optical ring resonators are the same as those behind whispering galleries except that they use light and obey the properties behind constructive interference and total internal reflection. When light of the resonant wavelength is passed through the loop from the input waveguide, the light builds up in intensity over multiple round-trips owing to constructive interference and is output to the output bus waveguide which serves as a detector waveguide. Because only a select few wavelengths will be at resonance within the loop, the optical ring resonator functions as a filter. Additionally, as implied earlier, two or more ring waveguides can be coupled to each other to form an add/drop optical filter. Background Optical ring resonators work on the principles behind total ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Optical Fiber
An optical fiber, or optical fibre in Commonwealth English, is a flexible, transparent fiber made by drawing glass (silica) or plastic to a diameter slightly thicker than that of a human hair. Optical fibers are used most often as a means to transmit light between the two ends of the fiber and find wide usage in fiber-optic communications, where they permit transmission over longer distances and at higher bandwidths (data transfer rates) than electrical cables. Fibers are used instead of metal wires because signals travel along them with less loss; in addition, fibers are immune to electromagnetic interference, a problem from which metal wires suffer. Fibers are also used for illumination and imaging, and are often wrapped in bundles so they may be used to carry light into, or images out of confined spaces, as in the case of a fiberscope. Specially designed fibers are also used for a variety of other applications, some of them being fiber optic sensors and fiber lasers. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrogen Molecule
Hydrogen is the chemical element with the symbol H and atomic number 1. Hydrogen is the lightest element. At standard conditions hydrogen is a gas of diatomic molecules having the formula . It is colorless, odorless, tasteless, non-toxic, and highly combustible. Hydrogen is the most abundant chemical substance in the universe, constituting roughly 75% of all normal matter.However, most of the universe's mass is not in the form of baryons or chemical elements. See dark matter and dark energy. Stars such as the Sun are mainly composed of hydrogen in the plasma state. Most of the hydrogen on Earth exists in molecular forms such as water and organic compounds. For the most common isotope of hydrogen (symbol 1H) each atom has one proton, one electron, and no neutrons. In the early universe, the formation of protons, the nuclei of hydrogen, occurred during the first second after the Big Bang. The emergence of neutral hydrogen atoms throughout the universe occurred about 370,00 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Antibonding Molecular Orbital
In chemical bonding theory, an antibonding orbital is a type of molecular orbital that weakens the chemical bond between two atoms and helps to raise the energy of the molecule relative to the separated atoms. Such an orbital has one or more nodes in the bonding region between the nuclei. The density of the electrons in the orbital is concentrated outside the bonding region and acts to pull one nucleus away from the other and tends to cause mutual repulsion between the two atoms. This is in contrast to a bonding molecular orbital, which has a lower energy than that of the separate atoms, and is responsible for chemical bonds. Diatomic molecules Antibonding molecular orbitals (MOs) are normally ''higher'' in energy than bonding molecular orbitals. Bonding and antibonding orbitals form when atoms combine into molecules. If two hydrogen atoms are initially far apart, they have identical atomic orbitals. However, as the spacing between the two atoms becomes smaller, the electron w ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
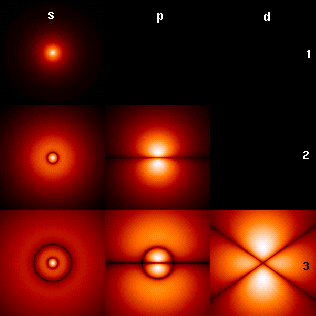
Hydrogen Atom
A hydrogen atom is an atom of the chemical element hydrogen. The electrically neutral atom contains a single positively charged proton and a single negatively charged electron bound to the nucleus by the Coulomb force. Atomic hydrogen constitutes about 75% of the baryonic mass of the universe. In everyday life on Earth, isolated hydrogen atoms (called "atomic hydrogen") are extremely rare. Instead, a hydrogen atom tends to combine with other atoms in compounds, or with another hydrogen atom to form ordinary ( diatomic) hydrogen gas, H2. "Atomic hydrogen" and "hydrogen atom" in ordinary English use have overlapping, yet distinct, meanings. For example, a water molecule contains two hydrogen atoms, but does not contain atomic hydrogen (which would refer to isolated hydrogen atoms). Atomic spectroscopy shows that there is a discrete infinite set of states in which a hydrogen (or any) atom can exist, contrary to the predictions of classical physics. Attempts to develop a theore ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Molecules
A molecule is a group of two or more atoms held together by attractive forces known as chemical bonds; depending on context, the term may or may not include ions which satisfy this criterion. In quantum physics, organic chemistry, and biochemistry, the distinction from ions is dropped and ''molecule'' is often used when referring to polyatomic ions. A molecule may be homonuclear, that is, it consists of atoms of one chemical element, e.g. two atoms in the oxygen molecule (O2); or it may be heteronuclear, a chemical compound composed of more than one element, e.g. water (molecule), water (two hydrogen atoms and one oxygen atom; H2O). In the kinetic theory of gases, the term ''molecule'' is often used for any gaseous particle regardless of its composition. This relaxes the requirement that a molecule contains two or more atoms, since the noble gases are individual atoms. Atoms and complexes connected by non-covalent interactions, such as hydrogen bonds or ionic bonds, are typic ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Rydberg Blockade
A Rydberg atom is an excited atom with one or more electrons that have a very high principal quantum number, ''n''. The higher the value of ''n'', the farther the electron is from the nucleus, on average. Rydberg atoms have a number of peculiar properties including an exaggerated response to electric and magnetic fields, long decay periods and electron wavefunctions that approximate, under some conditions, classical orbits of electrons about the nuclei. The core electrons shield the outer electron from the electric field of the nucleus such that, from a distance, the electric potential looks identical to that experienced by the electron in a hydrogen atom. In spite of its shortcomings, the Bohr model of the atom is useful in explaining these properties. Classically, an electron in a circular orbit of radius ''r'', about a hydrogen nucleus of charge +'' e'', obeys Newton's second law: : \mathbf=m\mathbf \Rightarrow = where ''k'' = 1/(4π ε0). Orbital momentum is quantiz ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |