|
Periodic Table Of The Elements
The periodic table, also known as the periodic table of the (chemical) elements, is a rows and columns arrangement of the chemical elements. It is widely used in chemistry, physics, and other sciences, and is generally seen as an icon of chemistry. It is a graphic formulation of the periodic law, which states that the properties of the chemical elements exhibit an approximate periodic dependence on their atomic numbers. The table is divided into four roughly rectangular areas called blocks. The rows of the table are called periods, and the columns are called groups. Elements from the same group of the periodic table show similar chemical characteristics. Trends run through the periodic table, with nonmetallic character (keeping their own electrons) increasing from left to right across a period, and from down to up across a group, and metallic character (surrendering electrons to other atoms) increasing in the opposite direction. The underlying reason for these trends is ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Simple Periodic Table Chart-blocks
Simple or SIMPLE may refer to: *Simplicity, the state or quality of being simple Arts and entertainment * Simple (album), ''Simple'' (album), by Andy Yorke, 2008, and its title track * Simple (Florida Georgia Line song), "Simple" (Florida Georgia Line song), 2018 * "Simple", a song by Johnny Mathis from the 1984 album ''A Special Part of Me'' * "Simple", a song by Collective Soul from the 1995 album ''Collective Soul (1995 album), Collective Soul'' * "Simple", a song by Katy Perry from the 2005 soundtrack to ''The Sisterhood of the Traveling Pants (film), The Sisterhood of the Traveling Pants'' * "Simple", a song by Khalil from the 2017 album ''Prove It All'' * "Simple", a song by Kreesha Turner from the 2008 album ''Passion (Kreesha Turner album), Passion'' * "Simple", a song by Ty Dolla Sign from the 2017 album ''Beach House 3'' deluxe version * Simple (video game series), ''Simple'' (video game series), budget-priced console games Businesses and organisations * Simple ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
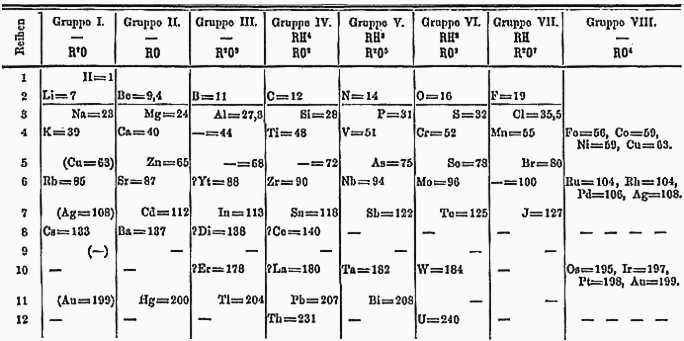
Dmitri Mendeleev
Dmitri Ivanovich Mendeleev (sometimes transliterated as Mendeleyev or Mendeleef) ( ; russian: links=no, Дмитрий Иванович Менделеев, tr. , ; 8 February Old_Style_and_New_Style_dates">O.S._27_January.html" ;"title="Old_Style_and_New_Style_dates.html" ;"title="nowiki/> O.S._27_January">Old_Style_and_New_Style_dates.html"_;"title="nowiki/>Old_Style_and_New_Style_dates">O.S._27_January18342_February_[O.S._20_January.html" ;"title="Old Style and New Style dates">O.S. 27 January">Old_Style_and_New_Style_dates.html" ;"title="nowiki/>Old Style and New Style dates">O.S. 27 January18342 February [O.S. 20 January">Old Style and New Style dates">O.S. 27 January">Old_Style_and_New_Style_dates.html" ;"title="nowiki/>Old Style and New Style dates">O.S. 27 January18342 February [O.S. 20 January1907) was a Russian chemist and inventor. He is best known for formulating the Periodic Law and creating a version of the periodic table, periodic table of elements. He used th ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chemical Reaction
A chemical reaction is a process that leads to the IUPAC nomenclature for organic transformations, chemical transformation of one set of chemical substances to another. Classically, chemical reactions encompass changes that only involve the positions of electrons in the forming and breaking of chemical bonds between atoms, with no change to the Atomic nucleus, nuclei (no change to the elements present), and can often be described by a chemical equation. Nuclear chemistry is a sub-discipline of chemistry that involves the chemical reactions of unstable and radioactive Chemical element, elements where both electronic and nuclear changes can occur. The substance (or substances) initially involved in a chemical reaction are called reagent, reactants or reagents. Chemical reactions are usually characterized by a chemical change, and they yield one or more Product (chemistry), products, which usually have properties different from the reactants. Reactions often consist of a sequence o ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Neutron
The neutron is a subatomic particle, symbol or , which has a neutral (not positive or negative) charge, and a mass slightly greater than that of a proton. Protons and neutrons constitute the nuclei of atoms. Since protons and neutrons behave similarly within the nucleus, and each has a mass of approximately one atomic mass unit, they are both referred to as nucleons. Their properties and interactions are described by nuclear physics. Protons and neutrons are not elementary particles; each is composed of three quarks. The chemical properties of an atom are mostly determined by the configuration of electrons that orbit the atom's heavy nucleus. The electron configuration is determined by the charge of the nucleus, which is determined by the number of protons, or atomic number. The number of neutrons is the neutron number. Neutrons do not affect the electron configuration, but the sum of atomic and neutron numbers is the mass of the nucleus. Atoms of a chemical element t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Proton
A proton is a stable subatomic particle, symbol , H+, or 1H+ with a positive electric charge of +1 ''e'' elementary charge. Its mass is slightly less than that of a neutron and 1,836 times the mass of an electron (the proton–electron mass ratio). Protons and neutrons, each with masses of approximately one atomic mass unit, are jointly referred to as "nucleons" (particles present in atomic nuclei). One or more protons are present in the nucleus of every atom. They provide the attractive electrostatic central force which binds the atomic electrons. The number of protons in the nucleus is the defining property of an element, and is referred to as the atomic number (represented by the symbol ''Z''). Since each element has a unique number of protons, each element has its own unique atomic number, which determines the number of atomic electrons and consequently the chemical characteristics of the element. The word ''proton'' is Greek for "first", and this name was given to the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Atomic Nucleus
The atomic nucleus is the small, dense region consisting of protons and neutrons at the center of an atom, discovered in 1911 by Ernest Rutherford based on the 1909 Geiger–Marsden gold foil experiment. After the discovery of the neutron in 1932, models for a nucleus composed of protons and neutrons were quickly developed by Dmitri Ivanenko and Werner Heisenberg. An atom is composed of a positively charged nucleus, with a cloud of negatively charged electrons surrounding it, bound together by electrostatic force. Almost all of the mass of an atom is located in the nucleus, with a very small contribution from the electron cloud. Protons and neutrons are bound together to form a nucleus by the nuclear force. The diameter of the nucleus is in the range of () for hydrogen (the diameter of a single proton) to about for uranium. These dimensions are much smaller than the diameter of the atom itself (nucleus + electron cloud), by a factor of about 26,634 (uranium atomic radiu ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Atom
Every atom is composed of a nucleus and one or more electrons bound to the nucleus. The nucleus is made of one or more protons and a number of neutrons. Only the most common variety of hydrogen has no neutrons. Every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. Atoms are extremely small, typically around 100 picometers across. They are so small that accurately predicting their behavior using classical physics, as if they were tennis balls for example, is not possible due to quantum effects. More than 99.94% of an atom's mass is in the nucleus. The protons have a positive electric charge, the electrons have a negative electric charge, and the neutrons have no electric charge. If the number of protons and electrons are equal, then the atom is electrically neutral. If an atom has more or fewer electrons than protons, then it has an overall negative or positive charge, respectively – such atoms are called ions. The electrons of an atom are a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Alternative Periodic Tables
Alternative periodic tables are tabulations of chemical elements differing in their organization from the traditional depiction of the periodic system. Over a thousand have been devised, often for didactic reasons, as not all correlations between the chemical elements are effectively captured by the standard periodic table. Major alternative structures Left-step periodic table (Janet, 1928) Charles Janet's left-step periodic table is the most widely used alternative to the traditional depiction of the periodic system. It organizes elements according to an idealized orbital filling (instead of valence). For example, the elements Sc to Zn are shown as a 3d block implying orbital occupancy r4s2 3dx. (Although Cr and Cu are exceptions in the gas-phase, the idealized configurations are not too far away from the ground state, and the energy difference is small enough to be controlled by the chemical environment.) Compared to the common layout, the left-step table has these ch ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Extended Periodic Table
An extended periodic table theorises about chemical elements beyond those currently known in the periodic table and proven. , the element with the highest atomic number known is oganesson (''Z'' = 118), which completes the seventh period (row) in the periodic table. All elements in the eighth period and beyond thus remain purely hypothetical. Elements beyond 118 will be placed in additional periods when discovered, laid out (as with the existing periods) to illustrate periodically recurring trends in the properties of the elements concerned. Any additional periods are expected to contain a larger number of elements than the seventh period, as they are calculated to have an additional so-called ''g-block'', containing at least 18 elements with partially filled g- orbitals in each period. An ''eight-period table'' containing this block was suggested by Glenn T. Seaborg in 1969. The first element of the g-block may have atomic number 121, and thus would have the systematic name unb ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Synthetic Element
A synthetic element is one of 24 known chemical elements that do not occur naturally on Earth: they have been created by human manipulation of fundamental particles in a nuclear reactor, a particle accelerator, or the explosion of an atomic bomb; thus, they are called "synthetic", "artificial", or "man-made". The synthetic elements are those with atomic numbers 95–118, as shown in purple on the accompanying periodic table: these 24 elements were first created between 1944 and 2010. The mechanism for the creation of a synthetic element is to force additional protons into the nucleus of an element with an atomic number lower than 95. All synthetic elements are unstable, but they decay at widely varying rates: the half-lives of their longest-lived isotopes range from microseconds to millions of years. Five more elements that were created artificially are strictly speaking not ''synthetic'' because they were later found in nature in trace quantities: 43Tc, 61Pm, 85At, 93Np, and ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Actinide
The actinide () or actinoid () series encompasses the 15 metallic chemical elements with atomic numbers from 89 to 103, actinium through lawrencium. The actinide series derives its name from the first element in the series, actinium. The informal chemical symbol An is used in general discussions of actinide chemistry to refer to any actinide. The 1985 IUPAC ''Red Book'' recommends that ''actinoid'' be used rather than ''actinide'', since the suffix ''-ide'' normally indicates a negative ion. However, owing to widespread current use, ''actinide'' is still allowed. Since ''actinoid'' literally means ''actinium-like'' (cf. ''humanoid'' or ''android''), it has been argued for semantic reasons that actinium cannot logically be an actinoid, but IUPAC acknowledges its inclusion based on common usage. All the actinides are f-block elements, except the final one (lawrencium) which is a d-block element. Actinium has sometimes been considered d-block instead of lawrencium, but the class ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |