|
Polysulfide
Polysulfides are a class of chemical compounds containing chains of sulfur atoms. There are two main classes of polysulfides: inorganic and organic. Among the inorganic polysulfides, there are ones which contain anions, which have the general formula . These anions are the conjugate bases of the hydrogen polysulfides . Organic polysulfides generally have the formulae , where R = alkyl or aryl. Polysulfide salts and complexes The alkali metal polysulfides arise by treatment of a solution of sulfide, e.g. sodium sulfide, with elemental sulfur: : In some cases, these anions have been obtained as organic salts, which are soluble in organic solvents. The energy released in the reaction of sodium and elemental sulfur is the basis of battery technology. The sodium–sulfur battery and the lithium–sulfur battery require high temperatures to maintain liquid polysulfide and -conductive membranes that are unreactive toward sodium, sulfur, and sodium sulfide. Polysulfides are ligands i ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Lithium–sulfur Battery
The lithium–sulfur battery (Li–S battery) is a type of rechargeable battery. It is notable for its high specific energy. The low atomic weight of lithium and moderate atomic weight of sulfur means that Li–S batteries are relatively light (about the density of water). They were used on the longest and highest-altitude unmanned solar-powered aeroplane flight (at the time) by Airbus Zephyr, Zephyr 6 in August 2008. Lithium–sulfur batteries may displace lithium-ion cells because of their higher energy density and reduced cost. This is due to the use of sulfur instead of cobalt, a common element in Lithium-ion battery, lithium-ion batteries. Li–S batteries offer specific energies on the order of 550Watt hour, Wh/kilogram, kg, while lithium-ion batteries are in the range of 150260Wh/kg. Li–S batteries with up to 1,500 charge and discharge cycles were demonstrated in 2017, but cycle life tests at commercial scale and with lean electrolyte have not been completed. As of ear ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sodium–sulfur Battery
A sodium–sulfur battery is a type of molten-salt battery constructed from liquid sodium (Na) and sulfur (S). This type of battery has a high energy density (its energy density is 5 times that of a lead-acid battery), high efficiency of charge/discharge and is fabricated from inexpensive and non-toxic materials. The operating temperatures of 300 to 350 °C and the highly corrosive nature of the sodium polysulfides, primarily make them suitable for stationary energy storage applications. The cell becomes more economical with increasing size. Commercially available cells are typically large with high capacities (up to 500Ah). This is because of the Square-cube law: large cells have less relative heat loss, so maintaining their high operating temperatures is easier. These batteries, although having a reasonably long long cycle life (>1000 on average) are prone to disastrous failures due to a reaction between molten sodium and molten sulfur , and primarily for this reasons ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Thiokol (polymer)
''Thiokol'' is a trade mark for various organic polysulfide polymers. Thiokol polymers are used as an elastomer in seals and sealants. The distinction between the polymers first commercialized by the Thiokol Corporation and subsequent polysulfide materials is often unclear. Preparation and structure A variety of thiokols are recognized. Typically they are prepared by the combination of 2-chloroethanol, formaldehyde, and sodium polysulfide (Na2S''x''). The chloroethanol is produced in situ from ethylene oxide and hydrogen chloride. The rank ''x'' of the polysulfide is an important variable. Crosslinking agents are used, such as 1,2,3-trichloropropane. An idealized polymer is represented by this formula HS(CH2CH2OCH2OCH2CH2SS)''n''CH2CH2OCH2OCH2CH2SH. Thiol-terminated resins can be cured oxidatively. History and etymology In 1838, Swiss chemists reported the preparation of hydrophobic rubbery materials by the alkylation of sodium polysulfide with 1,2-dichloroethane. In 1926 chemis ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sulfur
Sulfur (or sulphur in British English) is a chemical element with the symbol S and atomic number 16. It is abundant, multivalent and nonmetallic. Under normal conditions, sulfur atoms form cyclic octatomic molecules with a chemical formula S8. Elemental sulfur is a bright yellow, crystalline solid at room temperature. Sulfur is the tenth most abundant element by mass in the universe and the fifth most on Earth. Though sometimes found in pure, native form, sulfur on Earth usually occurs as sulfide and sulfate minerals. Being abundant in native form, sulfur was known in ancient times, being mentioned for its uses in ancient India, ancient Greece, China, and ancient Egypt. Historically and in literature sulfur is also called brimstone, which means "burning stone". Today, almost all elemental sulfur is produced as a byproduct of removing sulfur-containing contaminants from natural gas and petroleum.. Downloahere The greatest commercial use of the element is the production o ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sulfide
Sulfide (British English also sulphide) is an inorganic anion of sulfur with the chemical formula S2− or a compound containing one or more S2− ions. Solutions of sulfide salts are corrosive. ''Sulfide'' also refers to chemical compounds large families of inorganic and organic compounds, e.g. lead sulfide and dimethyl sulfide. Hydrogen sulfide (H2S) and bisulfide (SH−) are the conjugate acids of sulfide. Chemical properties The sulfide ion, S2−, does not exist in aqueous alkaline solutions of Na2S. Instead sulfide converts to hydrosulfide: :S2− + H2O → SH− + OH− Upon treatment with an acid, sulfide salts convert to hydrogen sulfide: :S2− + H+ → SH− :SH− + H+ → H2S Oxidation of sulfide is a complicated process. Depending on the conditions, the oxidation can produce elemental sulfur, polysulfides, polythionates, sulfite, or sulfate. Metal sulfides react with halogens, forming sulfur and metal salts. :8 MgS + 8 I2 → S8 + 8 M ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sodium Sulfide
Sodium sulfide is a chemical compound with the formula Na2 S, or more commonly its hydrate Na2S·9 H2O. Both the anhydrous and the hydrated salts in pure crystalline form are colorless solids, although technical grades of sodium sulfide are generally yellow to brick red owing to the presence of polysulfides and commonly supplied as a crystalline mass, in flake form, or as a fused solid. They are water-soluble, giving strongly alkaline solutions. When exposed to moist air, Na2S and its hydrates emit hydrogen sulfide, an extremely toxic, flammable and corrosive gas which smells like rotten eggs. Some commercial samples are specified as Na2S·''x''H2O, where a weight percentage of Na2S is specified. Commonly available grades have around 60% Na2S by weight, which means that ''x'' is around 3. These grades of sodium sulfide are often marketed as 'sodium sulfide flakes'. Structure Na2S adopts the antifluorite structure, which means that the Na+ centers occupy sites of the fluoride in ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Curing (chemistry)
Curing is a chemical process employed in polymer chemistry and process engineering that produces the toughening or hardening of a polymer material by cross-linking of polymer chains. Even if it is strongly associated with the production of thermosetting polymers, the term "curing" can be used for all the processes where a solid product is obtained from a liquid solution, such as with PVC plastisols. Curing process During the curing process, single monomers and oligomers, mixed with or without a curing agent, react to form a tridimensional polymeric network. In the very first part of the reaction branches of molecules with various architectures are formed, and their molecular weight increases in time with the extent of the reaction until the network size is equal to the size of the system. The system has lost its solubility and its viscosity tends to infinite. The remaining molecules start to coexist with the macroscopic network until they react with the network creating other c ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Poly(p-phenylene Sulfide)
Polyphenylene sulfide (PPS) is an organic polymer consisting of aromatic rings linked by sulfides. Synthetic fiber and textiles derived from this polymer resist chemical and thermal attack. PPS is used in filter fabric for coal boilers, papermaking felts, electrical insulation, film capacitors, specialty membranes, gaskets, and packings. PPS is the precursor to a conductive polymer of the semi-flexible rod polymer family. The PPS, which is otherwise insulating, can be converted to the semiconducting form by oxidation or use of dopants.David Parker, Jan Bussink, Hendrik T. van de Grampel, Gary W. Wheatley, Ernst-Ulrich Dorf, Edgar Ostlinning, Klaus Reinking, "Polymers, High-Temperature" in Ullmann's Encyclopedia of Industrial Chemistry 2002, Wiley-VCH: Weinheim. Polyphenylene sulfide is an engineering plastic, commonly used today as a high-performance thermoplastic. PPS can be molded, extruded, or machined to tight tolerances. In its pure solid form, it may be opaque white to ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Titanocene Pentasulfide
Titanocene pentasulfide is the organotitanium compound with the formula (C5H5)2TiS5, commonly abbreviated as Cp2TiS5. This metallocene exists as a bright red solid that is soluble in organic solvents. It is of academic interest as a precursor to unusual allotropes of elemental sulfur as well as some related inorganic rings. Preparation and structure Titanocene pentasulfide is prepared by treating Cp2TiCl2 with polysulfide salts: It was first produced by the addition of elemental sulfur to titanocene dicarbonyl: :(C5H5)2Ti(CO)2 + S8 → (C5H5)2TiS5 + 2 CO The complex is viewed as a pseudotetrahedral complex of Ti(IV). The Ti–S distances are 2.420 and 2.446 Å and the S–S bond distances are of a normal range, 2.051–2.059 Å. The molecule exhibits a dynamic NMR spectrum owing to the chair–chair equilibrium of the TiS5 ring which equivalizes the Cp signals at high temperatures. Reactions Cp2TiS5 reacts with sulfur and selenium chlorides, E''x''Cl2, to ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Rubber
Rubber, also called India rubber, latex, Amazonian rubber, ''caucho'', or ''caoutchouc'', as initially produced, consists of polymers of the organic compound isoprene, with minor impurities of other organic compounds. Thailand, Malaysia, and Indonesia are three of the leading rubber producers. Types of polyisoprene that are used as natural rubbers are classified as elastomers. Currently, rubber is harvested mainly in the form of the latex from the rubber tree (''Hevea brasiliensis'') or others. The latex is a sticky, milky and white colloid drawn off by making incisions in the bark and collecting the fluid in vessels in a process called "tapping". The latex then is refined into the rubber that is ready for commercial processing. In major areas, latex is allowed to coagulate in the collection cup. The coagulated lumps are collected and processed into dry forms for sale. Natural rubber is used extensively in many applications and products, either alone or in combination wit ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cross-link
In chemistry and biology a cross-link is a bond or a short sequence of bonds that links one polymer chain to another. These links may take the form of covalent bonds or ionic bonds and the polymers can be either synthetic polymers or natural polymers (such as proteins). In polymer chemistry "cross-linking" usually refers to the use of cross-links to promote a change in the polymers' physical properties. When "crosslinking" is used in the biological field, it refers to the use of a probe to link proteins together to check for protein–protein interactions, as well as other creative cross-linking methodologies. Although the term is used to refer to the "linking of polymer chains" for both sciences, the extent of crosslinking and specificities of the crosslinking agents vary greatly. As with all science, there are overlaps, and the following delineations are a starting point to understanding the subtleties. Polymer chemistry Crosslinking is the general term for the process of ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
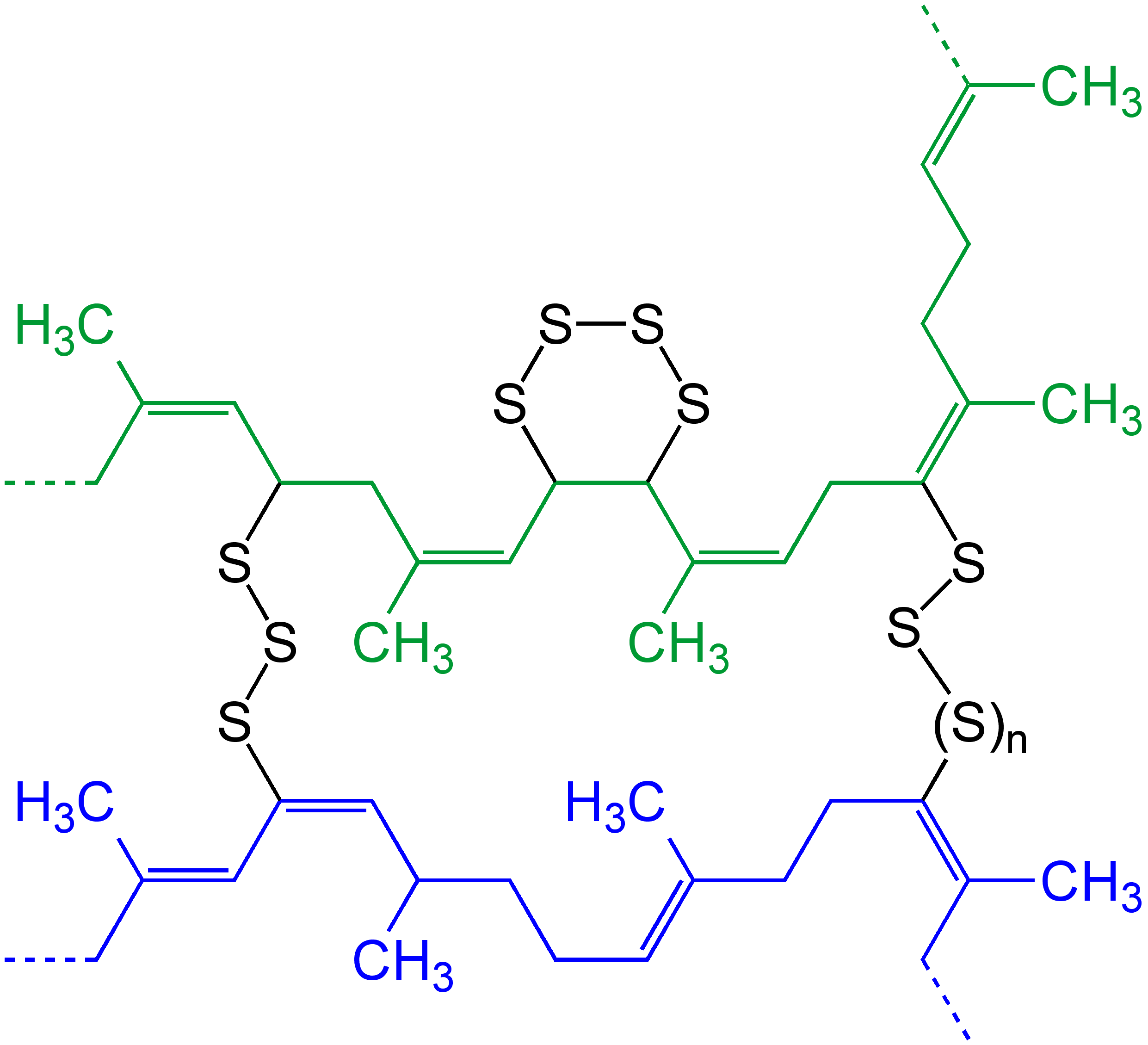
Vulcanization Of POLYIsoprene V
Vulcanization (British: Vulcanisation) is a range of processes for hardening rubbers. The term originally referred exclusively to the treatment of natural rubber with sulfur, which remains the most common practice. It has also grown to include the hardening of other (synthetic) rubbers via various means. Examples include silicone rubber via room temperature vulcanizing and chloroprene rubber (neoprene) using metal oxides. Vulcanization can be defined as the curing of elastomers, with the terms 'vulcanization' and 'curing' sometimes used interchangeably in this context. It works by forming cross-links between sections of polymer chain which results in increased rigidity and durability, as well as other changes in the mechanical and electrical properties of the material. Vulcanization, in common with the curing of other thermosetting polymers, is generally irreversible. The word vulcanization is derived from Vulcan, the Roman god of fire and forge. History Rubber—latex— ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |