|
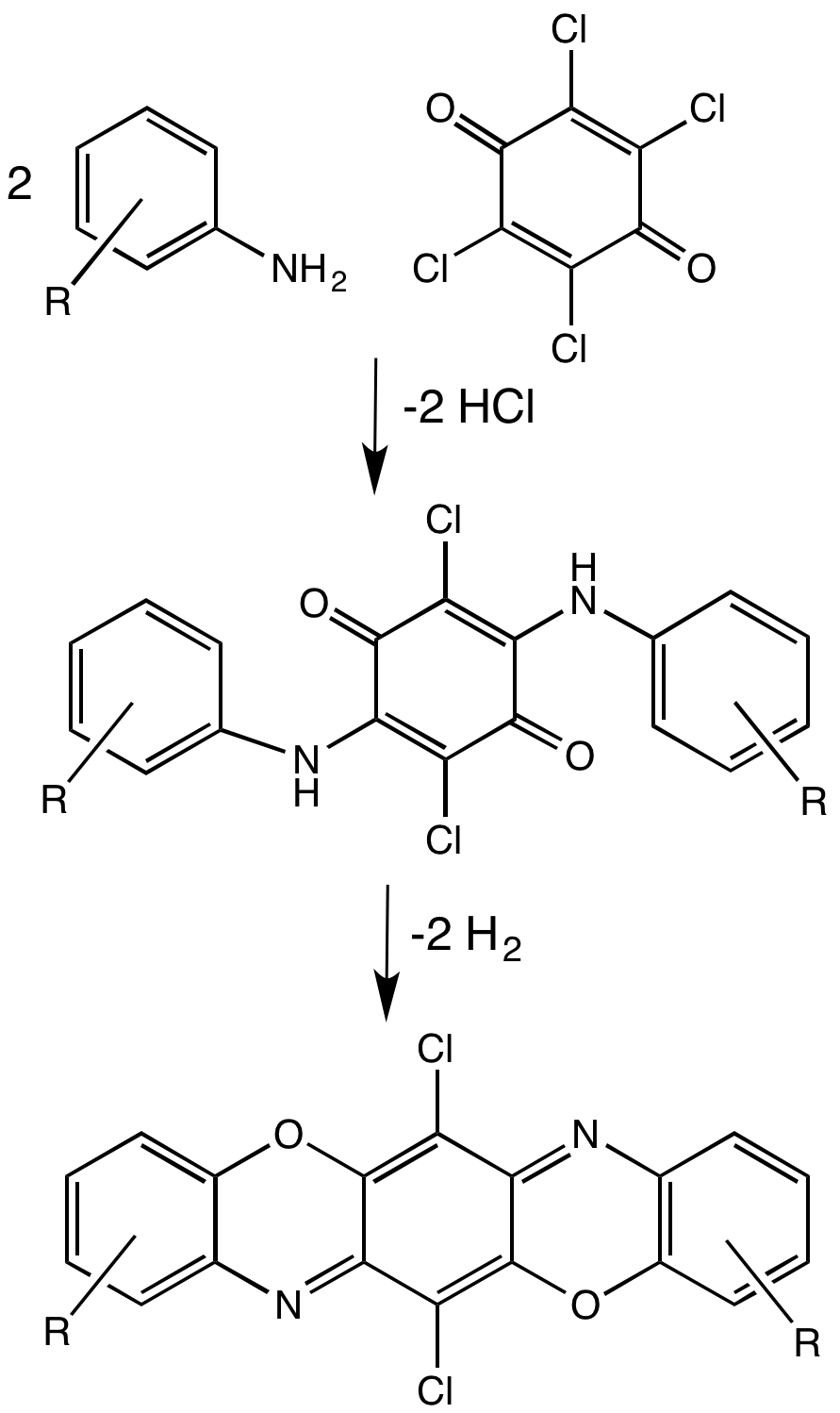
P-chloranil
Chloranil is a quinone with the molecular formula C6Cl4O2. Also known as tetrachloro-1,4-benzoquinone, it is a yellow solid. Like the parent benzoquinone, chloranil is a planar molecule J.-M. Lü, S. V. Rosokha, I. S. Neretin and J. K. Kochi, "Quinones as Electron Acceptors. X-Ray Structures, Spectral (EPR, UV-vis) Characteristics and Electron-Transfer Reactivities of Their Reduced Anion Radicals as Separated vs Contact Ion Pairs" Journal of the American Chemical Society 2006 128, 16708-16719. that functions as a mild oxidant. Synthesis and use as reagent Chloranil is produced by chlorination of phenol to give hexachlorocyclohexa-2,5-dien-1-one ("hexachlorophenol"). Hydrolysis of the dichloromethylene group in this dienone gives chloranil: :C6H5OH + 6 Cl2 → C6Cl6O + 6 HCl :C6Cl6O + H2O → C6Cl4O2 + 2 HCl Chloroanil serves as a hydrogen acceptor. It is more electrophilic than quinone itself. It is used for the aromatization reactions, such as the conversion of cy ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sigma-Aldrich
Sigma-Aldrich (formally MilliporeSigma) is an American chemical, life science, and biotechnology company that is owned by the German chemical conglomerate Merck Group. Sigma-Aldrich was created in 1975 by the merger of Sigma Chemical Company and Aldrich Chemical Company. It grew through various acquisitions until it had over 9,600 employees and was listed on the Fortune 1000. The company is headquartered in St. Louis and has operations in approximately 40 countries. In 2015, the German chemical conglomerate Merck Group acquired Sigma-Aldrich for $17 billion. The company is currently a part of Merck's life science business and in combination with Merck's earlier acquired Millipore Corporation, Millipore, operates as MilliporeSigma. History Sigma Chemical Company of St. Louis and Aldrich Chemical Company of Milwaukee were both American specialty chemical companies when they merged in August 1975. The company grew throughout the 1980s and 1990s, with significant expansion in fac ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Quinone
The quinones are a class of organic compounds that are formally "derived from aromatic compounds [such as benzene or naphthalene] by conversion of an even number of –CH= groups into –C(=O)– groups with any necessary rearrangement of double bonds, resulting in "a fully Conjugated system, conjugated cyclic diketone, dione structure". The archetypical member of the class is 1,4-benzoquinone or cyclohexadienedione, often called simply "quinone" (thus the name of the class). Other important examples are 1,2-benzoquinone (''ortho''-quinone), 1,4-naphthoquinone and anthraquinone, 9,10-anthraquinone. The name is derived from that of quinic acid (with the suffix "-one" indicating a ketone), since it is one of the compounds obtained upon oxidation of quinic acid. Quinic acid, like quinine is obtained from cinchona bark, called wikt:quinaquina, quinaquina in the indigenous languages of Peruvian tribes. Properties Quinones are oxidized derivatives of aromatic compounds and are often re ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hexachlorocyclohexa-2,5-dien-1-one
Hexachlorocyclohexa-2,5-dien-1-one, sometimes informally called hexachlorophenol (HCP) is an organochlorine compound. It can be prepared from phenol. Despite the informal name, the compound is not a phenol but is a ketone. The informal name is derived from its method of preparation which includes phenol as a reagent. Preparation HCP is normally produced by chlorination of phenol by chlorine in the presence of metal chloride catalyst, such as ferric chloride. It can also be produced by alkaline hydrolysis of polychlorinatedbenzenes at high temperature and pressure, by conversion of diazonium salts of chlorinated anilines, or by chlorination of phenolsulfonic acids and benzenesulfonic acids followed by removal of the sulfonic acid group. The hydrolysis of HCP gives chloranil.{{Ullmann, authors=François Muller, Liliane Caillard, title=Chlorophenols, year=2011, doi=10.1002/14356007.a07_001.pub2 References See also * Pentachlorophenol * Hexachlorobenzene Hexachlorobenzene, or per ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cyclohexadiene
{{chemistry index ...
Cyclohexadiene may refer to: * 1,3-Cyclohexadiene, * 1,4-Cyclohexadiene, See also * Benzene or its theoretical isomer ''1,3,5-Cyclohexatriene'' * Cyclohexene Cyclohexene is a hydrocarbon with the formula C6H10. This cycloalkene is a colorless liquid with a sharp smell. It is an intermediate in various industrial processes. Cyclohexene is not very stable upon long term storage with exposure to light an ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Proline
Proline (symbol Pro or P) is an organic acid classed as a proteinogenic amino acid (used in the biosynthesis of proteins), although it does not contain the amino group but is rather a secondary amine. The secondary amine nitrogen is in the protonated form (NH2+) under biological conditions, while the carboxyl group is in the deprotonated −COO− form. The "side chain" from the α carbon connects to the nitrogen forming a pyrrolidine loop, classifying it as a aliphatic amino acid. It is non-essential in humans, meaning the body can synthesize it from the non-essential amino acid L-glutamate. It is encoded by all the codons starting with CC (CCU, CCC, CCA, and CCG). Proline is the only proteinogenic secondary amino acid which is a secondary amine, as the nitrogen atom is attached both to the α-carbon and to a chain of three carbons that together form a five-membered ring. History and etymology Proline was first isolated in 1900 by Richard Willstätter who obtained the amino ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pigment Violet 23
Pigment violet 23 is an organic compound that is a commercial pigment. It is member of the dioxazine family of heterocyclic compounds, but derived from carbazoles. It is prepared by condensation of chloranil and 3-amino-''N''-ethylcarbazole. It has a centrosymmetric angular structure. For many years, the structure was assigned, incorrectly, as having a "linear structure" (European Community number, EC no. 228-767-9, CAS RN 6358-30-1) which differ in terms of the carbazole ring fusion. Pigment violet 23 is prepared by condensation of an aniline with chloranil.Horst Tappe, Walter Helmling, Peter Mischke, Karl Rebsamen, Uwe Reiher, Werner Russ, Ludwig Schläfer and Petra Vermehren "Reactive Dyes"in Ullmann's Encyclopedia of Industrial Chemistry 2000, Wiley-VCH, Weinheim. {{doi, 10.1002/14356007.a22_651 References [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chloranilic Acid
Chloranilic acid is an organic compound with the chemical formula . It is a red-orange solid. The compound is obtained by hydrolysis of chloranil: : It is centrosymmetric, planar molecule. It also crystallizes as a dihydrate. Chloranilic acid is a noteworthy hydroxyquinone that is somewhat acidic owing to the presence of the two chloride substituents. The conjugate base, C6H2Cl2O4]2- readily forms coordination complexes often linking pairs of many metal ions. See also * Chloranil Chloranil is a quinone with the molecular formula C6Cl4O2. Also known as tetrachloro-1,4-benzoquinone, it is a yellow solid. Like the parent benzoquinone, chloranil is a planar molecule J.-M. Lü, S. V. Rosokha, I. S. Neretin and J. K. Kochi, "Qui ... References {{ketone-stub Chlorobenzenes 1,4-Benzoquinones Hydroxybenzoquinones ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
2,3-Dichloro-5,6-dicyano-1,4-benzoquinone
2,3-Dichloro-5,6-dicyano-1,4-benzoquinone (or DDQ) is the chemical reagent with formula C6Cl2(CN)2O2. This oxidant is useful for the dehydrogenation of alcohols, phenols, and steroid ketones. DDQ decomposes in water, but is stable in aqueous mineral acid. Preparation Synthesis of DDQ involves cyanation of chloranil. J. Thiele and F. Günther first reported a 6-step preparation in 1906. The substance did not receive interest until its potential as a dehydrogenation agent was discovered. A single-step chlorination from 2,3-dicyanohydroquinone was reported in 1965. Reactions The reagent removes pairs of H atoms from organic molecules. The stoichiometry of its action is illustrated by the conversion of tetralin to naphthalene: :2 C6Cl2(CN)2O2 + C10H12 → 2 C6Cl2(CN)2(OH)2 + C10H8 The resulting hydroquinone is poorly soluble in typical reaction solvents (dioxane, benzene, alkanes), which facilitates workup. Solutions of DDQ in benzene are red, due to the formation of a charge-tra ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Organochlorides
An organochloride, organochlorine compound, chlorocarbon, or chlorinated hydrocarbon is an organic compound containing at least one covalently bonded atom of chlorine. The chloroalkane class (alkanes with one or more hydrogens substituted by chlorine) provides common examples. The wide structural variety and divergent chemical properties of organochlorides lead to a broad range of names, applications, and properties. Organochlorine compounds have wide use in many applications, though some are of profound environmental concern, with TCDD being one of the most notorious. Physical and chemical properties Chlorination modifies the physical properties of hydrocarbons in several ways. These compounds are typically denser than water due to the higher atomic weight of chlorine versus hydrogen. Aliphatic organochlorides are often alkylating agents as chlorine can act as a leaving group, which can result in cellular damage. Natural occurrence Many organochlorine compounds have been isolate ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Fungicides
Fungicides are biocidal chemical compounds or biological organisms used to kill parasitic fungi or their spores. A fungistatic inhibits their growth. Fungi can cause serious damage in agriculture, resulting in critical losses of yield, quality, and profit. Fungicides are used both in agriculture and to fight fungal infections in animals. Chemicals used to control oomycetes, which are not fungi, are also referred to as fungicides, as oomycetes use the same mechanisms as fungi to infect plants. Fungicides can either be contact, translaminar or systemic. Contact fungicides are not taken up into the plant tissue and protect only the plant where the spray is deposited. Translaminar fungicides redistribute the fungicide from the upper, sprayed leaf surface to the lower, unsprayed surface. Systemic fungicides are taken up and redistributed through the xylem vessels. Few fungicides move to all parts of a plant. Some are locally systemic, and some move upwardly. Most fungicides that can ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |