|
Organomolybdenum Compound
Organomolybdenum chemistry is the chemistry of chemical compounds with Mo-C bonds. The heavier group 6 elements molybdenum and tungsten form organometallic compounds similar to those in organochromium chemistry but higher oxidation states tend to be more common. Mo(0) and more reduced states Molybdenum hexacarbonyl is the precursor to many substituted derivatives. It reacts with organolithium reagents to give anionic acyls which can be O-alkylated to give Fischer carbenes. 144px, Structure of (mesitylene)molybdenum tricarbonyl. Mo(CO)6 reacts with arenes to give piano-stool complexes such as (mesitylene)molybdenum tricarbonyl. Cycloheptatrienemolybdenum tricarbonyl, which is related to (arene)Mo(CO)3, reacts with trityl salts to give the cycloheptatrienyl complex: :(C7H8)Mo(CO)3 + (C6H5)3C+ → C7H7)Mo(CO)3sup>+ + (C6H5)3CH file:CHTMo(CO)3.png, 144px, Structure of Cycloheptatrienemolybdenum tricarbonyl. Reduction of Mo(CO)6 gives [Mo(CO)5]2− which is formally M ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Molybdenum(V) Chloride
Molybdenum(V) chloride is the inorganic compound with the empirical formula . This dark volatile solid is used in research to prepare other molybdenum compounds. It is moisture-sensitive and soluble in chlorinated solvents. Structure Usually called molybdenum pentachloride, it is in fact partly a dimer with the molecular formula . In the dimer, each molybdenum has local octahedral symmetry and two chlorides bridge between the molybdenum centers. A similar structure is also found for the pentachlorides of W, Nb and Ta. In the gas phase and partly in solution, the dimers partially dissociate to give a monomeric . The monomer is paramagnetic, with one unpaired electron per Mo center, reflecting the fact that the formal oxidation state is +5, leaving one valence electron on the metal center. Preparation and properties is prepared by chlorination of Mo metal but also chlorination of . The unstable hexachloride is not produced in this way. is reduced by acetonitrile to afford an or ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Stoichiometric
Stoichiometry refers to the relationship between the quantities of reactants and products before, during, and following chemical reactions. Stoichiometry is founded on the law of conservation of mass where the total mass of the reactants equals the total mass of the products, leading to the insight that the relations among quantities of reactants and products typically form a ratio of positive integers. This means that if the amounts of the separate reactants are known, then the amount of the product can be calculated. Conversely, if one reactant has a known quantity and the quantity of the products can be empirically determined, then the amount of the other reactants can also be calculated. This is illustrated in the image here, where the balanced equation is: : Here, one molecule of methane reacts with two molecules of oxygen gas to yield one molecule of carbon dioxide and two molecules of water. This particular chemical equation is an example of complete combustion. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
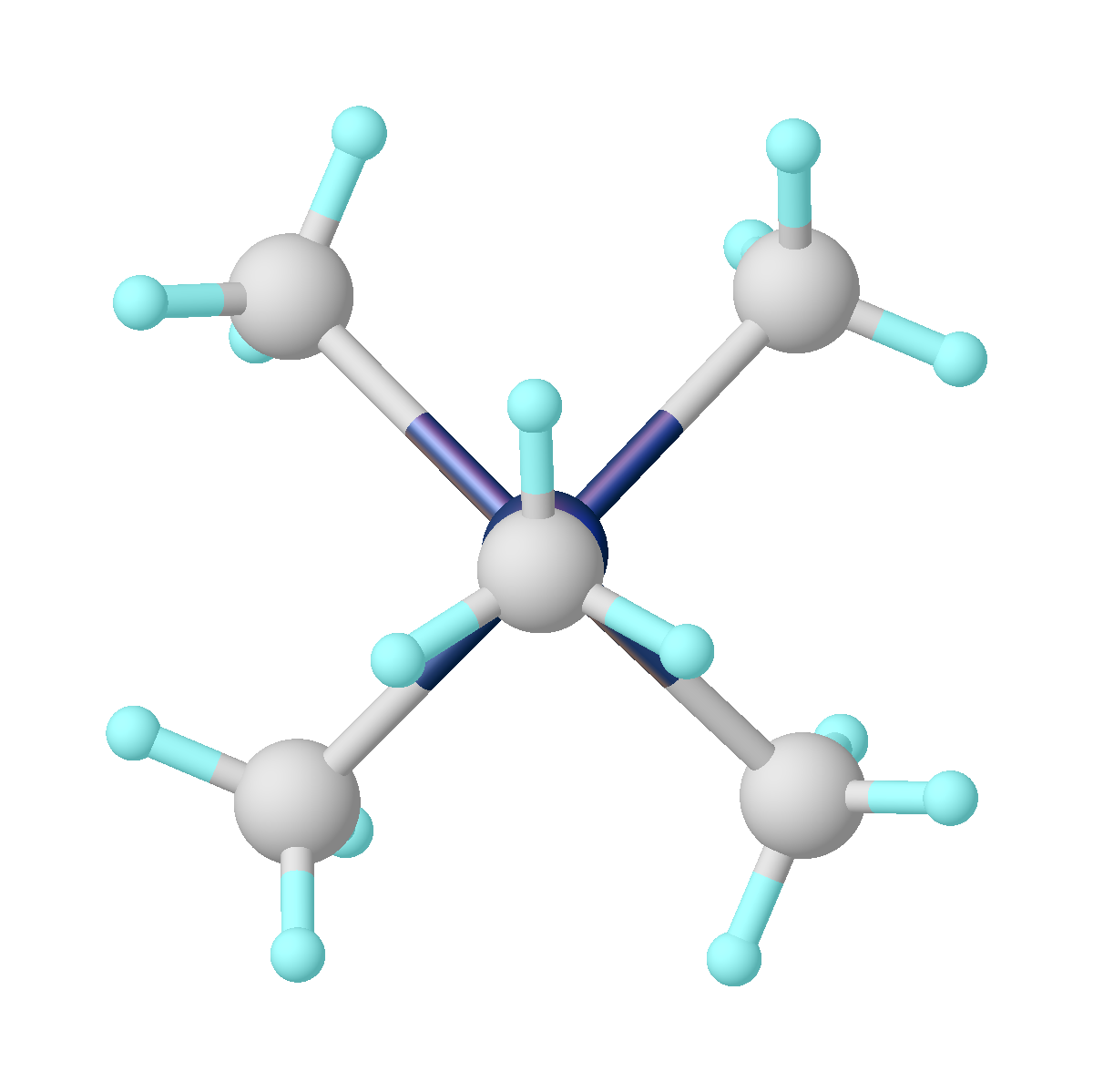
Cummins First Mo Example
Cummins Inc. is an American multinational corporation that designs, manufactures, and distributes engines, filtration, and power generation products. Cummins also services engines and related equipment, including fuel systems, controls, air handling, filtration, emission control, electrical power generation systems, and trucks. Headquartered in Columbus, Indiana, Cummins sells in approximately 190 countries and territories through a network of more than 600 company-owned and independent distributors and approximately 7,200 dealers. Cummins reported a net income of $2.13 billion on sales of $24.02 billion in 2021. History The Cummins Engine Company was founded in Columbus, Indiana, on February 3, 1919, by mechanic Clessie Cummins and banker William Glanton Irwin. The company focused on developing the diesel engine invented 20 years earlier, but despite several well-publicized endurance trials, it was not until 1933, that their Model H engine, used in small railroad swi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Alkylidene
A transition metal carbene complex is an organometallic compound featuring a divalent organic ligand. The divalent organic ligand coordinated to the metal center is called a carbene. Carbene complexes for almost all transition metals have been reported. Many methods for synthesizing them and reactions utilizing them have been reported. The term carbene ligand is a formalism since many are not derived from carbenes and almost none exhibit the reactivity characteristic of carbenes. Described often as , they represent a class of organic ligands intermediate between alkyls and carbynes . They feature in some catalytic reactions, especially alkene metathesis, and are of value in the preparation of some fine chemicals. Classification Metal carbene complexes are often classified into two types. The Fischer carbenes named after Ernst Otto Fischer feature strong π-acceptors at the metal and being electrophilic at the carbene carbon atom. Schrock carbenes, named after Richard R. Schroc ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Alkoxide
In chemistry, an alkoxide is the conjugate base of an alcohol and therefore consists of an organic group bonded to a negatively charged oxygen atom. They are written as , where R is the organic substituent. Alkoxides are strong bases and, when R is not bulky, good nucleophiles and good ligands. Alkoxides, although generally not stable in protic solvents such as water, occur widely as intermediates in various reactions, including the Williamson ether synthesis. Transition metal alkoxides are widely used for coatings and as catalysts. Enolates are unsaturated alkoxides derived by deprotonation of a bond adjacent to a ketone or aldehyde. The nucleophilic center for simple alkoxides is located on the oxygen, whereas the nucleophilic site on enolates is delocalized onto both carbon and oxygen sites. Ynolates are also unsaturated alkoxides derived from acetylenic alcohols. Phenoxides are close relatives of the alkoxides, in which the alkyl group is replaced by a derivative ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Olefin Metathesis
Olefin metathesis is an organic reaction that entails the redistribution of fragments of alkenes (olefins) by the scission and regeneration of carbon-carbon double bonds. Because of the relative simplicity of olefin metathesis, it often creates fewer undesired by-products and hazardous wastes than alternative organic reactions. For their elucidation of the reaction mechanism and their discovery of a variety of highly active catalysts, Yves Chauvin, Robert H. Grubbs, and Richard R. Schrock were collectively awarded the 2005 Nobel Prize in Chemistry. Catalysts The reaction requires metal catalysts. Most commercially important processes employ heterogeneous catalysts. The heterogeneous catalysts are often prepared by in-situ activation of a metal halides (MClx) using organoaluminium or organotin compounds, e.g. combining MClx–EtAlCl2. A typical catalyst support is alumina. Commercial catalysts are often based on molybdenum and ruthenium. Well-defined organometallic compo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Transition Metal Imido Complex
In coordination chemistry and organometallic chemistry, transition metal imido complexes is a coordination compound containing an imido ligand. Imido ligands can be terminal or bridging ligands. The parent imido ligand has the formula NH, but most imido ligands have alkyl or aryl groups in place of H. The imido ligand is generally viewed as a dianion, akin to oxide. Structural classes Complexes with terminal imido ligands In some terminal imido complexes, the M=N−C angle is 180° but often the angle is decidedly bent. Complexes of the type M=NH are assumed to be intermediates in nitrogen fixation by synthetic catalysts.Nugent, W. A.; Mayer, J. M., "Metal-Ligand Multiple Bonds," J. Wiley: New York, 1988. 220px, Typical Schrock-style olefin metathesis catalyst features imides as spectator ligands.">spectator_ligand.html" ;"title="olefin metathesis catalyst features imides as spectator ligand">olefin metathesis catalyst features imides as spectator ligands. Complexes with bridg ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Metal Oxo Complex
A transition metal oxo complex is a coordination complex containing an oxo ligand. Formally O2-, an oxo ligand can be bound to one or more metal centers, i.e. it can exist as a terminal or (most commonly) as bridging ligands (Fig. 1). Oxo ligands stabilize high oxidation states of a metal.Nugent, W. A., Mayer, J. M. "Metal-Ligand Multiple Bonds." John Wiley & Sons, New York, 1988. They are also found in several metalloproteins, for example in molybdenum cofactors and in many iron-containing enzymes. One of the earliest synthetic compounds to incorporate an oxo ligand is potassium ferrate (K2FeO4), which was likely prepared by Georg E. Stahl in 1702. Reactivity Olation and acid-base reactions A common reaction exhibited by metal-oxo compounds is olation, the condensation process that converts low molecular weight oxides to polymers with M-O-M linkages. Olation often begins with the deprotonation of a metal-hydroxo complex. It is the basis for mineralization and the precipita ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cp2MoH2
Molybdocene dihydride is the organomolybdenum compound with the formula ( η5-C5H5)2MoH2. Commonly abbreviated as Cp2MoH2, it is a yellow air-sensitive solid that dissolves in some organic solvents. The compound is prepared by combining molybdenum pentachloride, sodium cyclopentadienide, and sodium borohydride. The dihydride converts to molybdocene dichloride upon treatment with chloroform Chloroform, or trichloromethane, is an organic compound with formula C H Cl3 and a common organic solvent. It is a colorless, strong-smelling, dense liquid produced on a large scale as a precursor to PTFE. It is also a precursor to various .... The compound adopts a "clamshell" structure where the Cp rings are not parallel.K. Prout, T. S. Cameron, R. A. Forder, and in parts S. R. Critchley, B. Denton and G. V. Rees "The crystal and molecular structures of bent bis-π-cyclopentadienyl-metal complexes: (a) bis-π-cyclopentadienyldibromorhenium(V) tetrafluoroborate, (b) bis-π-cyclopen ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ansa Metallocene
An ''ansa''-metallocene is a type of organometallic compound containing two cyclopentadienyl ligands that are linked by a bridging group such that both cyclopentadienyl groups are bound to the same metal. The link prevents rotation of the cyclopentadienyl ligand and often modifies the structure and reactivity of the metal center. Some ansa-metallocenes are active in Ziegler-Natta catalysis, although none are used commercially. The term ''ansa''-metallocene (ansa being Greek for "handle") was coined by Lüttringhaus and Kullick to describe alkylidene-bridged ferrocenes, which were developed in the 1950s. Often ''ansa''-metallocenes are described in terms of the angle defined by the two Cp rings. In titanocene dichloride, this angle is 58.5° whereas in the ''ansa''-titanocene Me2Si(C5H4)2TiCl2 the angle is 51.2°. The prototypical linker groups are of the type (CH2)n where n = 1, 2, and 3. More easily installed are linker groups consisting of heteroatoms, e.g. (CH3)2Si. One ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Molybdocene Dihydride
Molybdocene dihydride is the organomolybdenum compound with the formula ( η5-C5H5)2MoH2. Commonly abbreviated as Cp2MoH2, it is a yellow air-sensitive solid that dissolves in some organic solvents. The compound is prepared by combining molybdenum pentachloride, sodium cyclopentadienide, and sodium borohydride. The dihydride converts to molybdocene dichloride upon treatment with chloroform Chloroform, or trichloromethane, is an organic compound with formula C H Cl3 and a common organic solvent. It is a colorless, strong-smelling, dense liquid produced on a large scale as a precursor to PTFE. It is also a precursor to various .... The compound adopts a "clamshell" structure where the Cp rings are not parallel.K. Prout, T. S. Cameron, R. A. Forder, and in parts S. R. Critchley, B. Denton and G. V. Rees "The crystal and molecular structures of bent bis-π-cyclopentadienyl-metal complexes: (a) bis-π-cyclopentadienyldibromorhenium(V) tetrafluoroborate, (b) bis-π-cyclopen ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |


imidazol-2-ylidene_(aka_IMes).png)


