|
Nuclear Shell
In nuclear physics, atomic physics, and nuclear chemistry, the nuclear shell model is a model of the atomic nucleus which uses the Pauli exclusion principle to describe the structure of the nucleus in terms of energy levels. The first shell model was proposed by Dmitri Ivanenko (together with E. Gapon) in 1932. The model was developed in 1949 following independent work by several physicists, most notably Eugene Paul Wigner, Maria Goeppert Mayer and J. Hans D. Jensen, who shared the 1963 Nobel Prize in Physics for their contributions. The nuclear shell model is partly analogous to the atomic shell model, which describes the arrangement of electrons in an atom in that filled shell results in better stability. When adding nucleons (protons or neutrons) to a nucleus, there are certain points where the binding energy of the next nucleon is significantly less than the last one. This observation that there are specific magic quantum numbers of nucleons (2, 8, 20, 28, 50, 82, 126) wh ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nuclear Physics
Nuclear physics is the field of physics that studies atomic nuclei and their constituents and interactions, in addition to the study of other forms of nuclear matter. Nuclear physics should not be confused with atomic physics, which studies the atom as a whole, including its electrons. Discoveries in nuclear physics have led to applications in many fields. This includes nuclear power, nuclear weapons, nuclear medicine and magnetic resonance imaging, industrial and agricultural isotopes, ion implantation in materials engineering, and radiocarbon dating in geology and archaeology. Such applications are studied in the field of nuclear engineering. Particle physics evolved out of nuclear physics and the two fields are typically taught in close association. Nuclear astrophysics, the application of nuclear physics to astrophysics, is crucial in explaining the inner workings of stars and the origin of the chemical elements. History The history of nuclear physics as a discipl ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Flerovium
Flerovium is a superheavy chemical element with symbol Fl and atomic number 114. It is an extremely radioactive synthetic element. It is named after the Flerov Laboratory of Nuclear Reactions of the Joint Institute for Nuclear Research in Dubna, Russia, where the element was discovered in 1998. The lab's name, in turn, honours Russian physicist Georgy Flyorov ( in Cyrillic, hence the transliteration of " yo" to "e"). IUPAC adopted the name on 30 May 2012. The name and symbol had previously been proposed for element 102 (nobelium), but was not accepted by IUPAC at that time. It is a transactinide in the p-block of the periodic table. It is in period 7; the heaviest known member of the carbon group, and the last element whose chemistry has been investigated. Initial chemical studies in 2007–2008 indicated that flerovium was unexpectedly volatile for a group 14 element; in preliminary results it even seemed to exhibit properties similar to noble gases. More recent results show ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Periodic Table
The periodic table, also known as the periodic table of the (chemical) elements, is a rows and columns arrangement of the chemical elements. It is widely used in chemistry, physics, and other sciences, and is generally seen as an icon of chemistry. It is a graphic formulation of the periodic law, which states that the properties of the chemical elements exhibit an approximate periodic dependence on their atomic numbers. The table is divided into four roughly rectangular areas called blocks. The rows of the table are called periods, and the columns are called groups. Elements from the same group of the periodic table show similar chemical characteristics. Trends run through the periodic table, with nonmetallic character (keeping their own electrons) increasing from left to right across a period, and from down to up across a group, and metallic character (surrendering electrons to other atoms) increasing in the opposite direction. The underlying reason for these trends is ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Neutron
The neutron is a subatomic particle, symbol or , which has a neutral (not positive or negative) charge, and a mass slightly greater than that of a proton. Protons and neutrons constitute the nuclei of atoms. Since protons and neutrons behave similarly within the nucleus, and each has a mass of approximately one atomic mass unit, they are both referred to as nucleons. Their properties and interactions are described by nuclear physics. Protons and neutrons are not elementary particles; each is composed of three quarks. The chemical properties of an atom are mostly determined by the configuration of electrons that orbit the atom's heavy nucleus. The electron configuration is determined by the charge of the nucleus, which is determined by the number of protons, or atomic number. The number of neutrons is the neutron number. Neutrons do not affect the electron configuration, but the sum of atomic and neutron numbers is the mass of the nucleus. Atoms of a chemical element t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Proton
A proton is a stable subatomic particle, symbol , H+, or 1H+ with a positive electric charge of +1 ''e'' elementary charge. Its mass is slightly less than that of a neutron and 1,836 times the mass of an electron (the proton–electron mass ratio). Protons and neutrons, each with masses of approximately one atomic mass unit, are jointly referred to as "nucleons" (particles present in atomic nuclei). One or more protons are present in the nucleus of every atom. They provide the attractive electrostatic central force which binds the atomic electrons. The number of protons in the nucleus is the defining property of an element, and is referred to as the atomic number (represented by the symbol ''Z''). Since each element has a unique number of protons, each element has its own unique atomic number, which determines the number of atomic electrons and consequently the chemical characteristics of the element. The word ''proton'' is Greek for "first", and this name was given to the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
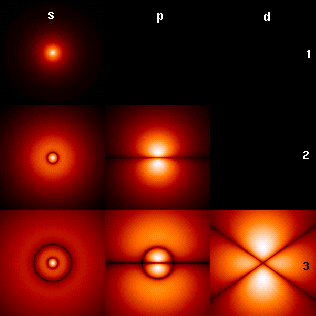
Azimuthal Quantum Number
The azimuthal quantum number is a quantum number for an atomic orbital that determines its orbital angular momentum and describes the shape of the orbital. The azimuthal quantum number is the second of a set of quantum numbers that describe the unique quantum state of an electron (the others being the principal quantum number, the magnetic quantum number, and the spin quantum number). It is also known as the orbital angular momentum quantum number, orbital quantum number or second quantum number, and is symbolized as ℓ (pronounced ''ell''). Derivation Connected with the energy states of the atom's electrons are four quantum numbers: ''n'', ''ℓ'', ''m''''ℓ'', and ''m''''s''. These specify the complete, unique quantum state of a single electron in an atom, and make up its wavefunction or ''orbital''. When solving to obtain the wave function, the Schrödinger equation reduces to three equations that lead to the first three quantum numbers. Therefore, the equations for t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Woods–Saxon Potential
The Woods–Saxon potential is a mean field potential for the nucleons (protons and neutrons) inside the atomic nucleus, which is used to describe approximately the forces applied on each nucleon, in the nuclear shell model for the structure of the nucleus. The potential is named after Roger D. Woods and David S. Saxon. The form of the potential, in terms of the distance ''r'' from the center of nucleus, is: V(r) = -\frac where ''V''0 (having dimension of energy) represents the potential well depth, ''a'' is a length representing the "surface thickness" of the nucleus, and R = r_0 A^ is the nuclear radius where and ''A'' is the mass number. Typical values for the parameters are: , . For large atomic number ''A'' this potential is similar to a potential well. It has the following desired properties * It is monotonically increasing with distance, i.e. attracting. * For large ''A'', it is approximately flat in the center. * Nucleons near the surface of the nucleus (i.e. having ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Spin–orbit Interaction
In quantum physics, the spin–orbit interaction (also called spin–orbit effect or spin–orbit coupling) is a relativistic interaction of a particle's spin with its motion inside a potential. A key example of this phenomenon is the spin–orbit interaction leading to shifts in an electron's atomic energy levels, due to electromagnetic interaction between the electron's magnetic dipole, its orbital motion, and the electrostatic field of the positively charged nucleus. This phenomenon is detectable as a splitting of spectral lines, which can be thought of as a Zeeman effect product of two relativistic effects: the apparent magnetic field seen from the electron perspective and the magnetic moment of the electron associated with its intrinsic spin. A similar effect, due to the relationship between angular momentum and the strong nuclear force, occurs for protons and neutrons moving inside the nucleus, leading to a shift in their energy levels in the nucleus shell model. In the fi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
3D Isotropic Harmonic Oscillator
3-D, 3D, or 3d may refer to: Science, technology, and mathematics Relating to three-dimensionality * Three-dimensional space ** 3D computer graphics, computer graphics that use a three-dimensional representation of geometric data ** 3D film, a motion picture that gives the illusion of three-dimensional perception ** 3D modeling, developing a representation of any three-dimensional surface or object ** 3D printing, making a three-dimensional solid object of a shape from a digital model ** 3D display, a type of information display that conveys depth to the viewer ** 3D television, television that conveys depth perception to the viewer ** Stereoscopy, any technique capable of recording three-dimensional visual information or creating the illusion of depth in an image Other uses in science and technology or commercial products * 3D projection * 3D rendering * 3D scanning, making a digital representation of three-dimensional objects * 3D video game (other) * 3-D Secure, a s ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Shell Gap
Shell may refer to: Architecture and design * Shell (structure), a thin structure ** Concrete shell, a thin shell of concrete, usually with no interior columns or exterior buttresses ** Thin-shell structure Science Biology * Seashell, a hard outer layer of a marine animal, found on beaches * Eggshell * Nutshell * Exoskeleton, an external covering of some animals ** Mollusc shell *** Bivalve shell *** Gastropod shell ** Shell, of a brachiopod ** Turtle shell Physics and chemistry * Electron shell or a principal energy level of electrons outside an atom's nucleus * Nuclear shell model, a principal energy level of nucleons within an atom's nucleus * On shell and off shell, quantum field theory concepts depending on whether classical equations of motion are obeyed Mathematics * Spherical shell Organisations * Shell plc, a British multinational oil and gas company ** Shell USA ** Shell Australia ** Shell Canada ** Shell Nigeria * Shell corporation, a type of compan ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Quantum Harmonic Oscillator
量子調和振動子 は、 古典調和振動子 の 量子力学 類似物です。任意の滑らかな ポテンシャル は通常、安定した 平衡点 の近くで 調和ポテンシャル として近似できるため、最も量子力学における重要なモデル系。さらに、これは正確な 解析解法が知られている数少ない量子力学系の1つである。 author=Griffiths, David J. , title=量子力学入門 , エディション=2nd , 出版社=プレンティス・ホール , 年=2004 , isbn=978-0-13-805326-0 , author-link=David Griffiths (物理学者) , URL アクセス = 登録 , url=https://archive.org/details/introductiontoel00grif_0 One-dimensional harmonic oscillator Hamiltonian and energy eigenstates 粒子の ハミルトニアン は次のとおりです。 \hat H = \frac + \frac k ^2 = \frac + \frac m \omega^2 ^2 \, , ここで、 は粒子の質量、 は力定数、\omega = \sqrt は ��動子の [角周波数 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Square Well
In quantum mechanics, the particle in a box model (also known as the infinite potential well or the infinite square well) describes a particle free to move in a small space surrounded by impenetrable barriers. The model is mainly used as a hypothetical example to illustrate the differences between classical and quantum systems. In classical systems, for example, a particle trapped inside a large box can move at any speed within the box and it is no more likely to be found at one position than another. However, when the well becomes very narrow (on the scale of a few nanometers), quantum effects become important. The particle may only occupy certain positive energy levels. Likewise, it can never have zero energy, meaning that the particle can never "sit still". Additionally, it is more likely to be found at certain positions than at others, depending on its energy level. The particle may never be detected at certain positions, known as spatial nodes. The particle in a box ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |