|
Neonium
Neon compounds are chemical compounds containing the chemical element, element neon (Ne) with other molecules or elements from the periodic table. Compounds of the noble gas neon were believed not to exist, but there are now known to be molecular ions containing neon, as well as temporary excited neon-containing molecules called excimers. Several neutral neon molecules have also been predicted to be stable, but are yet to be discovered in nature. Neon has been shown to crystallize with other substances and form clathrates or Van der Waals molecule, Van der Waals solids. Neon has a high first ionization potential of 21.564 eV, which is only exceeded by that of helium (24.587 eV), requiring too much energy to make stable ionic compounds. Neon's polarisability of 0.395 Å3 is the second lowest of any element (only helium's is more extreme). Low polarisability means there will be little tendency to link to other atoms. Neon has a Lewis basicity or proton affinity of 2.06 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chemical Compound
A chemical compound is a chemical substance composed of many identical molecules (or molecular entities) containing atoms from more than one chemical element held together by chemical bonds. A molecule consisting of atoms of only one element is therefore not a compound. A compound can be transformed into a different substance by a chemical reaction, which may involve interactions with other substances. In this process, bonds between atoms may be broken and/or new bonds formed. There are four major types of compounds, distinguished by how the constituent atoms are bonded together. Molecular compounds are held together by covalent bonds; ionic compounds are held together by ionic bonds; intermetallic compounds are held together by metallic bonds; coordination complexes are held together by coordinate covalent bonds. Non-stoichiometric compounds form a disputed marginal case. A chemical formula specifies the number of atoms of each element in a compound molecule, usi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dodecahedrane
Dodecahedrane is a chemical compound, a hydrocarbon with formula , whose carbon atoms are arranged as the vertices (corners) of a regular dodecahedron. Each carbon is bound to three neighbouring carbon atoms and to a hydrogen atom. This compound is one of the three possible Platonic hydrocarbons, the other two being cubane and tetrahedrane. Dodecahedrane does not occur in nature and has no significant uses. It was synthesized by Leo Paquette in 1982, primarily for the "aesthetically pleasing symmetry of the dodecahedral framework". For many years, dodecahedrane was the simplest real carbon-based molecule with full icosahedral symmetry. Buckminsterfullerene (), discovered in 1985, also has the same symmetry, but has three times as many carbons and 50% more atoms overall. The synthesis of the C20 fullerene in 2000, from brominated dodecahedrane, may have demoted to second place. Structure The angle between the C-C bonds in each carbon atom is 108°, which is the angle be ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Carbonaceous Chondrite
Carbonaceous chondrites or C chondrites are a class of chondritic meteorites comprising at least 8 known groups and many ungrouped meteorites. They include some of the most primitive known meteorites. The C chondrites represent only a small proportion (4.6%) of meteorite falls. Some famous carbonaceous chondrites are: Allende, Murchison, Orgueil, Ivuna, Murray, Tagish Lake, Sutter's Mill and Winchcombe. General description C chondrites contain a high proportion of carbon (up to 3%), which is in the form of graphite, carbonates and organic compounds, including amino acids. In addition, they contain water and minerals that have been modified by the influence of water. The carbonaceous chondrites were not exposed to higher temperatures, so that they are hardly changed by thermal processes. Some carbonaceous chondrites, such as the Allende meteorite, contain calcium-aluminum-rich inclusions (CAIs). These are compounds that emerged early from the primeval solar nebula, c ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Neon-22
Neon (10Ne) possesses three stable isotopes: , , and . In addition, 17 radioactive isotopes have been discovered, ranging from to , all short-lived. The longest-lived is with a half-life of . All others are under a minute, most under a second. The least stable is with a half-life of (). See isotopes of carbon for notes about the measurement. Light radioactive neon isotopes usually decay to fluorine or oxygen, while heavier ones decay to sodium. List of isotopes , - , , style="text-align:right" , 10 , style="text-align:right" , 5 , , [] , proton emission, 2p , , (3/2−) , , , - , , style="text-align:right" , 10 , style="text-align:right" , 6 , , > [ , - , , style="text-align:right" , 10 , style="text-align:right" , 11 , , colspan=3 align=center, Stable , 3/2+ , , ref name="Isotopic Composition of Elements" /> , - , , style="text-align:right" , 10 , style="text-align:right" , 12 , , colspan=3 align=center, Stable , 0+ , , ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
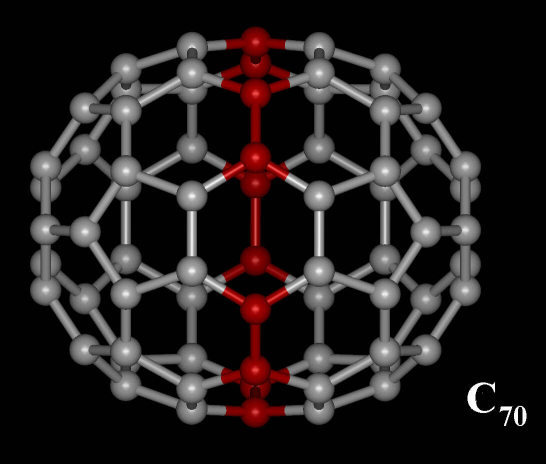
C70 Fullerene
C70 fullerene is the fullerene molecule consisting of 70 carbon atoms. It is a cage-like fused-ring structure which resembles a rugby ball, made of 25 hexagons and 12 pentagons, with a carbon atom at the vertices of each polygon and a bond along each polygon edge. A related fullerene molecule, named buckminsterfullerene (C60 fullerene), consists of 60 carbon atoms. It was first intentionally prepared in 1985 by Harold Kroto, James R. Heath, Sean O'Brien, Robert Curl and Richard Smalley at Rice University. Kroto, Curl and Smalley were awarded the 1996 Nobel Prize in Chemistry for their roles in the discovery of cage-like fullerenes. The name is a homage to Buckminster Fuller, whose geodesic domes these molecules resemble. History Theoretical predictions of buckyball molecules appeared in the late 1960s to early 1970s,Katz, 363 but they went largely unnoticed. In the early 1970s, the chemistry of unsaturated carbon configurations was studied by a group at the University of ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Buckminsterfullerene
Buckminsterfullerene is a type of fullerene with the formula C60. It has a cage-like fused-ring structure (truncated icosahedron) made of twenty hexagons and twelve pentagons, and resembles a soccer ball. Each of its 60 carbon atoms is bonded to its three neighbors. Buckminsterfullerene is a black solid that dissolves in hydrocarbon solvents to produce a violet solution. The compound was discovered in 1985 and has received intense study, although few real world applications have been found. Occurrence Buckminsterfullerene is the most common naturally occurring fullerene. Small quantities of it can be found in soot. It also exists in space. Neutral C60 has been observed in planetary nebulae and several types of star. The ionised form, C60+, has been identified in the interstellar medium, where it is the cause of several absorption features known as diffuse interstellar bands in the near-infrared. History Theoretical predictions of buckyball molecules appeared i ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Fullerenes
A fullerene is an allotrope of carbon whose molecule consists of carbon atoms connected by single and double bonds so as to form a closed or partially closed mesh, with fused rings of five to seven atoms. The molecule may be a hollow sphere, ellipsoid, tube, or many other shapes and sizes. Graphene (isolated atomic layers of graphite), which is a flat mesh of regular hexagonal rings, can be seen as an extreme member of the family. Fullerenes with a closed mesh topology are informally denoted by their empirical formula C''n'', often written C''n'', where ''n'' is the number of carbon atoms. However, for some values of ''n'' there may be more than one isomer. The family is named after buckminsterfullerene (C60), the most famous member, which in turn is named after Buckminster Fuller. The closed fullerenes, especially C60, are also informally called buckyballs for their resemblance to the standard ball of association football ("soccer"). Nested closed fullerenes have been na ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Helium Compounds
Helium is the smallest and the lightest noble gas and one of the most unreactive elements, so it was commonly considered that helium compounds cannot exist at all, or at least under normal conditions. Helium's first ionization energy of 24.57 eV is the highest of any element. Helium has a complete shell of electrons, and in this form the atom does not readily accept any extra electrons nor join with anything to make covalent compounds. The electron affinity is 0.080 eV, which is very close to zero. The helium atom is small with the radius of the outer electron shell at 0.29 Å. Helium is a very hard atom with a Pearson hardness of 12.3 eV. It has the lowest polarizability of any kind of atom, however, very weak van der Waals forces exist between helium and other atoms. This force may exceed repulsive forces, so at extremely low temperatures helium may form van der Waals molecules. Helium has the lowest boiling point (4.2 K) of any known substance. Repulsive fo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrogen Clathrate
A hydrogen clathrate is a clathrate containing hydrogen in a water lattice. It is not possible to store commercial quantities of hydrogen by this method hydrogen in a hydrogen economy. A recent review that accounts the state-of-the-art and future prospects and challenges of hydrogen storage as clathrate hydrates is reported by Veluswamy et al. (2014). Another unusual characteristic is that multiple hydrogen molecules can occur at each cage site in the ice, one of only a very few guest molecule that forms clathrates with this property. The maximum ratio of hydrogen to water is 6 H2 to 17 H2O. It can be formed at 250K in a diamond anvil at a pressure of 300MPa (3000 Bars). It takes about 30 minutes to form, so this method is impractical for rapid manufacture. The percent of weight of hydrogen is 3.77%. The cage compartments are hexakaidecahedral and hold from two to four molecules of hydrogen. At temperatures above 160K the molecules rotate around inside the cage. Below 120K the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ice II
Ice II is a rhombohedral crystalline form of ice with a highly ordered structure. It is formed from ice Ih by compressing it at a temperature of 198 K at 300 MPa or by decompressing ice V. When heated it undergoes transformation to ice III. Ordinary water ice is known as ice Ih, (in the Bridgman nomenclature). Different types of ice, from ice II to ice XIX, have been created in the laboratory at different temperatures and pressures. It is thought that the cores of icy moons like Jupiter's Ganymede may be made of ice II. History The properties of ice II were first described and recorded by Gustav Heinrich Johann Apollon Tammann in 1900 during his experiments with ice under high pressure and low temperatures. Having produced ice III, Tammann then tried condensing the ice at a temperature between under of pressure. Tammann noted that in this state ice II was denser than he had observed ice III to be. He also found that both types of ice can be kept at normal atmospheric ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |