|
Ionisation Chamber
The ionization chamber is the simplest type of gas-filled radiation detector, and is widely used for the detection and measurement of certain types of ionizing radiation, including X-rays, gamma rays, and beta particles. Conventionally, the term "ionization chamber" refers exclusively to those detectors which collect all the charges created by ''direct ionization'' within the gas through the application of an electric field. It only uses the discrete charges created by each interaction between the incident radiation and the gas. Gaseous ionization detectors include ionization chambers and devices that use gas multiplication, namely the proportional counter and the Geiger counter. Ion chambers have a good uniform response to radiation over a wide range of energies and are the preferred means of measuring high levels of gamma radiation. They are widely used in the nuclear power industry, research labs, radiography, radiobiology, and environmental monitoring. Principle of operation ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ionizing Radiation
Ionizing radiation (or ionising radiation), including nuclear radiation, consists of subatomic particles or electromagnetic waves that have sufficient energy to ionize atoms or molecules by detaching electrons from them. Some particles can travel up to 99% of the speed of light, and the electromagnetic waves are on the high-energy portion of the electromagnetic spectrum. Gamma rays, X-rays, and the higher energy ultraviolet part of the electromagnetic spectrum are ionizing radiation, whereas the lower energy ultraviolet, visible light, nearly all types of laser light, infrared, microwaves, and radio waves are non-ionizing radiation. The boundary between ionizing and non-ionizing radiation in the ultraviolet area is not sharply defined, as different molecules and atoms ionize at different energies. The energy of ionizing radiation starts between 10 electronvolts (eV) and 33 eV. Typical ionizing subatomic particles include alpha particles, beta particles, and neutrons. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electrometer
An electrometer is an electrical instrument for measuring electric charge or electrical potential difference. There are many different types, ranging from historical handmade mechanical instruments to high-precision electronic devices. Modern electrometers based on vacuum tube or solid-state technology can be used to make voltage and charge measurements with very low leakage currents, down to 1 femtoampere. A simpler but related instrument, the electroscope, works on similar principles but only indicates the relative magnitudes of voltages or charges. Historical electrometers Gold-leaf electroscope The gold-leaf electroscope was one of the instruments used to indicate electric charge. It is still used for science demonstrations but has been superseded in most applications by electronic measuring instruments. The instrument consists of two thin leaves of gold foil suspended from an electrode. When the electrode is charged by induction or by contact, the leaves acquire simi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ionisation Chamber Made By Pierre Curie, C 1895-1900
Ionization, or Ionisation is the process by which an atom or a molecule acquires a negative or positive charge by gaining or losing electrons, often in conjunction with other chemical changes. The resulting electrically charged atom or molecule is called an ion. Ionization can result from the loss of an electron after collisions with subatomic particles, collisions with other atoms, molecules and ions, or through the interaction with electromagnetic radiation. Heterolytic bond cleavage and heterolytic substitution reactions can result in the formation of ion pairs. Ionization can occur through radioactive decay by the internal conversion process, in which an excited nucleus transfers its energy to one of the inner-shell electrons causing it to be ejected. Uses Everyday examples of gas ionization are such as within a fluorescent lamp or other electrical discharge lamps. It is also used in radiation detectors such as the Geiger-Müller counter or the ionization chamber. The ionizati ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
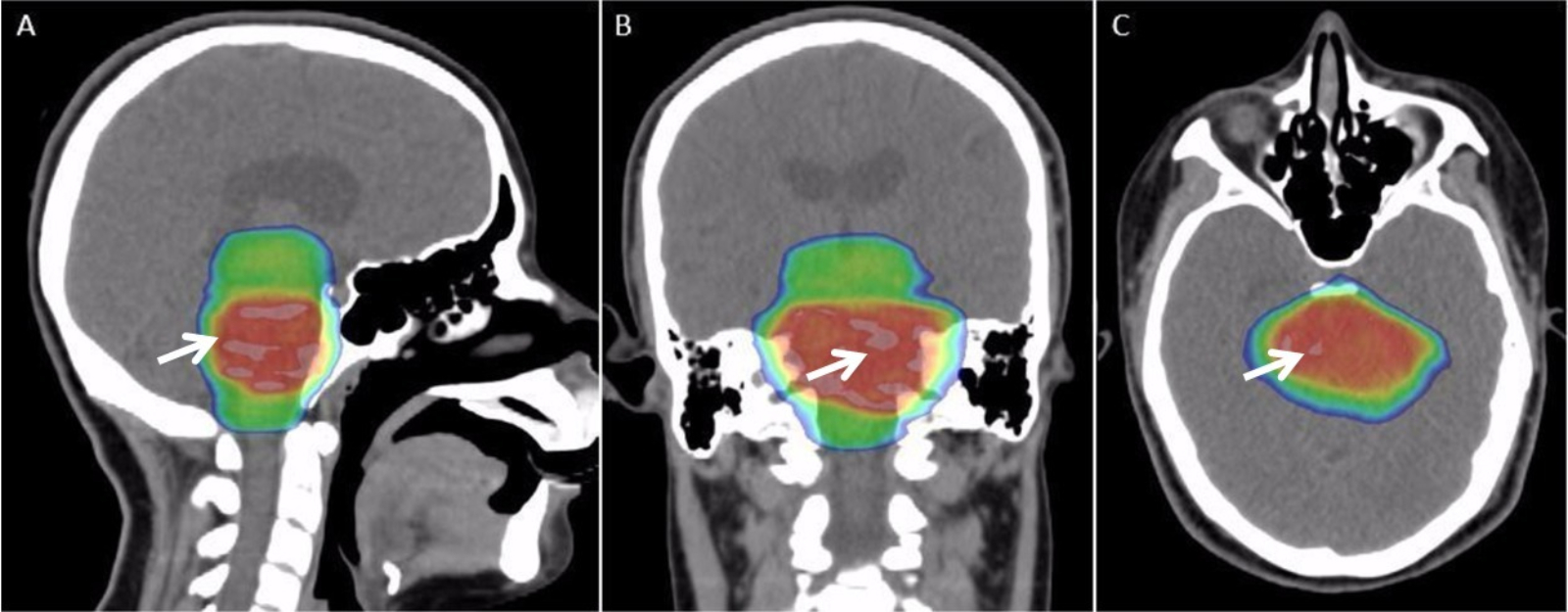
Radiotherapy
Radiation therapy or radiotherapy, often abbreviated RT, RTx, or XRT, is a therapy using ionizing radiation, generally provided as part of cancer treatment to control or kill malignant cells and normally delivered by a linear accelerator. Radiation therapy may be curative in a number of types of cancer if they are localized to one area of the body. It may also be used as part of adjuvant therapy, to prevent tumor recurrence after surgery to remove a primary malignant tumor (for example, early stages of breast cancer). Radiation therapy is synergistic with chemotherapy, and has been used before, during, and after chemotherapy in susceptible cancers. The subspecialty of oncology concerned with radiotherapy is called radiation oncology. A physician who practices in this subspecialty is a radiation oncologist. Radiation therapy is commonly applied to the cancerous tumor because of its ability to control cell growth. Ionizing radiation works by damaging the DNA of cancerous tissue l ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Linear Accelerator
A linear particle accelerator (often shortened to linac) is a type of particle accelerator that accelerates charged subatomic particles or ions to a high speed by subjecting them to a series of oscillating electric potentials along a linear beamline. The principles for such machines were proposed by Gustav Ising in 1924, while the first machine that worked was constructed by Rolf Widerøe in 1928 at the RWTH Aachen University. Linacs have many applications: they generate X-rays and high energy electrons for medicinal purposes in radiation therapy, serve as particle injectors for higher-energy accelerators, and are used directly to achieve the highest kinetic energy for light particles (electrons and positrons) for particle physics. The design of a linac depends on the type of particle that is being accelerated: electrons, protons or ions. Linacs range in size from a cathode ray tube (which is a type of linac) to the linac at the SLAC National Accelerator Laboratory in Menlo Park ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Radiation Therapy
Radiation therapy or radiotherapy, often abbreviated RT, RTx, or XRT, is a therapy using ionizing radiation, generally provided as part of cancer treatment to control or kill malignant cells and normally delivered by a linear accelerator. Radiation therapy may be curative in a number of types of cancer if they are localized to one area of the body. It may also be used as part of adjuvant therapy, to prevent tumor recurrence after surgery to remove a primary malignant tumor (for example, early stages of breast cancer). Radiation therapy is synergistic with chemotherapy, and has been used before, during, and after chemotherapy in susceptible cancers. The subspecialty of oncology concerned with radiotherapy is called radiation oncology. A physician who practices in this subspecialty is a radiation oncologist. Radiation therapy is commonly applied to the cancerous tumor because of its ability to control cell growth. Ionizing radiation works by damaging the DNA of cancerous tissue ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electronegative
Electronegativity, symbolized as , is the tendency for an atom of a given chemical element to attract shared electrons (or electron density) when forming a chemical bond. An atom's electronegativity is affected by both its atomic number and the distance at which its valence electrons reside from the charged nucleus. The higher the associated electronegativity, the more an atom or a substituent group attracts electrons. Electronegativity serves as a simple way to quantitatively estimate the bond energy, and the sign and magnitude of a bond's chemical polarity, which characterizes a bond along the continuous scale from covalent to ionic bonding. The loosely defined term electropositivity is the opposite of electronegativity: it characterizes an element's tendency to donate valence electrons. On the most basic level, electronegativity is determined by factors like the nuclear charge (the more protons an atom has, the more "pull" it will have on electrons) and the number and location ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Noble Gas
The noble gases (historically also the inert gases; sometimes referred to as aerogens) make up a class of chemical elements with similar properties; under standard conditions, they are all odorless, colorless, monatomic gases with very low chemical reactivity. The six naturally occurring noble gases are helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe), and the radioactive radon (Rn). Oganesson (Og) is a synthetically produced highly radioactive element. Although IUPAC has used the term "noble gas" interchangeably with "group 18" and thus included oganesson, it may not be significantly chemically noble and is predicted to break the trend and be reactive due to relativistic effects. Because of the extremely short 0.7 ms half-life of its only known isotope, its chemistry has not yet been investigated. For the first six periods of the periodic table, the noble gases are exactly the members of group 18. Noble gases are typically highly unreactive except when u ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Desiccant
A desiccant is a hygroscopic substance that is used to induce or sustain a state of dryness (desiccation) in its vicinity; it is the opposite of a humectant. Commonly encountered pre-packaged desiccants are solids that absorb water. Desiccants for specialized purposes may be in forms other than solid, and may work through other principles, such as chemical bonding of water molecules. They are commonly encountered in foods to retain crispness. Industrially, desiccants are widely used to control the level of water in gas streams. Types of desiccants Although some desiccants are chemically inert, others are extremely reactive and require specialized handling techniques. The most common desiccant is silica gel, an otherwise inert, nontoxic, water-insoluble white solid. Tens of thousands of tons are produced annually for this purpose. Other common desiccants include activated charcoal, calcium sulfate, calcium chloride, and molecular sieves (typically, zeolites). Desiccants ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Smoke Detector
A smoke detector is a device that senses smoke, typically as an indicator of fire. Smoke detectors are usually housed in plastic enclosures, typically shaped like a disk about in diameter and thick, but shape and size vary. Smoke can be detected either optically (photoelectric) or by physical process (ionization). Detectors may use one or both sensing methods. Sensitive alarms can be used to detect and deter smoking in banned areas. Smoke detectors in large commercial and industrial buildings are usually connected to a central fire alarm system. Household smoke detectors, also known as ''smoke alarms'', generally issue an audible or visual alarm from the detector itself or several detectors if there are multiple devices interlinked. Household smoke detectors range from individual battery-powered units to several interlinked units with battery backup. With interlinked units, if any unit detects smoke, alarms will trigger at all of the units. This happens even if household power ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Geiger–Müller Tube
The Geiger–Müller tube or G–M tube is the sensing element of the Geiger counter instrument used for the detection of ionizing radiation. It is named after Hans Geiger, who invented the principle in 1908, and Walther Müller, who collaborated with Geiger in developing the technique further in 1928 to produce a practical tube that could detect a number of different radiation types. It is a gaseous ionization detector and uses the Townsend avalanche phenomenon to produce an easily detectable electronic pulse from as little as a single ionizing event due to a radiation particle. It is used for the detection of gamma radiation, X-rays, and alpha and beta particles. It can also be adapted to detect neutrons. The tube operates in the "Geiger" region of ion pair generation. This is shown on the accompanying plot for gaseous detectors showing ion current against applied voltage. While it is a robust and inexpensive detector, the G–M is unable to measure high radiation rates efficient ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |