|
Insulin Receptor Substrate
Insulin receptor substrate (IRS) is an important ligand in the insulin response of human cells. IRS-1, for example, is an IRS protein that contains a phosphotyrosine binding-domain ( PTB-domain). In addition, the insulin receptor contains a NPXY motif Motif may refer to: General concepts * Motif (chess composition), an element of a move in the consideration of its purpose * Motif (folkloristics), a recurring element that creates recognizable patterns in folklore and folk-art traditions * Moti .... The PTB-domain binds the NPXY sequence. Thus, the insulin receptor binds IRS. Genes * (see also Insulin receptor substrate 1) * (see also Insulin receptor substrate 2) * - a pseudogene * References * {{cite journal , vauthors=van der Geer P, Wiley S, Pawson T , title=Re-engineering the target specificity of the insulin receptor by modification of a PTB domain binding site , journal=Oncogene , volume=18 , issue=20 , pages=3071–5 , year=1999 , pmid=10340378 , doi=10.1038/sj.onc ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ligand (biochemistry)
In biochemistry and pharmacology, a ligand is a substance that forms a complex with a biomolecule to serve a biological purpose. The etymology stems from ''ligare'', which means 'to bind'. In protein-ligand binding, the ligand is usually a molecule which produces a signal by binding to a site on a target protein. The binding typically results in a change of conformational isomerism (conformation) of the target protein. In DNA-ligand binding studies, the ligand can be a small molecule, ion, or protein which binds to the DNA double helix. The relationship between ligand and binding partner is a function of charge, hydrophobicity, and molecular structure. Binding occurs by intermolecular forces, such as ionic bonds, hydrogen bonds and Van der Waals forces. The association or docking is actually reversible through dissociation. Measurably irreversible covalent bonding between a ligand and target molecule is atypical in biological systems. In contrast to the definition ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
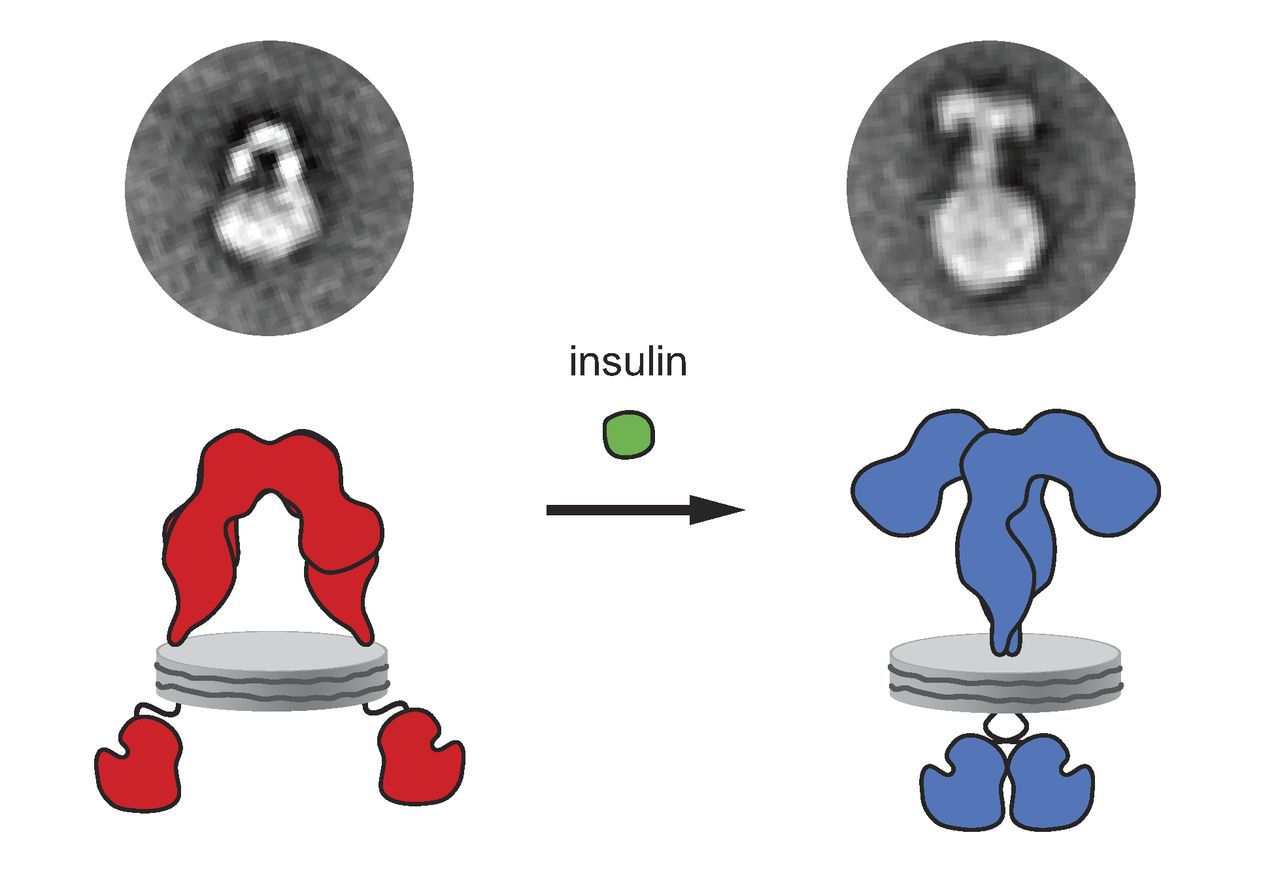
Insulin
Insulin (, from Latin ''insula'', 'island') is a peptide hormone produced by beta cells of the pancreatic islets encoded in humans by the ''INS'' gene. It is considered to be the main anabolic hormone of the body. It regulates the metabolism of carbohydrates, fats and protein by promoting the absorption of glucose from the blood into liver, fat and skeletal muscle cells. In these tissues the absorbed glucose is converted into either glycogen via glycogenesis or fats ( triglycerides) via lipogenesis, or, in the case of the liver, into both. Glucose production and secretion by the liver is strongly inhibited by high concentrations of insulin in the blood. Circulating insulin also affects the synthesis of proteins in a wide variety of tissues. It is therefore an anabolic hormone, promoting the conversion of small molecules in the blood into large molecules inside the cells. Low insulin levels in the blood have the opposite effect by promoting widespread catabolism, espe ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
IRS-1
Insulin receptor substrate 1 (IRS-1) is a signaling adapter protein that in humans is encoded by the ''IRS-1'' gene. It is a 131 kDa protein with amino acid sequence of 1242 residues. It contains a single pleckstrin homology (PH) domain at the N-terminus and a PTB domain ca. 40 residues downstream of this, followed by a poorly conserved C-terminus tail. Together with IRS2, IRS3 (pseudogene) and IRS4, it is homologous to the ''Drosophila'' protein ''chico'', whose disruption extends the median lifespan of flies up to 48%. Similarly, Irs1 mutant mice experience moderate life extension and delayed age-related pathologies. Function Insulin receptor substrate 1 plays a key role in transmitting signals from the insulin and insulin-like growth factor-1 (IGF-1) receptors to intracellular pathways PI3K / Akt and Erk MAP kinase pathways. Tyrosine phosphorylation of IRS-1 by insulin receptor (IR) introduces multiple binding sites for proteins bearing SH2 homology domain, such as PI3K, Grb- ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phosphotyrosine
-Tyrosine or tyrosine (symbol Tyr or Y) or 4-hydroxyphenylalanine is one of the 20 standard amino acids that are used by cells to synthesize proteins. It is a non-essential amino acid with a polar side group. The word "tyrosine" is from the Greek ''tyrós'', meaning ''cheese'', as it was first discovered in 1846 by German chemist Justus von Liebig in the protein casein from cheese. It is called tyrosyl when referred to as a functional group or side chain. While tyrosine is generally classified as a hydrophobic amino acid, it is more hydrophilic than phenylalanine. It is encoded by the codons UAC and UAU in messenger RNA. Functions Aside from being a proteinogenic amino acid, tyrosine has a special role by virtue of the phenol functionality. It occurs in proteins that are part of signal transduction processes and functions as a receiver of phosphate groups that are transferred by way of protein kinases. Phosphorylation of the hydroxyl group can change the activity of the ta ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
PTB Domain
In molecular biology, Phosphotyrosine-binding domains are protein domains which bind to phosphotyrosine. The phosphotyrosine-binding domain (PTB, also phosphotyrosine-interaction or PI domain) in the protein tensin tends to be found at the C-terminus. Tensin is a multi-domain protein that binds to actin filaments and functions as a focal-adhesion molecule (focal adhesions are regions of plasma membrane through which cells attach to the extracellular matrix). Human tensin has actin-binding sites, an SH2 () domain and a region similar to the tumour suppressor PTEN. The PTB domain interacts with the cytoplasmic tails of beta integrin by binding to an NPXY motif. The phosphotyrosine-binding domain of insulin receptor substrate-1 is not related to the phosphotyrosine-binding domain of tensin. Insulin receptor substrate-1 proteins contain both a pleckstrin homology domain and a phosphotyrosine binding (PTB) domain. The PTB domains facilitate interaction with the activated tyr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Insulin Receptor
The insulin receptor (IR) is a transmembrane receptor that is activated by insulin, IGF-I, IGF-II and belongs to the large class of receptor tyrosine kinase. Metabolically, the insulin receptor plays a key role in the regulation of glucose homeostasis, a functional process that under degenerate conditions may result in a range of clinical manifestations including diabetes and cancer. Insulin signalling controls access to blood glucose in body cells. When insulin falls, especially in those with high insulin sensitivity, body cells begin only to have access to lipids that do not require transport across the membrane. So, in this way, insulin is the key regulator of fat metabolism as well. Biochemically, the insulin receptor is encoded by a single gene , from which alternate splicing during transcription results in either IR-A or IR-B isoforms. Downstream post-translational events of either isoform result in the formation of a proteolytically cleaved α and β subunit, which upon com ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Protein Motif
In a chain-like biological molecule, such as a protein or nucleic acid, a structural motif is a common three-dimensional structure that appears in a variety of different, evolutionarily unrelated molecules. A structural motif does not have to be associated with a sequence motif; it can be represented by different and completely unrelated sequences in different proteins or RNA. In nucleic acids Depending upon the sequence and other conditions, nucleic acids can form a variety of structural motifs which is thought to have biological significance. ;Stem-loop: Stem-loop intramolecular base pairing is a pattern that can occur in single-stranded DNA or, more commonly, in RNA. The structure is also known as a hairpin or hairpin loop. It occurs when two regions of the same strand, usually complementary in nucleotide sequence when read in opposite directions, base-pair to form a double helix that ends in an unpaired loop. The resulting structure is a key building block of many ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Insulin Receptor Substrate 1
Insulin receptor substrate 1 (IRS-1) is a signaling adapter protein that in humans is encoded by the ''IRS-1'' gene. It is a 131 kDa protein with amino acid sequence of 1242 residues. It contains a single pleckstrin homology (PH) domain at the N-terminus and a PTB domain ca. 40 residues downstream of this, followed by a poorly conserved C-terminus tail. Together with IRS2, IRS3 (pseudogene) and IRS4, it is homologous to the ''Drosophila'' protein ''chico'', whose disruption extends the median lifespan of flies up to 48%. Similarly, Irs1 mutant mice experience moderate life extension and delayed age-related pathologies. Function Insulin receptor substrate 1 plays a key role in transmitting signals from the insulin and insulin-like growth factor-1 (IGF-1) receptors to intracellular pathways PI3K / Akt and Erk MAP kinase pathways. Tyrosine phosphorylation of IRS-1 by insulin receptor (IR) introduces multiple binding sites for proteins bearing SH2 homology domain, such as PI3K, Grb- ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
IRS2
Insulin receptor substrate 2 is a protein that in humans is encoded by the ''IRS2'' gene. Function This gene encodes the insulin receptor substrate 2, a cytoplasmic signaling molecule that mediates effects of insulin, insulin-like growth factor 1, and other cytokines by acting as a molecular adaptor between diverse receptor tyrosine kinases and downstream effectors. The product of this gene is phosphorylated by the insulin receptor tyrosine kinase upon receptor stimulation, as well as by an interleukin 4 receptor-associated kinase in response to IL4 treatment. Mice lacking IRS2 have a diabetic phenotype as well as a 40% reduction in brain mass. Interactions IRS2 has been shown to interact with: * PLCG1, * SOCS1, and * PIK3R1 Phosphatidylinositol 3-kinase regulatory subunit alpha is an enzyme that in humans is encoded by the ''PIK3R1'' gene. Function Phosphatidylinositol 3-kinase phosphorylates the inositol ring of phosphatidylinositol at the 3-prime position. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Coordination Chemistry
A coordination complex consists of a central atom or ion, which is usually metallic and is called the ''coordination centre'', and a surrounding array of chemical bond, bound molecules or ions, that are in turn known as ''ligands'' or complexing agents. Many metal-containing chemical compound, compounds, especially those that include transition metals (elements like titanium that belong to the Block (periodic table), Periodic Table's d-block), are coordination complexes. Nomenclature and terminology Coordination complexes are so pervasive that their structures and reactions are described in many ways, sometimes confusingly. The atom within a ligand that is bonded to the central metal atom or ion is called the donor atom. In a typical complex, a metal ion is bonded to several donor atoms, which can be the same or different. A Ligand#Polydentate and polyhapto ligand motifs and nomenclature, polydentate (multiple bonded) ligand is a molecule or ion that bonds to the central atom ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |