|
Hydronium Perchlorate
Hydronium perchlorate is the chemical compound with the formula . It is an unusual salt due to it being a solid and stable hydronium salt. Production Hydronium perchlorate is produced by the reaction of anhydrous perchloric acid and water in a 1:1 molar ratio: : This method was found to produce some byproducts and is not a reliable method. A more reliable method was reported by the cyclocondensation of sodium 2,6-diformyl-4-methylphenolate(produced by the reaction of 2,6-diformyl-4-methylphenol and sodium hydroxide in ethanol) and p-phenylenediamine via the sodium template method. It resulted in a yellow solid which was transmetallated with copper(II) perchlorate then refluxed in nitrogen for 1 hour. The resulting solution was filtered and cooled to form the colorless crystals of hydronium perchlorate. It can also be produced by the reaction of anhydrous nitric acid and perchloric acid Perchloric acid is a mineral acid with the formula H Cl O4. Usually found as an aqueous ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydronium Chloride
Hydrochloric acid, also known as muriatic acid, is an aqueous solution of hydrogen chloride. It is a colorless solution with a distinctive pungency, pungent smell. It is classified as a acid strength, strong acid. It is a component of the gastric acid in the digestive systems of most animal species, including humans. Hydrochloric acid is an important laboratory reagent and industrial chemical. History In the early tenth century, the Persian physician and alchemist Abu Bakr al-Razi ( 865–925, Latin: Rhazes) conducted experiments with sal ammoniac (ammonium chloride) and vitriol (hydrated sulfates of various metals), which he distilled together, thus producing the gas hydrogen chloride. In doing so, al-Razi may have stumbled upon a primitive method for producing hydrochloric acid, as perhaps manifested in the following recipe from his ("The Book of Secrets"): However, it appears that in most of his experiments al-Razi disregarded the gaseous products, concentrating instead o ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Perchloric Acid
Perchloric acid is a mineral acid with the formula H Cl O4. Usually found as an aqueous solution, this colorless compound is a stronger acid than sulfuric acid, nitric acid and hydrochloric acid. It is a powerful oxidizer when hot, but aqueous solutions up to approximately 70% by weight at room temperature are generally safe, only showing strong acid features and no oxidizing properties. Perchloric acid is useful for preparing perchlorate salts, especially ammonium perchlorate, an important rocket fuel component. Perchloric acid is dangerously corrosive and readily forms potentially explosive mixtures. History Perchloric acid was first synthesized (together with potassium perchlorate) by Austrian chemist and called "oxygenated chloric acid" in mid-1810s; French pharmacist Georges-Simon Serullas introduced the modern designation along with discovering its solid monohydrate (which he, however, mistook for an anhydride). Production Perchloric acid is produced industrially by two r ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydronium
In chemistry, hydronium (hydroxonium in traditional British English) is the common name for the aqueous cation , the type of oxonium ion produced by protonation of water. It is often viewed as the positive ion present when an Arrhenius acid is dissolved in water, as Arrhenius acid molecules in solution give up a proton (a positive hydrogen ion, ) to the surrounding water molecules (). In fact, acids must be surrounded by more than a single water molecule in order to ionize, yielding aqueous and conjugate base. Three main structures for the aqueous proton have garnered experimental support: The Eigen cation, which is a tetrahydrate, H3O+(H2O)3; the Zundel cation, which is a symmetric dihydrate, H+(H2O)2; and the Stoyanov cation, an expanded Zundel cation, which is a hexahydrate: H+(H2O)2(H2O)4. Spectroscopic evidence from well-defined IR spectra overwhelmingly supports the Stoyanov cation as the predominant form. For this reason, it has been suggested that wherever possible ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Perchloric Acid
Perchloric acid is a mineral acid with the formula H Cl O4. Usually found as an aqueous solution, this colorless compound is a stronger acid than sulfuric acid, nitric acid and hydrochloric acid. It is a powerful oxidizer when hot, but aqueous solutions up to approximately 70% by weight at room temperature are generally safe, only showing strong acid features and no oxidizing properties. Perchloric acid is useful for preparing perchlorate salts, especially ammonium perchlorate, an important rocket fuel component. Perchloric acid is dangerously corrosive and readily forms potentially explosive mixtures. History Perchloric acid was first synthesized (together with potassium perchlorate) by Austrian chemist and called "oxygenated chloric acid" in mid-1810s; French pharmacist Georges-Simon Serullas introduced the modern designation along with discovering its solid monohydrate (which he, however, mistook for an anhydride). Production Perchloric acid is produced industrially by two r ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
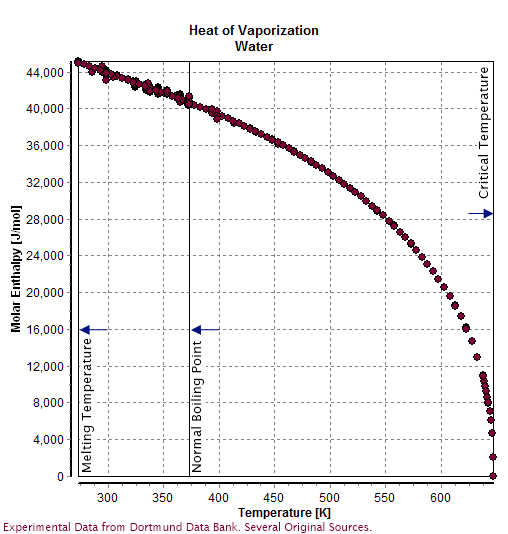
Properties Of Water
Water () is a polar inorganic compound that is at room temperature a tasteless and odorless liquid, which is nearly colorless apart from an inherent hint of blue. It is by far the most studied chemical compound and is described as the "universal solvent" and the "solvent of life". It is the most abundant substance on the surface of Earth and the only common substance to exist as a solid, liquid, and gas on Earth's surface. It is also the third most abundant molecule in the universe (behind molecular hydrogen and carbon monoxide). Water molecules form hydrogen bonds with each other and are strongly polar. This polarity allows it to dissociate ions in salts and bond to other polar substances such as alcohols and acids, thus dissolving them. Its hydrogen bonding causes its many unique properties, such as having a solid form less dense than its liquid form, a relatively high boiling point of 100 °C for its molar mass, and a high heat capacity. Water is amphoteric, meani ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sodium Hydroxide
Sodium hydroxide, also known as lye and caustic soda, is an inorganic compound with the formula NaOH. It is a white solid ionic compound consisting of sodium cations and hydroxide anions . Sodium hydroxide is a highly caustic base and alkali that decomposes proteins at ordinary ambient temperatures and may cause severe chemical burns. It is highly soluble in water, and readily absorbs moisture and carbon dioxide from the air. It forms a series of hydrates . The monohydrate crystallizes from water solutions between 12.3 and 61.8 °C. The commercially available "sodium hydroxide" is often this monohydrate, and published data may refer to it instead of the anhydrous compound. As one of the simplest hydroxides, sodium hydroxide is frequently used alongside neutral water and acidic hydrochloric acid to demonstrate the pH scale to chemistry students. Sodium hydroxide is used in many industries: in the manufacture of pulp and paper, textiles, drinking water, soaps and deterge ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ethanol
Ethanol (abbr. EtOH; also called ethyl alcohol, grain alcohol, drinking alcohol, or simply alcohol) is an organic compound. It is an Alcohol (chemistry), alcohol with the chemical formula . Its formula can be also written as or (an ethyl group linked to a hydroxyl group). Ethanol is a Volatility (chemistry), volatile, Combustibility and flammability, flammable, colorless liquid with a characteristic wine-like odor and pungent taste. It is a psychoactive recreational drug, the active ingredient in alcoholic drinks. Ethanol is naturally produced by the fermentation process of Carbohydrate, sugars by yeasts or via Petrochemistry, petrochemical processes such as ethylene hydration. It has medical applications as an antiseptic and disinfectant. It is used as a chemical solvent and in the Chemical synthesis, synthesis of organic compounds, and as a Alcohol fuel, fuel source. Ethanol also can be dehydrated to make ethylene, an important chemical feedstock. As of 2006, world produ ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
P-phenylenediamine
''p''-Phenylenediamine (PPD) is an organic compound with the formula C6H4(NH2)2. This derivative of aniline is a white solid, but samples can darken due to air oxidation. It is mainly used as a component of engineering polymers and composites like kevlar. It is also an ingredient in hair dyes and is occasionally used as a substitute for henna. Production PPD is produced via three routes. Most commonly, 4-nitrochlorobenzene is treated with ammonia and the resulting 4-nitroaniline is then hydrogenated: :ClC6H4NO2 + 2 NH3 → H2NC6H4NO2 + NH4Cl :H2NC6H4NO2 + 3 H2 → H2NC6H4NH2 + 2 H2O In the DuPont route, aniline is converted to diphenyltriazine, which is then converted by acid-catalysis to 4-aminoazobenzene. Hydrogenation of the latter affords PPD.Robert A. Smiley "Phenylene- and Toluenediamines" in ''Ullmann's Encyclopedia of Industrial Chemistry'', 2002, Wiley-VCH, Weinheim. Uses Precursor to polymers PPD is a precursor to aramid plastics and fibers such as ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Transmetalation
Transmetalation (alt. spelling: transmetallation) is a type of organometallic reaction that involves the transfer of ligands from one metal to another. It has the general form: :M1–R + M2–R′ → M1–R′ + M2–R where R and R′ can be, but are not limited to, an alkyl, aryl, alkynyl, allyl, halogen, or pseudohalogen group. The reaction is usually an irreversible process due to thermodynamic and kinetic reasons. Thermodynamics will favor the reaction based on the electronegativities of the metals and kinetics will favor the reaction if there are empty orbitals on both metals. There are different types of transmetalation including redox-transmetalation and redox-transmetalation/ligand exchange. During transmetalation the metal-carbon bond is activated, leading to the formation of new metal-carbon bonds. Transmetalation is commonly used in catalysis, synthesis of main group complexes, and synthesis of transition metal complexes. Types of transmetalation There are two main types ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Copper(II) Perchlorate
Copper(II) perchlorate is a salt of copper and perchloric acid. It is a hygroscopic crystalline blue solid, most commonly copper(II) perchlorate hexahydrate, Cu(ClO4)2·6H2O. Like any perchlorate A perchlorate is a chemical compound containing the perchlorate ion, . The majority of perchlorates are commercially produced salts. They are mainly used as oxidizers for pyrotechnic devices and to control static electricity in food packaging. Per ..., it is a strong oxidizing agent. References Copper(II) compounds Perchlorates {{inorganic-compound-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |