|
Heating Elements
A heating element converts electrical energy into heat through the process of Joule heating. Electric current through the element encounters resistance, resulting in heating of the element. Unlike the Peltier effect, this process is independent of the direction of current. Heating elements types Metal Resistance wire: Metallic resistance heating elements may be wire or ribbon, straight or coiled. They are used in common heating devices like toasters and hair dryers, furnaces for industrial heating, floor heating, roof heating, pathway heating to melt snow, dryers, etc. The most common classes of materials used include: * Nichrome: Most resistance wire heating elements usually use nichrome 80/20 (80% Nickel, 20% Chromium) wire, ribbon, or strip. Nichrome 80/20 is an ideal material, because it has relatively high resistance and forms an adherent layer of chromium oxide when it is heated for the first time. Material beneath this layer will not oxidize, preventing the wir ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Lead Titanate
Lead(II) titanate is an inorganic compound with the chemical formula PbTiO3. It is the lead salt of titanic acid. Lead(II) titanate is a yellow powder that is insoluble in water. At high temperatures, lead titanate adopts a cubic perovskite structure. At 760 K, the material undergoes a second order phase transition to a tetragonal perovskite structure which exhibits ferroelectricity. Lead titanate is one of the end members of the lead zirconate titanate (, , PZT) system, which is technologically one of the most important ferroelectric and piezoelectric ceramics; PbTiO3 has a high ratio of k33 to kp with a high kt. Lead titanate occurs in nature as mineral macedonite. Toxicity Lead titanate is toxic, like other lead compounds. It irritates skin, mucous membrane A mucous membrane or mucosa is a membrane that lines various cavities in the body of an organism and covers the surface of internal organs. It consists of one or more layers of epithelial cells overlying a lay ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Barium Titanate
Barium titanate (BTO) is an inorganic compound with chemical formula BaTiO3. Barium titanate appears white as a powder and is transparent when prepared as large crystals. It is a Ferroelectricity, ferroelectric, Pyroelectricity, pyroelectric, and Piezoelectricity, piezoelectric ceramic material that exhibits the photorefractive effect. It is used in capacitors, electromechanical transducers and nonlinear optics. Structure The solid exists in one of four polymorphs depending on temperature. From high to low temperature, these crystal symmetries of the four polymorphs are Cubic crystal system, cubic, Tetragonal crystal system, tetragonal, Orthorhombic crystal system, orthorhombic and Trigonal crystal system, rhombohedral crystal structure. All of these phases exhibit the ferroelectric effect apart from the cubic phase. The high temperature cubic phase is easiest to describe, as it consists of regular corner-sharing octahedral TiO6 units that define a cube with O vertices and Ti- ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ceramic
A ceramic is any of the various hard, brittle, heat-resistant and corrosion-resistant materials made by shaping and then firing an inorganic, nonmetallic material, such as clay, at a high temperature. Common examples are earthenware, porcelain, and brick. The earliest ceramics made by humans were pottery objects (''pots,'' ''vessels or vases'') or figurines made from clay, either by itself or mixed with other materials like silica, hardened and sintered in fire. Later, ceramics were glazed and fired to create smooth, colored surfaces, decreasing porosity through the use of glassy, amorphous ceramic coatings on top of the crystalline ceramic substrates. Ceramics now include domestic, industrial and building products, as well as a wide range of materials developed for use in advanced ceramic engineering, such as in semiconductors. The word "'' ceramic''" comes from the Greek word (), "of pottery" or "for pottery", from (), "potter's clay, tile, pottery". The earliest kno ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Positive Temperature Coefficient
A temperature coefficient describes the relative change of a physical property that is associated with a given change in temperature. For a property ''R'' that changes when the temperature changes by ''dT'', the temperature coefficient α is defined by the following equation: :\frac = \alpha\,dT Here α has the dimension of an inverse temperature and can be expressed e.g. in 1/K or K−1. If the temperature coefficient itself does not vary too much with temperature and \alpha\Delta T \ll 1, a linear approximation will be useful in estimating the value ''R'' of a property at a temperature ''T'', given its value ''R''0 at a reference temperature ''T''0: :R(T) = R(T_0)(1 + \alpha\Delta T), where Δ''T'' is the difference between ''T'' and ''T''0. For strongly temperature-dependent α, this approximation is only useful for small temperature differences Δ''T''. Temperature coefficients are specified for various applications, including electric and magnetic properties of materials a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
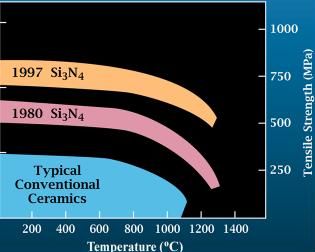
Silicon Nitride
Silicon nitride is a chemical compound of the elements silicon and nitrogen. is the most thermodynamically stable and commercially important of the silicon nitrides, and the term "silicon nitride" commonly refers to this specific composition. It is a white, high-melting-point solid that is relatively chemically inert, being attacked by dilute HF and hot . It is very hard (8.5 on the mohs scale). It has a high thermal stability with strong optical nonlinearities for all-optical applications. Production Silicon nitride is prepared by heating powdered silicon between 1300 °C and 1400 °C in a nitrogen atmosphere: :3 Si + 2 → The silicon sample weight increases progressively due to the chemical combination of silicon and nitrogen. Without an iron catalyst, the reaction is complete after several hours (~7), when no further weight increase due to nitrogen absorption (per gram of silicon) is detected. In addition to , several other silicon nitride phases (with chemical ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Silicon Carbide
Silicon carbide (SiC), also known as carborundum (), is a hard chemical compound containing silicon and carbon. A semiconductor, it occurs in nature as the extremely rare mineral moissanite, but has been mass-produced as a powder and crystal since 1893 for use as an abrasive. Grains of silicon carbide can be bonded together by sintering to form very hard ceramics that are widely used in applications requiring high endurance, such as car brakes, car clutches and ceramic plates in bulletproof vests. Large single crystals of silicon carbide can be grown by the Lely method and they can be cut into gems known as synthetic moissanite. Electronic applications of silicon carbide such as light-emitting diodes (LEDs) and Cat's whisker detector, detectors in early radios were first demonstrated around 1907. SiC is used in semiconductor electronics devices that operate at high temperatures or high voltages, or both. Natural occurrence Naturally occurring moissanite is found in only minut ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Metallurgical Furnace
A metallurgical furnace, more commonly referred to as a furnace, is a device used to heat and melt metal ore to remove gangue, primarily in Metal, iron and steel production. The heat energy to fuel a furnace may be supplied directly by fuel combustion, by electricity such as the electric arc furnace, or through induction heating in induction furnaces. There are several different types of furnaces used in metallurgy to work with specific metal and ores. Smelting furnaces Smelting furnaces are used in smelting to extract metal from ore. Smelting furnaces include: * The blast furnace, used to Redox, reduce iron ore to pig iron ** Cold blast ** Hot blast * Steelmaking furnaces, including: ** Puddling furnace ** Reverberatory furnace ** ** Open hearth furnace ** Basic oxygen furnace ** Electric arc furnace ** Electric induction furnace Other furnaces * Furnaces used to remelt metal in Foundry, foundries. * Furnaces used to reheat and Heat treatment, heat treat metal for use in: ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Semiconductor
A semiconductor is a material which has an electrical resistivity and conductivity, electrical conductivity value falling between that of a electrical conductor, conductor, such as copper, and an insulator (electricity), insulator, such as glass. Its electrical resistivity and conductivity, resistivity falls as its temperature rises; metals behave in the opposite way. Its conducting properties may be altered in useful ways by introducing impurities ("doping (semiconductor), doping") into the crystal structure. When two differently doped regions exist in the same crystal, a semiconductor junction is created. The behavior of charge carriers, which include electrons, ions, and electron holes, at these junctions is the basis of diodes, transistors, and most modern electronics. Some examples of semiconductors are silicon, germanium, gallium arsenide, and elements near the so-called "metalloid staircase" on the periodic table. After silicon, gallium arsenide is the second-most common s ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Heat Treatment Furnaces
In thermodynamics, heat is defined as the form of energy crossing the boundary of a thermodynamic system by virtue of a temperature difference across the boundary. A thermodynamic system does not ''contain'' heat. Nevertheless, the term is also often used to refer to the thermal energy contained in a system as a component of its internal energy and that is reflected in the temperature of the system. For both uses of the term, heat is a form of energy. An example of formal vs. informal usage may be obtained from the right-hand photo, in which the metal bar is "conducting heat" from its hot end to its cold end, but if the metal bar is considered a thermodynamic system, then the energy flowing within the metal bar is called internal energy, not heat. The hot metal bar is also transferring heat to its surroundings, a correct statement for both the strict and loose meanings of ''heat''. Another example of informal usage is the term '' heat content'', used despite the fact that ph ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ceramic Sintering
A ceramic is any of the various hard, brittle, heat-resistant and corrosion-resistant materials made by shaping and then firing an inorganic, nonmetallic material, such as clay, at a high temperature. Common examples are earthenware, porcelain, and brick. The earliest ceramics made by humans were pottery objects (''pots,'' ''vessels or vases'') or figurines made from clay, either by itself or mixed with other materials like silica, hardened and sintered in fire. Later, ceramics were glazed and fired to create smooth, colored surfaces, decreasing porosity through the use of glassy, amorphous ceramic coatings on top of the crystalline ceramic substrates. Ceramics now include domestic, industrial and building products, as well as a wide range of materials developed for use in advanced ceramic engineering, such as in semiconductors. The word "''ceramic''" comes from the Greek word (), "of pottery" or "for pottery", from (), "potter's clay, tile, pottery". The earliest known men ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |