|
Half Metal
A half-metal is any substance that acts as a conductor to electrons of one spin orientation, but as an insulator or semiconductor to those of the opposite orientation. Although all half-metals are ferromagnetic (or ferrimagnetic), most ferromagnets are not half-metals. Many of the known examples of half-metals are oxides, sulfides, or Heusler alloys. In half-metals, the valence band for one spin orientation is partially filled while there is a gap in the density of states for the other spin orientation. This results in conducting behavior for only electrons in the first spin orientation. In some half-metals, the majority spin channel is the conducting one while in others the minority channel is. Half-metals were first described in 1983, as an explanation for the electrical properties of Mn-based Heusler alloys. Some notable half-metals are chromium(IV) oxide, magnetite, and lanthanum strontium manganite (LSMO), as well as chromium arsenide. Half-metals have attracted some i ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Half Metar Sattar
One half ( : halves) is the irreducible fraction resulting from dividing one by two or the fraction resulting from dividing any number by its double. Multiplication by one half is equivalent to division by two, or "halving"; conversely, division by one half is equivalent to multiplication by two, or "doubling". One half often appears in mathematical equations, recipes, measurements, etc. Half can also be said to be one part of something divided into two equal parts. For instance, the area ''S'' of a triangle is computed. :''S'' = × perpendicular height. One half also figures in the formula for calculating figurate numbers, such as triangular numbers and pentagonal numbers: : \frac and in the formula for computing magic constants for magic squares : M_2(n) = \frac \left(n^ + 1\right) The Riemann hypothesis states that every nontrivial complex root of the Riemann zeta function has a real part equal to . One half has two different decimal expansions, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
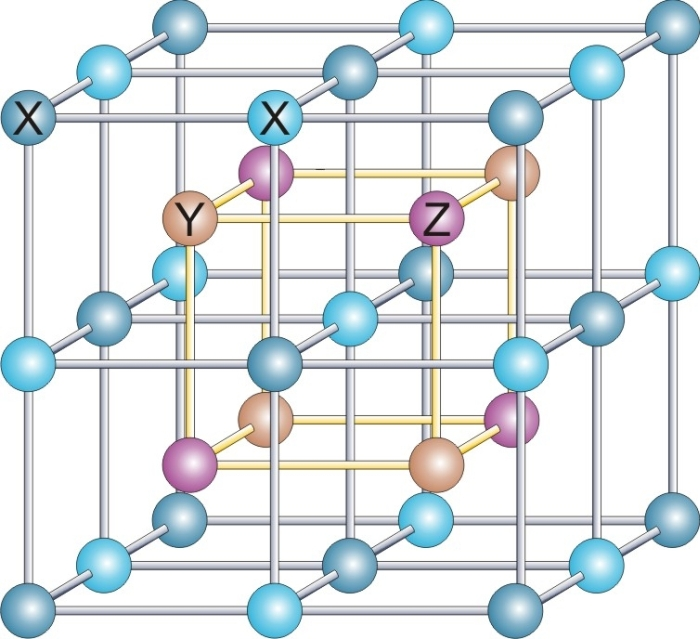
Heusler Alloy
Heusler compounds are magnetic intermetallics with face-centered cubic crystal structure and a composition of XYZ (half-Heuslers) or X2YZ (full-Heuslers), where X and Y are transition metals and Z is in the p-block. The term derives from the name of German mining engineer and chemist Friedrich Heusler, who studied such a compound (Cu2MnAl) in 1903. Many of these compounds exhibit properties relevant to spintronics, such as magnetoresistance, variations of the Hall effect, ferro-, antiferro-, and ferrimagnetism, half- and semimetallicity, semiconductivity with spin filter ability, superconductivity, topological band structure and are actively studied as Thermoelectric materials. Their magnetism results from a double-exchange mechanism between neighboring magnetic ions. Manganese, which sits at the body centers of the cubic structure, was the magnetic ion in the first Heusler compound discovered. (See the Bethe–Slater curve for details of why this happens.) Styles of ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Spintronics
Spintronics (a portmanteau meaning spin transport electronics), also known as spin electronics, is the study of the intrinsic spin of the electron and its associated magnetic moment, in addition to its fundamental electronic charge, in solid-state devices. The field of spintronics concerns spin-charge coupling in metallic systems; the analogous effects in insulators fall into the field of multiferroics. Spintronics fundamentally differs from traditional electronics in that, in addition to charge state, electron spins are exploited as a further degree of freedom, with implications in the efficiency of data storage and transfer. Spintronic systems are most often realised in dilute magnetic semiconductors (DMS) and Heusler alloys and are of particular interest in the field of quantum computing and neuromorphic computing. History Spintronics emerged from discoveries in the 1980s concerning spin-dependent electron transport phenomena in solid-state devices. This includes the observa ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chromium Arsenide
Chromium is a chemical element with the symbol Cr and atomic number 24. It is the first element in group 6. It is a steely-grey, lustrous, hard, and brittle transition metal. Chromium metal is valued for its high corrosion resistance and hardness. A major development in steel production was the discovery that steel could be made highly resistant to corrosion and discoloration by adding metallic chromium to form stainless steel. Stainless steel and chrome plating (electroplating with chromium) together comprise 85% of the commercial use. Chromium is also greatly valued as a metal that is able to be highly polished while resisting tarnishing. Polished chromium reflects almost 70% of the visible spectrum, and almost 90% of infrared light. The name of the element is derived from the Greek word χρῶμα, ''chrōma'', meaning color, because many chromium compounds are intensely colored. Industrial production of chromium proceeds from chromite ore (mostly FeCr2O4) to produce ferr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Lanthanum Strontium Manganite
Lanthanum strontium manganite (LSM or LSMO) is an oxide ceramic material with the general formula La1−xSrxMnO3, where ''x'' describes the doping level. It has a perovskite-based crystal structure, which has the general form ABO3. In the crystal, the 'A' sites are occupied by lanthanum and strontium atoms, and the 'B' sites are occupied by the smaller manganese atoms. In other words, the material consists of lanthanum manganite with some of the lanthanum atoms substitutionally doped with strontium atoms. The strontium (valence 2+) doping on lanthanum (valence 3+) introduces extra holes in the valence band and thus increases electronic conductivity. Depending on the x value in La1−xSrxMnO3, the unit cell of LSMO can be rhombohedral, cubic, or hexagonal. This change in the unit cell is explained on the basis of the Goldschmidt tolerance factor for perovskites. The change in the oxidation state of the Mn cation in LSMO can be readily observed through the position of the XPS pea ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Magnetite
Magnetite is a mineral and one of the main iron ores, with the chemical formula Fe2+Fe3+2O4. It is one of the oxides of iron, and is ferrimagnetic; it is attracted to a magnet and can be magnetized to become a permanent magnet itself. With the exception of extremely rare native iron deposits, it is the most magnetic of all the naturally occurring minerals on Earth. Naturally magnetized pieces of magnetite, called lodestone, will attract small pieces of iron, which is how ancient peoples first discovered the property of magnetism. Magnetite is black or brownish-black with a metallic luster, has a Mohs hardness of 5–6 and leaves a black streak. Small grains of magnetite are very common in igneous and metamorphic rocks. The chemical IUPAC name is iron(II,III) oxide and the common chemical name is ''ferrous-ferric oxide''. Properties In addition to igneous rocks, magnetite also occurs in sedimentary rocks, including banded iron formations and in lake and marine ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chromium(IV) Oxide
Chromium dioxide or chromium(IV) oxide is an inorganic compound with the formula CrO2. It is a black synthetic magnetic solid. It once was widely used in magnetic tape emulsion. With the increasing popularity of CDs and DVDs, the use of chromium(IV) oxide has declined. However, it is still used in data tape applications for enterprise-class storage systems. It is still considered by many oxide and tape manufacturers to have been one of the best magnetic recording particulates ever invented. Preparation and basic properties CrO2 was first prepared by Friedrich Wöhler by decomposition of chromyl chloride. Acicular chromium dioxide was first synthesized in 1956 by Norman L. Cox, a chemist at E.I. DuPont, by decomposing chromium trioxide in the presence of water at a temperature of and a pressure of 200 MPa. The balanced equation for the hydrothermal synthesis is: :3 CrO3 + Cr2O3 → 5 CrO2 + O2 The magnetic crystal that forms is a long, slender glass-like rod � ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Manganese
Manganese is a chemical element with the symbol Mn and atomic number 25. It is a hard, brittle, silvery metal, often found in minerals in combination with iron. Manganese is a transition metal with a multifaceted array of industrial alloy uses, particularly in stainless steels. It improves strength, workability, and resistance to wear. Manganese oxide is used as an oxidising agent; as a rubber additive; and in glass making, fertilisers, and ceramics. Manganese sulfate can be used as a fungicide. Manganese is also an essential human dietary element, important in macronutrient metabolism, bone formation, and free radical defense systems. It is a critical component in dozens of proteins and enzymes. It is found mostly in the bones, but also the liver, kidneys, and brain. In the human brain, the manganese is bound to manganese metalloproteins, most notably glutamine synthetase in astrocytes. Manganese was first isolated in 1774. It is familiar in the laboratory in the form ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sulfide
Sulfide (British English also sulphide) is an inorganic anion of sulfur with the chemical formula S2− or a compound containing one or more S2− ions. Solutions of sulfide salts are corrosive. ''Sulfide'' also refers to chemical compounds large families of inorganic and organic compounds, e.g. lead sulfide and dimethyl sulfide. Hydrogen sulfide (H2S) and bisulfide (SH−) are the conjugate acids of sulfide. Chemical properties The sulfide ion, S2−, does not exist in aqueous alkaline solutions of Na2S. Instead sulfide converts to hydrosulfide: :S2− + H2O → SH− + OH− Upon treatment with an acid, sulfide salts convert to hydrogen sulfide: :S2− + H+ → SH− :SH− + H+ → H2S Oxidation of sulfide is a complicated process. Depending on the conditions, the oxidation can produce elemental sulfur, polysulfides, polythionates, sulfite, or sulfate. Metal sulfides react with halogens, forming sulfur and metal salts. :8 MgS + 8 I2 → S8 + ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electrical Conductor
In physics and electrical engineering, a conductor is an object or type of material that allows the flow of charge (electric current) in one or more directions. Materials made of metal are common electrical conductors. Electric current is generated by the flow of negatively charged electrons, positively charged holes, and positive or negative ions in some cases. In order for current to flow within a closed electrical circuit, it is not necessary for one charged particle to travel from the component producing the current (the current source) to those consuming it (the loads). Instead, the charged particle simply needs to nudge its neighbor a finite amount, who will nudge ''its'' neighbor, and on and on until a particle is nudged into the consumer, thus powering it. Essentially what is occurring is a long chain of momentum transfer between mobile charge carriers; the Drude model of conduction describes this process more rigorously. This momentum transfer model makes metal ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Oxide
An oxide () is a chemical compound that contains at least one oxygen atom and one other element in its chemical formula. "Oxide" itself is the dianion of oxygen, an O2– (molecular) ion. with oxygen in the oxidation state of −2. Most of the Earth's crust consists of oxides. Even materials considered pure elements often develop an oxide coating. For example, aluminium foil develops a thin skin of Al2O3 (called a passivation layer) that protects the foil from further corrosion.Greenwood, N. N.; & Earnshaw, A. (1997). Chemistry of the Elements (2nd Edn.), Oxford:Butterworth-Heinemann. . Stoichiometry (the measurable relationship between reactants and chemical equations of a equation or reaction) Oxides are extraordinarily diverse in terms of stoichiometries and in terms of the structures of each stoichiometry. Most elements form oxides of more than one stoichiometry. A well known example is carbon monoxide and carbon dioxide.Greenwood, N. N.; & Earnshaw, A. (1997). Chemist ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ferrimagnetism
A ferrimagnetic material is a material that has populations of atoms with opposing magnetic moments, as in antiferromagnetism, but these moments are unequal in magnitude so a spontaneous magnetization remains. This can for example occur when the populations consist of different atoms or ions (such as Fe2+ and Fe3+). Ferrimagnetism has often been confused with ferromagnetism. The oldest known magnetic substance, magnetite (Fe3O4), was classified as a ferromagnet before Louis Néel discovered ferrimagnetism in 1948. Since the discovery, numerous uses have been found for ferrimagnetic materials, such as hard drive platters and biomedical applications. History Until the twentieth century, all naturally occurring magnetic substances were called ferromagnets. In 1936, Louis Néel published a paper proposing the existence of a new form of cooperative magnetism he called antiferromagnetism. While working with Mn2Sb, French physicist Charles Guillaud discovered that the curren ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |