|
Emission Peak
A fluorophore (or fluorochrome, similarly to a chromophore) is a fluorescent chemical compound that can re-emit light upon light excitation. Fluorophores typically contain several combined aromatic groups, or planar or cyclic molecules with several π bonds. Fluorophores are sometimes used alone, as a tracer in fluids, as a dye for staining of certain structures, as a substrate of enzymes, or as a probe or indicator (when its fluorescence is affected by environmental aspects such as polarity or ions). More generally they are covalently bonded to a macromolecule, serving as a marker (or dye, or tag, or reporter) for affine or bioactive reagents ( antibodies, peptides, nucleic acids). Fluorophores are notably used to stain tissues, cells, or materials in a variety of analytical methods, i.e., fluorescent imaging and spectroscopy. Fluorescein, via its amine-reactive isothiocyanate derivative fluorescein isothiocyanate (FITC), has been one of the most popular fluorophores. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
FISH 13 21
Fish are Aquatic animal, aquatic, craniate, gill-bearing animals that lack Limb (anatomy), limbs with Digit (anatomy), digits. Included in this definition are the living hagfish, lampreys, and Chondrichthyes, cartilaginous and bony fish as well as various extinct related groups. Approximately 95% of living fish species are ray-finned fish, belonging to the class Actinopterygii, with around 99% of those being teleosts. The earliest organisms that can be classified as fish were soft-bodied chordates that first appeared during the Cambrian period. Although they lacked a vertebrate, true spine, they possessed notochords which allowed them to be more agile than their invertebrate counterparts. Fish would continue to evolve through the Paleozoic era, diversifying into a wide variety of forms. Many fish of the Paleozoic developed placodermi, external armor that protected them from predators. The first fish with jaws appeared in the Silurian period, after which many (such as sharks) b ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Rhodamine
Rhodamine is a family of related dyes, a subset of the triarylmethane dyes. They are derivatives of xanthene. Important members of the rhodamine family are Rhodamine 6G, Rhodamine 123, and Rhodamine B. They are mainly used to dye paper and inks, but they lack the lightfastness for fabric dyeing. Use Aside from their major applications, they are often used as a tracer dye, e.g. to determine the rate and direction of flow and transport of water. Rhodamine dyes fluoresce and can thus be detected easily and inexpensively with instruments called fluorometers. Rhodamine dyes are used extensively in biotechnology applications such as fluorescence microscopy, flow cytometry, fluorescence correlation spectroscopy and ELISA. Rhodamine 123 is used in biochemistry to inhibit mitochondrion function. Rhodamine 123 appears to bind to the mitochondrial membranes and inhibit transport processes, especially the electron transport chain, thus slowing down cellular respiration. It is a substra ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Quenching (fluorescence)
Quenching refers to any process which decreases the fluorescence intensity of a given substance. A variety of processes can result in quenching, such as excited state reactions, energy transfer, complex-formation and collisional quenching. As a consequence, quenching is often heavily dependent on pressure and temperature. Molecular oxygen, iodide ions and acrylamide are common chemical quenchers. The chloride ion is a well known quencher for quinine fluorescence. Quenching poses a problem for non-instant spectroscopic methods, such as laser-induced fluorescence. Quenching is made use of in optode sensors; for instance the quenching effect of oxygen on certain ruthenium complexes allows the measurement of oxygen saturation in solution. Quenching is the basis for Förster resonance energy transfer (FRET) assays. Quenching and dequenching upon interaction with a specific molecular biological target is the basis for activatable optical contrast agents for molecular imaging. Many dyes ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
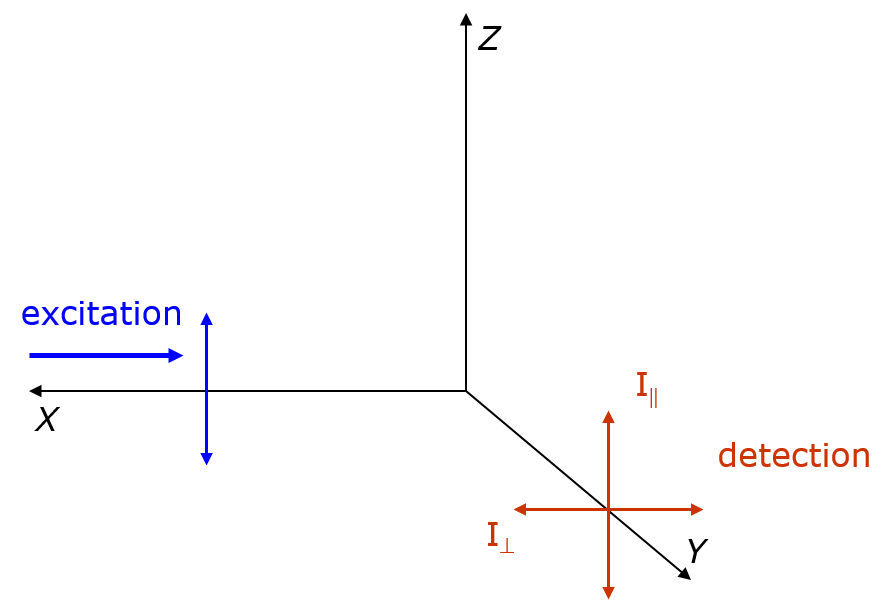
Fluorescence Anisotropy
Fluorescence anisotropy or fluorescence polarization is the phenomenon where the light emitted by a fluorophore has unequal intensities along different axes of polarization. Early pioneers in the field include Aleksander Jablonski, Gregorio Weber, and Andreas Albrecht. The principles of fluorescence polarization and some applications of the method are presented in Lakowicz's book.Lakowicz, J.R., 2006. ''Principles of Fluorescence Spectroscopy'' (3rd ed., Springer. Chapter 10-12 deal with fluorescence polarization spectroscopy.) Definition of fluorescence anisotropy The anisotropy (r) of a light source is defined as the ratio of the polarized component to the total intensity (I_T): r=\frac When the excitation is polarized along the z-axis, emission from the fluorophore is symmetric around the z-axis(Figure). Hence statistically we have I_x=I_y. As I_y=I_, and I_z=I_, we have r=\frac=\frac. Principle – Brownian motion and photoselection In fluorescence, a molecule absorbs ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Fluorescent Protein
Fluorescent proteins include: * Green fluorescent protein (GFP) * Yellow fluorescent protein Yellow fluorescent protein (YFP) is a genetic mutant of green fluorescent protein (GFP) originally derived from the jellyfish '' Aequorea victoria''. Its excitation peak is 513 nm and its emission peak is 527 nm. Like the parent GFP, YFP ... (YFP) * Red fluorescent protein (RFP) {{Short pages monitor ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Quantum Dots
Quantum dots (QDs) are semiconductor particles a few nanometres in size, having optical and electronic properties that differ from those of larger particles as a result of quantum mechanics. They are a central topic in nanotechnology. When the quantum dots are illuminated by UV light, an electron in the quantum dot can be excited to a state of higher energy. In the case of a semiconducting quantum dot, this process corresponds to the transition of an electron from the valence band to the conductance band. The excited electron can drop back into the valence band releasing its energy as light. This light emission ( photoluminescence) is illustrated in the figure on the right. The color of that light depends on the energy difference between the conductance band and the valence band, or the transition between discrete energy states when band structure is no longer a good definition in QDs. In the language of materials science, nanoscale semiconductor materials tightly confine eith ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nanometer
330px, Different lengths as in respect to the molecular scale. The nanometre (international spelling as used by the International Bureau of Weights and Measures; SI symbol: nm) or nanometer (American and British English spelling differences#-re, -er, American spelling) is a units of measurement, unit of length in the International System of Units (SI), equal to one billionth (short scale) of a metre () and to 1000 picometres. One nanometre can be expressed in scientific notation as , and as metres. History The nanometre was formerly known as the millimicrometre – or, more commonly, the millimicron for short – since it is of a micron (micrometre), and was often denoted by the symbol mμ or (more rarely and confusingly, since it logically should refer to a ''millionth'' of a micron) as μμ. Etymology The name combines the SI prefix ''nano-'' (from the Ancient Greek , ', "dwarf") with the parent unit name ''metre'' (from Greek , ', "unit of measurement"). ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Near Infrared
Infrared (IR), sometimes called infrared light, is electromagnetic radiation (EMR) with wavelengths longer than those of visible light. It is therefore invisible to the human eye. IR is generally understood to encompass wavelengths from around 1 millimeter (300 GHz) to the nominal red edge of the visible spectrum, around 700 nanometers (430 THz). Longer IR wavelengths (30 μm-100 μm) are sometimes included as part of the terahertz radiation range. Almost all black-body radiation from objects near room temperature is at infrared wavelengths. As a form of electromagnetic radiation, IR propagates energy and momentum, exerts radiation pressure, and has properties corresponding to both those of a wave and of a particle, the photon. It was long known that fires emit invisible heat; in 1681 the pioneering experimenter Edme Mariotte showed that glass, though transparent to sunlight, obstructed radiant heat. In 1800 the astronomer Sir William Herschel discovered ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Light
Light or visible light is electromagnetic radiation that can be perceived by the human eye. Visible light is usually defined as having wavelengths in the range of 400–700 nanometres (nm), corresponding to frequencies of 750–420 terahertz, between the infrared (with longer wavelengths) and the ultraviolet (with shorter wavelengths). In physics, the term "light" may refer more broadly to electromagnetic radiation of any wavelength, whether visible or not. In this sense, gamma rays, X-rays, microwaves and radio waves are also light. The primary properties of light are intensity, propagation direction, frequency or wavelength spectrum and polarization. Its speed in a vacuum, 299 792 458 metres a second (m/s), is one of the fundamental constants of nature. Like all types of electromagnetic radiation, visible light propagates by massless elementary particles called photons that represents the quanta of electromagnetic field, and can be analyzed as both waves and par ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
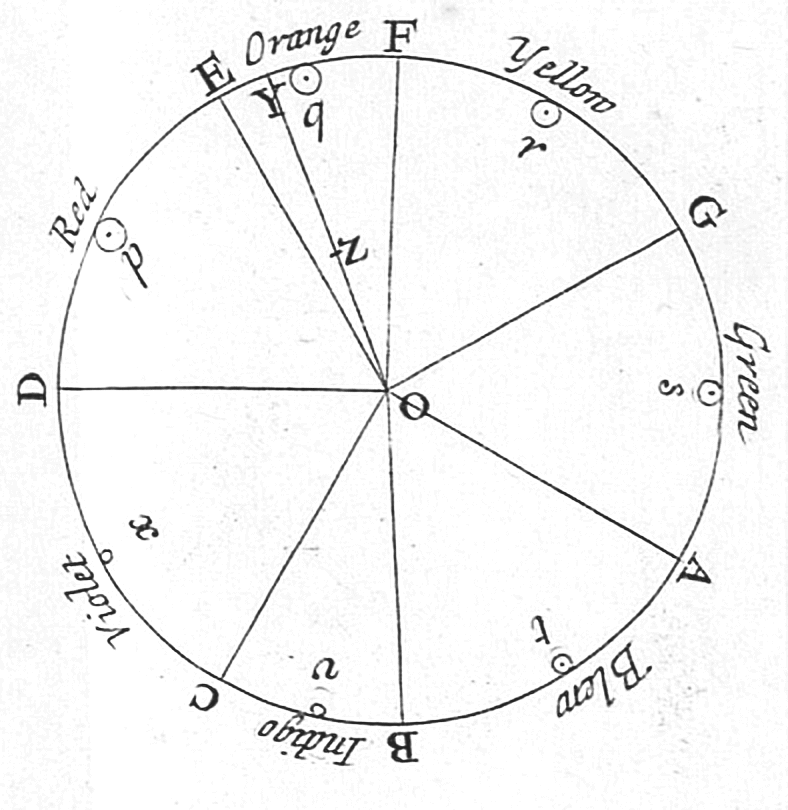
Visible Spectrum
The visible spectrum is the portion of the electromagnetic spectrum that is visual perception, visible to the human eye. Electromagnetic radiation in this range of wavelengths is called ''visible light'' or simply light. A typical human eye will respond to wavelengths from about 380 to about 750 nanometers. In terms of frequency, this corresponds to a band in the vicinity of 400–790 Terahertz (unit), terahertz. These boundaries are not sharply defined and may vary per individual. Under optimal conditions these limits of human perception can extend to 310 nm (ultraviolet) and 1100 nm (near infrared). The optical spectrum is sometimes considered to be the same as the visible spectrum, but some authors define the term more broadly, to include the ultraviolet and infrared parts of the electromagnetic spectrum as well. The spectrum does not contain all the colors that the human visual system can distinguish. ''Excitation purity, Unsaturated colors'' such as pink, or ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ultraviolet
Ultraviolet (UV) is a form of electromagnetic radiation with wavelength from 10 nanometer, nm (with a corresponding frequency around 30 Hertz, PHz) to 400 nm (750 Hertz, THz), shorter than that of visible light, but longer than X-rays. UV radiation is present in sunlight, and constitutes about 10% of the total electromagnetic radiation output from the Sun. It is also produced by electric arcs and specialized lights, such as mercury-vapor lamps, tanning lamps, and black lights. Although long-wavelength ultraviolet is not considered an ionizing radiation because its photons lack the energy to ionization, ionize atoms, it can cause chemical reactions and causes many substances to glow or fluorescence, fluoresce. Consequently, the chemical and biological effects of UV are greater than simple heating effects, and many practical applications of UV radiation derive from its interactions with organic molecules. Short-wave ultraviolet light damages DNA and sterilizes surf ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Quantum Efficiency
The term quantum efficiency (QE) may apply to incident photon to converted electron (IPCE) ratio of a photosensitive device, or it may refer to the TMR effect of a Magnetic Tunnel Junction. This article deals with the term as a measurement of a device's electrical sensitivity to light. In a charge-coupled device (CCD) or other photodetector, it is the ratio between the number of charge carriers collected at either terminal and the number of photons hitting the device's photoreactive surface. As a ratio, QE is dimensionless, but it is closely related to the responsivity, which is expressed in amps per watt. Since the energy of a photon is inversely proportional to its wavelength, QE is often measured over a range of different wavelengths to characterize a device's efficiency at each photon energy level. For typical semiconductor photodetectors, QE drops to zero for photons whose energy is below the band gap. A photographic film typically has a QE of much less than 10%, while C ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |



.jpg)