|
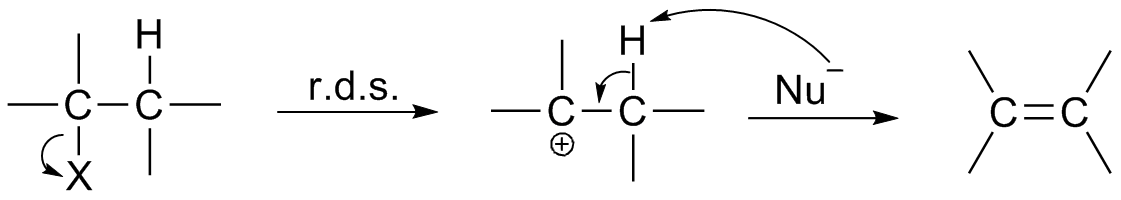
Elementary Step
A reaction step of a chemical reaction is defined as: ''"An elementary reaction, constituting one of the stages of a stepwise reaction in which a reaction intermediate (or, for the first step, the reactants) is converted into the next reaction intermediate (or, for the last step, the products) in the sequence of intermediates between reactants and products"''. – Gold Book
The International Union of Pure and Applied Chemistry publishes many books which contain its complete list of definitions. The definitions are divided into seven "colour books": Gold, Green, Blue, Purple, O ...
[...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chemical Reaction
A chemical reaction is a process that leads to the IUPAC nomenclature for organic transformations, chemical transformation of one set of chemical substances to another. Classically, chemical reactions encompass changes that only involve the positions of electrons in the forming and breaking of chemical bonds between atoms, with no change to the Atomic nucleus, nuclei (no change to the elements present), and can often be described by a chemical equation. Nuclear chemistry is a sub-discipline of chemistry that involves the chemical reactions of unstable and radioactive Chemical element, elements where both electronic and nuclear changes can occur. The substance (or substances) initially involved in a chemical reaction are called reagent, reactants or reagents. Chemical reactions are usually characterized by a chemical change, and they yield one or more Product (chemistry), products, which usually have properties different from the reactants. Reactions often consist of a sequence o ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Elementary Reaction
An elementary reaction is a chemical reaction in which one or more chemical species react directly to form products in a single reaction step and with a single transition state. In practice, a reaction is assumed to be elementary if no reaction intermediates have been detected or need to be postulated to describe the reaction on a molecular scale. An apparently elementary reaction may be in fact a stepwise reaction, i.e. a complicated sequence of chemical reactions, with reaction intermediates of variable lifetimes. In a unimolecular elementary reaction, a molecule dissociates or isomerises to form the products(s) :\mbox \rightarrow \mbox At constant temperature, the rate of such a reaction is proportional to the concentration of the species :\frac=-kmbox In a bimolecular elementary reaction, two atoms, molecules, ions or radicals, and , react together to form the product(s) :\mbox \rightarrow \mbox The rate of such a reaction, at constant temperature, is proportional to the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Stepwise Reaction
In chemistry, a stepwise reaction (also called an overall reaction, complex reaction, and multistep reaction, among others) is a chemical reaction with one or more reaction intermediates, which by definition involves at least two consecutive elementary reactions. In a stepwise reaction, not all bonds are broken and formed at the same time. Hence, intermediates appear in the reaction pathway going from the reactants to the products. A stepwise reaction distinguishes itself from an elementary reaction in which the transformation is assumed to occur in a single step and to pass through a single transition state. In contrast to elementary reactions which follow the law of mass action, the rate law of stepwise reactions is obtained by combining the rate laws of the multiple elementary steps, and can become rather complex. Moreover, when speaking about catalytic reactions, the diffusion may also limit the reaction. In general, however, there is one very slow step, which is the rate-de ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Reaction Intermediate
In chemistry, a reaction intermediate or an intermediate is a molecular entity that is formed from the reactants (or preceding intermediates) but is consumed in further reactions in stepwise chemical reactions that contain multiple elementary steps. Intermediates are the reaction product of one elementary step, but do not appear in the chemical equation for an overall chemical equation. For example, consider this hypothetical stepwise reaction: :A + B -> C + D The reaction includes two elementary steps: :A + B -> X :X -> C + D In this example, X is a reaction intermediate. IUPAC definition The IUPAC Gold Book defines an ''intermediate'' as a compound that has a lifetime greater than a molecular vibration that is formed (directly or indirectly) from the reactants and reacts further to give (either directly or indirectly) the products of a chemical reaction. The lifetime condition distinguishes true, chemically distinct intermediates from vibrational states or such transition st ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Product (chemistry)
Products are the species formed from chemical reactions. During a chemical reaction, reactants are transformed into products after passing through a high energy transition state. This process results in the consumption of the reactants. It can be a spontaneous reaction or mediated by catalysts which lower the energy of the transition state, and by solvents which provide the chemical environment necessary for the reaction to take place. When represented in chemical equations, products are by convention drawn on the right-hand side, even in the case of reversible reactions. The properties of products such as their energies help determine several characteristics of a chemical reaction, such as whether the reaction is exergonic or endergonic. Additionally, the properties of a product can make it easier to extract and purify following a chemical reaction, especially if the product has a different state of matter than the reactants. Spontaneous reaction : R \rightarrow P *Where R is r ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
IUPAC
The International Union of Pure and Applied Chemistry (IUPAC ) is an international federation of National Adhering Organizations working for the advancement of the chemical sciences, especially by developing nomenclature and terminology. It is a member of the International Science Council (ISC). IUPAC is registered in Zürich, Switzerland, and the administrative office, known as the "IUPAC Secretariat", is in Research Triangle Park, North Carolina, United States. This administrative office is headed by IUPAC's executive director, currently Lynn Soby. IUPAC was established in 1919 as the successor of the International Congress of Applied Chemistry for the advancement of chemistry. Its members, the National Adhering Organizations, can be national chemistry societies, national academies of sciences, or other bodies representing chemists. There are fifty-four National Adhering Organizations and three Associate National Adhering Organizations. IUPAC's Inter-divisional Committee on ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Gold Book
The International Union of Pure and Applied Chemistry publishes many books which contain its complete list of definitions. The definitions are divided into seven "colour books": Gold, Green, Blue, Purple, Orange, White, and Red. There is also an eighth book—the "Silver Book". The eight ''colour books'' ''Blue Book'' ''Nomenclature of Organic Chemistry'', commonly referred to by chemists as the ''Blue Book'', is a collection of recommendations on organic chemical nomenclature published at irregular intervals by the International Union of Pure and Applied Chemistry (IUPAC). A full edition was published in 1979, an abridged and updated version of which was published in 1993 as ''A Guide to IUPAC Nomenclature of Organic Compounds''. Both of these are now out-of-print in their paper versions, but are available free of charge in electronic versions. After the release of a draft version for public comment in 2004 and the publication of several revised sections in the journal ''Pure ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |