|
Epimerase
Epimerases and racemases are isomerase enzymes that catalyze the inversion of stereochemistry in biological molecules. Racemases catalyze the stereochemical inversion around the asymmetric carbon atom in a substrate having only one center of asymmetry. Epimerases catalyze the stereochemical inversion of the configuration about an asymmetric carbon atom in a substrate having more than one center of asymmetry, thus interconverting epimers. Human epimerases include methylmalonyl-CoA epimerase, involved in the metabolic breakdown of the amino acids alanine, isoleucine, methionine and valine, and UDP-glucose 4-epimerase, which is used in the final step of galactose metabolism - catalyzing the reversible conversion of UDP-galactose to UDP-glucose. See also * Galactose epimerase deficiency Galactose epimerase deficiency, also known as GALE deficiency, Galactosemia III and UDP-galactose-4-epimerase deficiency, is a rare, autosomal recessive form of galactosemia associated with a defici ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
UDP-glucose 4-epimerase
The enzyme UDP-glucose 4-epimerase (), also known as UDP-galactose 4-epimerase or GALE, is a homodimeric epimerase found in bacterial, fungal, plant, and mammalian cells. This enzyme performs the final step in the Leloir pathway of galactose metabolism, catalyzing the reversible conversion of UDP-galactose to UDP-glucose. GALE tightly binds nicotinamide adenine dinucleotide (NAD+), a co-factor required for catalytic activity. Additionally, human and some bacterial GALE isoforms reversibly catalyze the formation of UDP-''N''-acetylgalactosamine (UDP-GalNAc) from UDP-''N''-acetylglucosamine ( UDP-GlcNAc) in the presence of NAD+, an initial step in glycoprotein or glycolipid synthesis. Historical significance Dr. Luis Leloir deduced the role of GALE in galactose metabolism during his tenure at the Instituto de Investigaciones Bioquímicas del Fundación Campomar, initially terming the enzyme waldenase. Dr. Leloir was awarded the 1970 Nobel Prize in Chemistry for his discovery ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Methylmalonyl-CoA Epimerase
Methylmalonyl CoA epimerase (, ''methylmalonyl-CoA racemase'', ''methylmalonyl coenzyme A racemase'', ''DL-methylmalonyl-CoA racemase'', ''2-methyl-3-oxopropanoyl-CoA 2-epimerase ncorrect') is an enzyme involved in fatty acid catabolism that is encoded in human by the "MCEE" gene located on chromosome 2. It is routinely and incorrectly labeled as "methylmalonyl-CoA racemase". It is not a racemase because the CoA moiety has 5 other stereocenters. Structure The "MCEE" gene is located in the 2p13 region and contains 4 exons, and encodes for a protein that is approximately 18 kDa in size and located to the mitochondrial matrix. Several natural variants in amino acid sequences exist. The structure of the MCEE protein has been resolved by X-ray crystallography at 1.8-angstrom resolution. Function The MCEE gene encodes an enzyme that interconverts D- and L- methylmalonyl-CoA during the degradation of branched-chain amino acids, odd chain-length fatty acids, and other metabolites. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Galactose Epimerase Deficiency
Galactose epimerase deficiency, also known as GALE deficiency, Galactosemia III and UDP-galactose-4-epimerase deficiency, is a rare, autosomal recessive form of galactosemia associated with a deficiency of the enzyme '' galactose epimerase''. Symptoms and signs Symptoms of congenital Type III Galactosemia are apparent from birth, but vary in severity depending on whether the peripheral or generalized disease form is present. Symptoms may include: * Infantile jaundice * Infantile hypotonia * Dysmorphic features * Sensorineural hearing loss * Impaired growth * Cognitive deficiencies * Depletion of cerebellar Purkinje cells * Ovarian failure (POI) and hypertrophic hypergonadism * Liver failure * Renal failure * Splenomegaly * Cataracts Studies of Type III galactosemia symptoms are mostly descriptive, and precise pathogenic mechanisms remain unknown. This is largely due to a lack of functional animal models of classic galactosemia. The recent development of a ''Drosophila melanogaste ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Isomerase
Isomerases are a general class of enzymes that convert a molecule from one isomer to another. Isomerases facilitate intramolecular rearrangements in which bonds are broken and formed. The general form of such a reaction is as follows: A–B → B–A There is only one substrate yielding one product. This product has the same molecular formula as the substrate but differs in bond connectivity or spatial arrangement. Isomerases catalyze reactions across many biological processes, such as in glycolysis and carbohydrate metabolism. Isomerization Isomerases catalyze changes within one molecule. They convert one isomer to another, meaning that the end product has the same molecular formula but a different physical structure. Isomers themselves exist in many varieties but can generally be classified as structural isomers or stereoisomers. Structural isomers have a different ordering of bonds and/or different bond connectivity from one another, as in the case of hexane and it ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Enzyme
Enzymes () are proteins that act as biological catalysts by accelerating chemical reactions. The molecules upon which enzymes may act are called substrates, and the enzyme converts the substrates into different molecules known as products. Almost all metabolic processes in the cell need enzyme catalysis in order to occur at rates fast enough to sustain life. Metabolic pathways depend upon enzymes to catalyze individual steps. The study of enzymes is called ''enzymology'' and the field of pseudoenzyme analysis recognizes that during evolution, some enzymes have lost the ability to carry out biological catalysis, which is often reflected in their amino acid sequences and unusual 'pseudocatalytic' properties. Enzymes are known to catalyze more than 5,000 biochemical reaction types. Other biocatalysts are catalytic RNA molecules, called ribozymes. Enzymes' specificity comes from their unique three-dimensional structures. Like all catalysts, enzymes increase the reaction ra ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Stereochemistry
Stereochemistry, a subdiscipline of chemistry, involves the study of the relative spatial arrangement of atoms that form the structure of molecules and their manipulation. The study of stereochemistry focuses on the relationships between stereoisomers, which by definition have the same molecular formula and sequence of bonded atoms (constitution), but differ in structural formula (the three-dimensional orientations of their atoms in space). For this reason, it is also known as 3D chemistry—the prefix "stereo-" means "three-dimensionality". Stereochemistry spans the entire spectrum of organic, inorganic, biological, physical and especially supramolecular chemistry. Stereochemistry includes methods for determining and describing these relationships; the effect on the physical or biological properties these relationships impart upon the molecules in question, and the manner in which these relationships influence the reactivity of the molecules in question ( dynamic stereochemis ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Epimer
In stereochemistry, an epimer is one of a pair of diastereomers. The two epimers have opposite configuration at only one stereogenic center out of at least two. All other stereogenic centers in the molecules are the same in each. Epimerization is the interconversion of one epimer to the other epimer. Doxorubicin and epirubicin are two epimers that are used as drugs. Examples The stereoisomers β-D- glucopyranose and β-D- mannopyranose are epimers because they differ only in the stereochemistry at the C-2 position. The hydroxy group in β-D-glucopyranose is equatorial (in the "plane" of the ring), while in β-D-mannopyranose the C-2 hydroxy group is axial (up from the "plane" of the ring). These two molecules are epimers but, because they are not mirror images of each other, are not enantiomers. (Enantiomers have the same name, but differ in D and L classification.) They are also not sugar anomers, since it is not the anomeric carbon involved in the stereochemistry. Simila ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
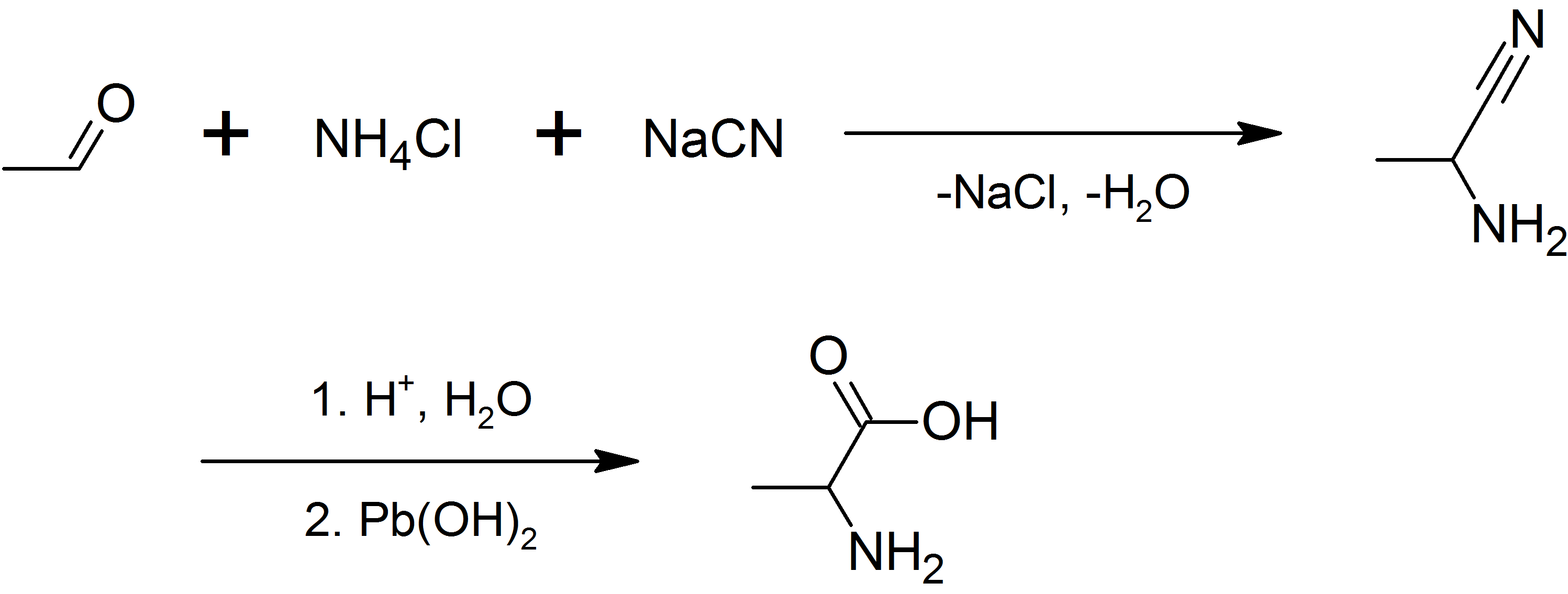
Alanine
Alanine (symbol Ala or A), or α-alanine, is an α-amino acid that is used in the biosynthesis of proteins. It contains an amine group and a carboxylic acid group, both attached to the central carbon atom which also carries a methyl group side chain. Consequently, its IUPAC systematic name is 2-aminopropanoic acid, and it is classified as a nonpolar, aliphatic α-amino acid. Under biological conditions, it exists in its zwitterionic form with its amine group protonated (as −NH3+) and its carboxyl group deprotonated (as −CO2−). It is non-essential to humans as it can be synthesised metabolically and does not need to be present in the diet. It is encoded by all codons starting with GC (GCU, GCC, GCA, and GCG). The L-isomer of alanine (left-handed) is the one that is incorporated into proteins. L-alanine is second only to leucine in rate of occurrence, accounting for 7.8% of the primary structure in a sample of 1,150 proteins. The right-handed form, D-alanine, occurs in p ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Isoleucine
Isoleucine (symbol Ile or I) is an α-amino acid that is used in the biosynthesis of proteins. It contains an α-amino group (which is in the protonated −NH form under biological conditions), an α-carboxylic acid group (which is in the deprotonated −COO form under biological conditions), and a hydrocarbon side chain with a branch (a central carbon atom bound to three other carbon atoms). It is classified as a non-polar, uncharged (at physiological pH), branched-chain, aliphatic amino acid. It is essential in humans, meaning the body cannot synthesize it, and must be ingested in our diet. Isoleucine is synthesized from pyruvate employing leucine biosynthesis enzymes in other organisms such as bacteria. It is encoded by the codons AUU, AUC, and AUA. Metabolism Biosynthesis As an essential nutrient, it is not synthesized in the body, hence it must be ingested, usually as a component of proteins. In plants and microorganisms, it is synthesized via several steps, startin ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Methionine
Methionine (symbol Met or M) () is an essential amino acid in humans. As the precursor of other amino acids such as cysteine and taurine, versatile compounds such as SAM-e, and the important antioxidant glutathione, methionine plays a critical role in the metabolism and health of many species, including humans. It is encoded by the codon AUG. Methionine is also an important part of angiogenesis, the growth of new blood vessels. Supplementation may benefit those suffering from copper poisoning. Overconsumption of methionine, the methyl group donor in DNA methylation, is related to cancer growth in a number of studies. Methionine was first isolated in 1921 by John Howard Mueller. Biochemical details Methionine (abbreviated as Met or M; encoded by the codon AUG) is an α-amino acid that is used in the biosynthesis of proteins. It contains a carboxyl group (which is in the deprotonated −COO− form under biological pH conditions), an amino group (which is in the protonated fo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Valine
Valine (symbol Val or V) is an α-amino acid that is used in the biosynthesis of proteins. It contains an α-amino group (which is in the protonated −NH3+ form under biological conditions), an α- carboxylic acid group (which is in the deprotonated −COO− form under biological conditions), and a side chain isopropyl group, making it a non-polar aliphatic amino acid. It is essential in humans, meaning the body cannot synthesize it: it must be obtained from the diet. Human dietary sources are foods that contain protein, such as meats, dairy products, soy products, beans and legumes. It is encoded by all codons starting with GU (GUU, GUC, GUA, and GUG). History and etymology Valine was first isolated from casein in 1901 by Hermann Emil Fischer. The name valine comes from valeric acid, which in turn is named after the plant valerian due to the presence of the acid in the roots of the plant. Nomenclature According to IUPAC, carbon atoms forming valine are numbered sequentially s ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |