|
Depolariser
A depolarizer or depolariser, in electrochemistry, according to an IUPAC definition, is a synonym of electroactive substance, i.e., a substance which changes its oxidation state, or partakes in a formation or breaking of chemical bonds, in a charge-transfer step of an electrochemical reaction. In the battery industry, the term "depolarizer" has been used to denote a substance used in a primary cell to prevent buildup of hydrogen gas bubbles."McGraw-Hill Dictionary of Scientific & Technical Terms", McGraw-Hill, Inc., 2003. A battery depolarizer takes up electrons during discharge of the cell; therefore, it is always an oxidizing agent. The term "depolarizer" can be considered as outdated or misleading, since it is based on the concept of " polarization" which is hardly realistic in many cases. Polarization Under certain conditions for some electrochemical cells, especially if they use an aqueous electrolyte, hydrogen ions can be converted into hydrogen atoms and H2 molecules. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dry Cell
upLine art drawing of a dry cell: 1. brass cap, 2. plastic seal, 3. expansion space, 4. porous cardboard, 5. zinc can, 6. carbon rod, 7. chemical mixture A dry cell is a type of electric battery, commonly used for portable electrical devices. Unlike wet cell batteries, which have a liquid electrolyte, dry cells use an electrolyte in the form of a paste, and are thus less susceptible to leakage. The dry cell was developed in 1886 by the German scientist Carl Gassner, after development of wet zinc–carbon batteries by Georges Leclanché in 1866. A type of dry cell was also developed by the Japanese Sakizō Yai in 1887. History left, upDry cell battery by Wilhelm Hellesen 1890 Many experimenters tried to immobilize the electrolyte of an electrochemical cell to make it more convenient to use. The Zamboni pile of 1812 is a high-voltage dry battery but capable of delivering only minute currents. Various experiments were made with cellulose, sawdust, spun glass, asbestos fiber ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Battery (electricity)
An electric battery is a source of electric power consisting of one or more electrochemical cells with external connections for powering electrical devices. When a battery is supplying power, its positive terminal is the cathode and its negative terminal is the anode. The terminal marked negative is the source of electrons that will flow through an external electric circuit to the positive terminal. When a battery is connected to an external electric load, a redox reaction converts high-energy reactants to lower-energy products, and the free-energy difference is delivered to the external circuit as electrical energy. Historically the term "battery" specifically referred to a device composed of multiple cells; however, the usage has evolved to include devices composed of a single cell. Primary (single-use or "disposable") batteries are used once and discarded, as the electrode materials are irreversibly changed during discharge; a common example is the alkaline battery used ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chromic Acid
The term chromic acid is usually used for a mixture made by adding concentrated sulfuric acid to a dichromate, which may contain a variety of compounds, including solid chromium trioxide. This kind of chromic acid may be used as a cleaning mixture for glass. Chromic acid may also refer to the molecular species, H2CrO4 of which the trioxide is the anhydride. Chromic acid features chromium in an oxidation state of +6 (or VI). It is a strong and corrosive oxidising agent. Molecular chromic acid Molecular chromic acid, H2CrO4, has much in common with sulfuric acid, H2SO4. Only sulfuric acid can be classified as part of the 7 strong acids list. Due to the laws pertinent to the concept of "first order ionization energy", the first proton is lost most easily. It behaves extremely similar to sulfuric acid deprotonation. Since the process of polyvalent acid-base titrations have more than one proton (especially when the acid is starting substance and the base is the titrant), proton ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Everyman's Encyclopaedia
''Everyman's Encyclopaedia'' is an encyclopedia published by Joseph Dent from 1913 as part of the Everyman's Library. The set was descended from the 1850s ''English Cyclopaedia'' of 1854, which in turn was based on the ''Penny Cyclopaedia'' of the 1830s. Originally published in 1913–14 by J.M. Dent in London and, simultaneously, E. P. Dutton in New York, the encyclopedia proved popular due to its low cost, small size and concise but highly accurate articles. The set did, however, lack much illustrative material. Andrew Boyle was credited as the editor. A second edition was published in 1931–32. The title was slightly different, changing from ''The Everyman Encyclopedia'' to ''Everyman's Encyclopaedia''. This set had 12 volumes, 7 million words, 9,000 pages and 50,000 articles. There was also an optional atlas volume. Athelstan Ridgway was credited as the editor. The third edition was published in 1950, again under Ridgways direction, and published in London by Dent, but no ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Clark Cell
The Clark cell, invented by English engineer Josiah Latimer Clark in 1873, is a wet-chemical cell (colloquially: ''battery'') that produces a highly stable voltage. In 1893, the output of the Clark cell at 15 °C was defined by the International Electrical Congress as 1.434 volts, and this definition became law in the United States in 1894. This definition was later supplanted by one based on the Weston cell. Chemistry Clark cells use a zinc, or zinc amalgam, anode and a mercury cathode in a saturated aqueous solution of zinc sulfate, with a paste of mercurous sulfate as depolarizer. Construction Original cell Clark's original cell was set up in a glass jar in a similar way to a gravity Daniell cell. The copper cathode was replaced by a pool of mercury at the bottom of the jar. Above this was the mercurous sulfate paste and, above that, the zinc sulfate solution. A short zinc rod dipped into the zinc sulfate solution. The zinc rod was supported by a cork with two holes � ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Weston Cell
The Weston standard cell is a wet-chemical cell that produces a highly stable voltage suitable as a laboratory standard for calibration In measurement technology and metrology, calibration is the comparison of measurement values delivered by a device under test with those of a calibration standard of known accuracy. Such a standard could be another measurement device of know ... of voltmeters. Invented by Edward Weston (chemist), Edward Weston in 1893, it was adopted as the International Standard for electromotive force, EMF from 1911 until superseded by the Josephson voltage standard in 1990. Chemistry The anode is an amalgam (chemistry), amalgam of cadmium with mercury (element), mercury with a cathode of pure mercury over which a paste of mercurous sulfate and mercury is placed. The electrolyte is a saturated solution of cadmium sulfate, and the depolarizer is a paste of mercurous sulfate. As shown in the illustration, the cell is set up in an H-shaped glass vessel ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Mercury(I) Sulfate
Mercury(I) sulfate, commonly called mercurous sulphate ( UK) or mercurous sulfate ( US) is the chemical compound Hg2SO4. Mercury(I) sulfate is a metallic compound that is a white, pale yellow or beige powder. It is a metallic salt of sulfuric acid formed by replacing both hydrogen atoms with mercury(I). It is highly toxic; it could be fatal if inhaled, ingested, or absorbed by skin. Structure In the crystal, mercurous sulfate is made up of Hg22+ center with an Hg-Hg distance of about 2.50 Å. The SO42− anions form both long and short Hg-O bonds ranging from 2.23 to 2.93 Å. Focusing on the shorter Hg-O bonds, the Hg – Hg – O bond angle is 165°±1°. Preparation One way to prepare mercury(I) sulfate is to mix the acidic solution of mercury(I) nitrate with 1 to 6 sulfuric acid solution:, [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Daniell Cell
The Daniell cell is a type of electrochemical cell invented in 1836 by John Frederic Daniell, a British chemist and meteorologist, and consists of a copper pot filled with a copper (II) sulfate solution, in which is immersed an unglazed earthenware container filled with sulfuric acid and a zinc electrode. He was searching for a way to eliminate the hydrogen bubble problem found in the voltaic pile, and his solution was to use a second electrolyte to consume the hydrogen produced by the first. Zinc sulfate may be substituted for the sulfuric acid. The Daniell cell was a great improvement over the existing technology used in the early days of battery development. A later variant of the Daniell cell called the gravity cell or crowfoot cell was invented in the 1860s by a Frenchman named Callaud and became a popular choice for electrical telegraphy. The Daniell cell is also the historical basis for the contemporary definition of the volt, which is the unit of electromotive force in t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Copper(II) Sulfate
Copper(II) sulfate, also known as copper sulphate, is an inorganic compound with the chemical formula . It forms hydrates , where ''n'' can range from 1 to 7. The pentahydrate (''n'' = 5), a bright blue crystal, is the most commonly encountered hydrate of copper(II) sulfate. Older names for the pentahydrate include blue vitriol, bluestone, vitriol of copper,Antoine-François de Fourcroy, tr. by Robert Heron (1796) "Elements of Chemistry, and Natural History: To which is Prefixed the Philosophy of Chemistry". J. Murray and others, Edinburgh. Page 348. and Roman vitriol.Oxford University Press,Roman vitriol, Oxford Living Dictionaries. Accessed on 2016-11-13 It exothermically dissolves in water to give the aquo complex , which has octahedral molecular geometry. The structure of the solid pentahydrate reveals a polymeric structure wherein copper is again octahedral but bound to four water ligands. The centers are interconnected by sulfate anions to form chains. Anhydrous copper sulfa ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Silver-oxide Battery
A silver-oxide battery (IEC code: S) is a primary cell using silver oxide as the cathode material and zinc for the anode. These cells maintain a nearly constant nominal voltage during discharge until fully depleted. They are available in small sizes as button cells, where the amount of silver used is minimal and not a prohibitively expensive contributor to the overall product cost. Silver-oxide primary batteries account for 30% of all primary battery sales in Japan (64 out of 212 million in February 2020). Silver-oxide batteries were used on Apollo program lunar missions for the lunar module and lunar rover power supplies because of their high energy-to-weight ratio.Lyons, Pete; "10 Best Ahead-of-Their-Time Machines", ''Car and Driver'', Jan. 1988, p.78 Chemistry A silver-oxide battery uses silver(I) oxide as the positive electrode (cathode), zinc as the negative electrode (anode), plus an alkaline electrolyte, usually sodium hydroxide (NaOH) or potassium hydroxide (KOH). ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Silver Oxide
Silver oxide is the chemical compound with the formula Ag2O. It is a fine black or dark brown powder that is used to prepare other silver compounds. Preparation Silver oxide can be prepared by combining aqueous solutions of silver nitrate and an alkali hydroxide. This reaction does not afford appreciable amounts of silver hydroxide due to the favorable energetics for the following reaction: :2 AgOH -> Ag2O + H2O ( p''K'' = 2.875) With suitably controlled conditions, this reaction can be used to prepare Ag2O powder with properties suitable for several uses including as a fine grained conductive paste filler. Structure and properties Ag2O features linear, two-coordinate Ag centers linked by tetrahedral oxides. It is isostructural with Cu2O. It "dissolves" in solvents that degrade it. It is slightly soluble in water due to the formation of the ion and possibly related hydrolysis products. It is soluble in ammonia solution, producing active compound of Tollens' reagent. A sl ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
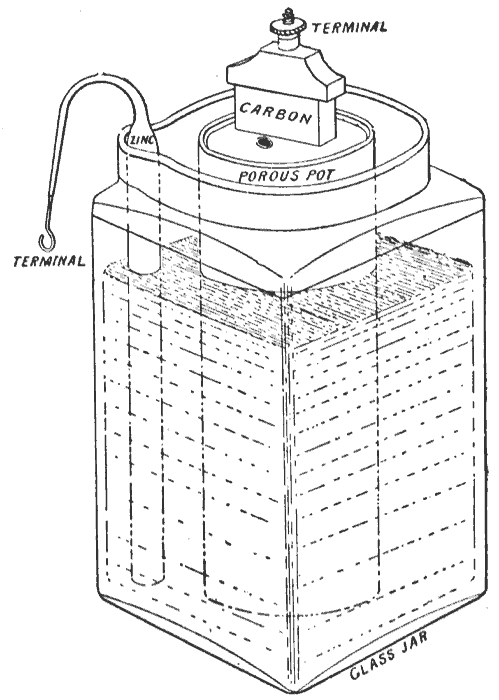
Leclanché Cell
The Leclanché cell is a battery invented and patented by the French scientist Georges Leclanché in 1866. The battery contained a conducting solution (electrolyte) of ammonium chloride, a cathode (positive terminal) of carbon, a depolarizer of manganese dioxide (oxidizer), and an anode (negative terminal) of zinc (reductant). The chemistry of this cell was later successfully adapted to manufacture a dry cell. History In 1866, Georges Leclanché invented a battery that consisted of a zinc anode and a manganese dioxide cathode wrapped in a porous material, dipped in a jar of ammonium chloride solution. The manganese dioxide cathode had a little carbon mixed into it as well, which improved conductivity and absorption. It provided a voltage of 1.4 volts.Simms, J.W. (1965) ''The Boy Electrician'' M.I.E.E. p. 61 This cell achieved very quick success in telegraphy, signalling and electric bell work. The dry cell form was used to power early telephones—usually from an adjacent woo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |



.png)