|
Chimerin
Chimerin is a type of nerve tissue protein. Chimerins are a family of non-protein kinase C phorbol ester receptors. They were the first phorbal ester receptors to be discovered within this family. They are represented as a family of four closely bound GAPs1 (GTPase-activating proteins). These small GTPases were once characterized as high affinity intracellular receptors for the second messenger diacylglycerol (DAG) and the phorbol ester tumor promoters. The name stems from their resemblance to the "chimera." Types Types include: * Chimerin 1 * Chimerin 2 There are four known isoforms of the chimerin protein. These include α1, α2, β1, and β2. α1-Chimerin was the first protein to be isolated from the brain. The other domains were discovered through alternative splicing. The α and β isoforms are almost identical, the key difference stems from the SH2 domain The SH2 (Src Homology 2) domain is a structurally conserved protein domain contained within the Src oncoprotein an ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chimerin 1
Chimerin 1 (CHN1), also known as alpha-1-chimerin, n-chimerin, is a protein which in humans is encoded by the ''CHN1'' gene. Chimerin 1 is a GTPase activating protein specific for RAC GTP-binding proteins. It is expressed primarily in the brain and may be involved in signal transduction. This gene encodes GTPase-activating protein for p21-rac and a phorbol ester receptor. It plays an important role in ocular motor axon pathfinding. Function CHN1 is a three-domain protein with the N-terminal SH2 domain, the C-terminal RhoGAP domain and the central C1 domain similar to protein kinase C In cell biology, Protein kinase C, commonly abbreviated to PKC (EC 2.7.11.13), is a family of protein kinase enzymes that are involved in controlling the function of other proteins through the phosphorylation of hydroxyl groups of serine and t .... When lipid diacylglycerol (DAG) binds to the C1 domain, CHN1 is transferred to the plasma membrane and negatively regulates Rho-family small ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nerve Tissue Protein
Nerve tissue is a biological molecule related to the function and maintenance of normal nervous tissue. An example would include, for example, the generation of myelin which insulates and protects nerves. These are typically calcium-binding proteins Calcium-binding proteins are proteins that participate in calcium cell signalling pathways by binding to Ca2+, the calcium ion that plays an important role in many cellular processes. Calcium-binding proteins have specific domains that bind to cal .... Myelination and Peripheral Nervous System There are two types of myelin. The first is oligodendrocyte, which can be found in the mammalian Central Nervous System (CNS). The second is Schwann cells, which are found in the Peripheral Nervous System (PNS). Myelination of axons by these Schwann cells are essential for normal nerve function. Peripheral nerves rely on communication between axons and Schwaan cells. Maintenance of myelin Prion protein triggers are an important factor in the sign ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chimerin 2
Chimerin 2 (beta-chimaerin) is a protein that in humans is encoded by the ''CHN2'' gene. This gene is a member of the chimerin family and encodes a protein with a phorbol-ester/diacylglycerol-type zinc finger, a Rho-GAP domain and an SH2 domain. This protein has GTPase-activating protein activity that is regulated by phospholipid binding and binding of diacylglycerol (DAG) induces translocation of the protein from the cytosol to the Golgi apparatus The Golgi apparatus (), also known as the Golgi complex, Golgi body, or simply the Golgi, is an organelle found in most eukaryotic cells. Part of the endomembrane system in the cytoplasm, it packages proteins into membrane-bound vesicles ins ... membrane. The protein plays a role in the proliferation and migration of smooth muscle cells. Decreased expression of this gene is associated with high-grade gliomas and breast tumors, and increased expression of this gene is associated with lymphomas. Mutations in this gene have been as ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
SH2 Domain
The SH2 (Src Homology 2) domain is a structurally conserved protein domain contained within the Src oncoprotein and in many other intracellular signal-transducing proteins. SH2 domains allow proteins containing those domains to dock to phosphorylated tyrosine residues on other proteins. SH2 domains are commonly found in adaptor proteins that aid in the signal transduction of receptor tyrosine kinase pathways. Background SH2 is conserved by signalization of protein tyrosine kinase, which are binding on phosphotyrosine (pTyr). In the human proteome the class of pTyr-selective recognition domains is represented by SH2 domains. The N-terminal SH2 domains of cytoplasmic tyrosine kinase was at the beginning of evolution evolved with the occurrence of tyrosine phosphorylation. At the beginning it was supposed that, these domains serve as a substrate for their target kinase. Protein-protein interactions play a major role in cellular growth and development. Modular domains, which are t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Protein Kinase C
In cell biology, Protein kinase C, commonly abbreviated to PKC (EC 2.7.11.13), is a family of protein kinase enzymes that are involved in controlling the function of other proteins through the phosphorylation of hydroxyl groups of serine and threonine amino acid residues on these proteins, or a member of this family. PKC enzymes in turn are activated by signals such as increases in the concentration of diacylglycerol (DAG) or calcium ions (Ca2+). Hence PKC enzymes play important roles in several signal transduction cascades. In biochemistry, the PKC family consists of fifteen isozymes in humans. They are divided into three subfamilies, based on their second messenger requirements: conventional (or classical), novel, and atypical. Conventional (c)PKCs contain the isoforms α, βI, βII, and γ. These require Ca2+, DAG, and a phospholipid such as phosphatidylserine for activation. Novel (n)PKCs include the δ, ε, η, and θ isoforms, and require DAG, but do not require Ca2+ for ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phorbol Ester
Phorbol esters are a class of chemical compounds found in a variety of plants, particularly in the families Euphorbiaceae and Thymelaeaceae. Chemically, they are ester derivatives of the tetracyclic diterpenoid phorbol. Biological activity Protein kinase C (PKC) is a phorbol ester receptor. Phorbol esters can stimulate PKC in a similar way to diglycerides. Phorbol esters are known for their ability to promote tumors. In particular, 12-O-tetradecanoylphorbol-13-acetate (TPA) is used as a biomedical research tool in models of carcinogenesis. Plants that contain phorbol esters are often poisonous Poison is a chemical substance that has a detrimental effect to life. The term is used in a wide range of scientific fields and industries, where it is often specifically defined. It may also be applied colloquially or figuratively, with a broa .... References {{Chem-stub Diterpenes Carboxylate esters Cyclopentenes Plant toxins ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
GTPase-activating Protein
GTPase-activating proteins or GTPase-accelerating proteins (GAPs) are a family of regulatory proteins whose members can bind to activated G proteins and stimulate their GTPase activity, with the result of terminating the signaling event. GAPs are also known as RGS protein, or RGS proteins,Kimple, A.J. "Structural Determinants of G-protein α Subunit Selectivity by Regulator of G-protein Signaling 2 (RGS2)". ''The Journal of Biological Chemistry''. 284 (2009): 19402-19411. and these proteins are crucial in controlling the activity of G proteins. Regulation of G proteins is important because these proteins are involved in a variety of important cellular processes. The large G proteins, for example, are involved in transduction of signaling from the G protein-coupled receptor for a variety of signaling processes like hormonal signaling, and small G proteins are involved in processes like cellular trafficking and cell cycling. GAP's role in this function is to turn the G protein's ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
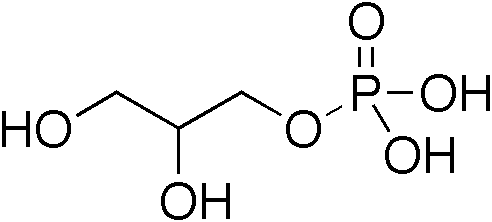
Diacylglycerol
A diglyceride, or diacylglycerol (DAG), is a glyceride consisting of two fatty acid chains covalently bonded to a glycerol molecule through ester linkages. Two possible forms exist, 1,2-diacylglycerols and 1,3-diacylglycerols. DAGs can act as surfactants and are commonly used as emulsifiers in processed foods. DAG-enriched oil (particularly 1,3-DAG) has been investigated extensively as a fat substitute due to its ability to suppress the accumulation of body fat; with total annual sales of approximately USD 200 million in Japan since its introduction in the late 1990s till 2009. Production Diglycerides are a minor component of many seed oils and are normally present at ~1–6%; or in the case of cottonseed oil as much as 10%. Industrial production is primarily achieved by a glycerolysis reaction between triglycerides and glycerol. The raw materials for this may be either vegetable oils or animal fats. Food additive Diglycerides, generally in a mix with monoglycerides (E471), a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
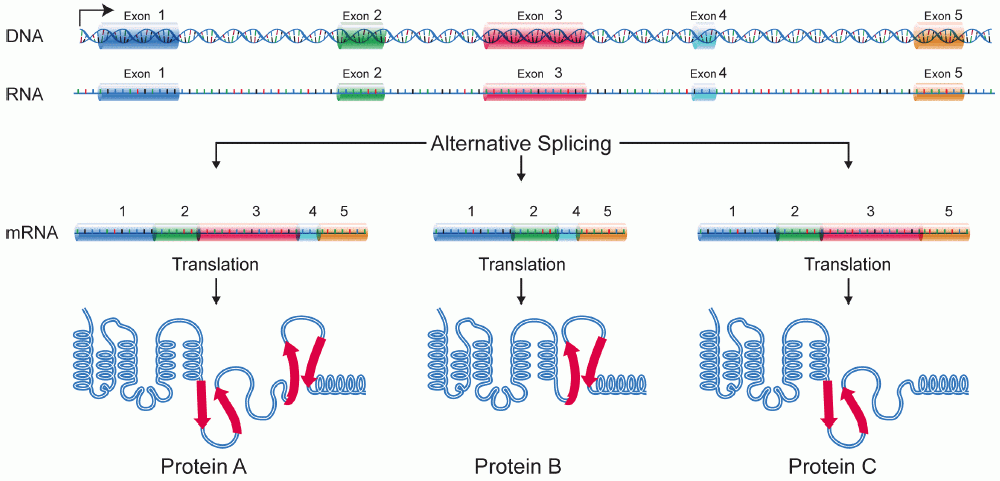
Protein Isoform
A protein isoform, or "protein variant", is a member of a set of highly similar proteins that originate from a single gene or gene family and are the result of genetic differences. While many perform the same or similar biological roles, some isoforms have unique functions. A set of protein isoforms may be formed from alternative splicings, variable promoter usage, or other post-transcriptional modifications of a single gene; post-translational modifications are generally not considered. (For that, see Proteoforms.) Through RNA splicing mechanisms, mRNA has the ability to select different protein-coding segments ( exons) of a gene, or even different parts of exons from RNA to form different mRNA sequences. Each unique sequence produces a specific form of a protein. The discovery of isoforms could explain the discrepancy between the small number of protein coding regions genes revealed by the human genome project and the large diversity of proteins seen in an organism: different ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Alternative Splicing
Alternative splicing, or alternative RNA splicing, or differential splicing, is an alternative splicing process during gene expression that allows a single gene to code for multiple proteins. In this process, particular exons of a gene may be included within or excluded from the final, processed messenger RNA (mRNA) produced from that gene. This means the exons are joined in different combinations, leading to different (alternative) mRNA strands. Consequently, the proteins translated from alternatively spliced mRNAs will contain differences in their amino acid sequence and, often, in their biological functions (see Figure). Biologically relevant alternative splicing occurs as a normal phenomenon in eukaryotes, where it increases the number of proteins that can be encoded by the genome. In humans, it is widely believed that ~95% of multi-exonic genes are alternatively spliced to produce functional alternative products from the same gene but many scientists believe that most o ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |