|
Cobalt Green
Cobalt green is an ambiguous term for either of two families of green inorganic pigments. Both are obtained by doping cobalt(II) oxide into colorless host oxides. Spinel-based cobalt green Doping Co(II) into Mg(II) and Zn(II) sites of Mg2TiO4 and Zn2TiO4, respectively gives one family of cobalt greens. These materials adopt the spinel structure.Völz, Hans G. ''et al.'' "Pigments, Inorganic" in ''Ullmann's Encyclopedia of Industrial Chemistry'', 2006 Wiley-VCH, Weinheim. . Rinman's green Rinman's green, also referred to as Rinmann's green, is obtained by doping cobalt(II) oxide into zinc oxide. Sven Rinman, a Swedish chemist, discovered this compound in 1780. Zinc oxide–derived pigments have been used in many industries and processes. It is rarely used because it is a weak chromophore and relatively expensive compared to chromium(III) oxide. The structure and color of compositions Zn1−xCoxO depends on the value of x. For x ≤ 0.3, the material adopts the Wurzite str ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pigment
A pigment is a colored material that is completely or nearly insoluble in water. In contrast, dyes are typically soluble, at least at some stage in their use. Generally dyes are often organic compounds whereas pigments are often inorganic compounds. Pigments of prehistoric and historic value include ochre, charcoal, and lapis lazuli. Economic impact In 2006, around 7.4 million tons of inorganic, organic, and special pigments were marketed worldwide. Estimated at around US$14.86 billion in 2018 and will rise at over 4.9% CAGR from 2019 to 2026. The global demand for pigments was roughly US$20.5 billion in 2009. According to an April 2018 report by ''Bloomberg Businessweek'', the estimated value of the pigment industry globally is $30 billion. The value of titanium dioxide – used to enhance the white brightness of many products – was placed at $13.2 billion per year, while the color Ferrari red is valued at $300 million each year. Physical principles ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
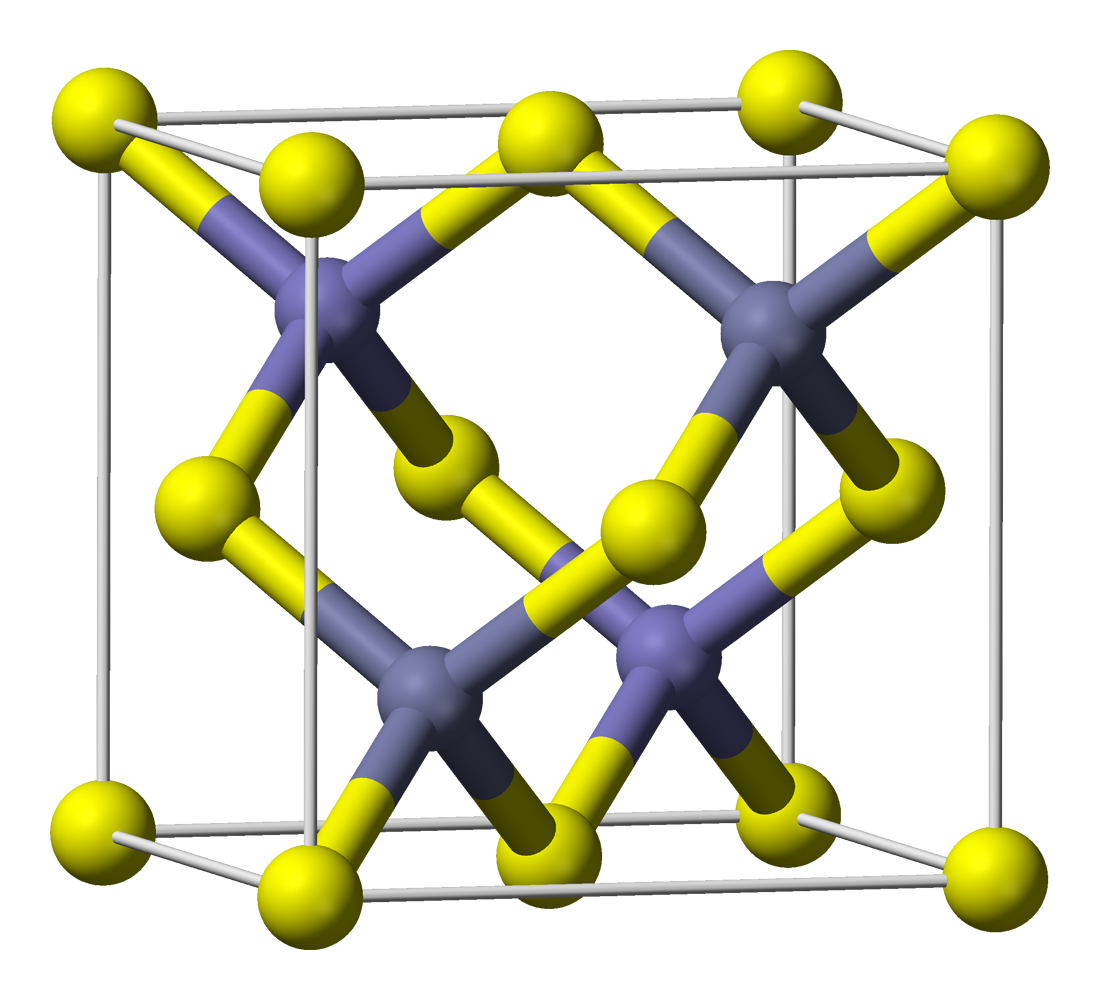
Spinel
Spinel () is the magnesium/aluminium member of the larger spinel group of minerals. It has the formula in the cubic crystal system. Its name comes from the Latin word , which means ''spine'' in reference to its pointed crystals. Properties Spinel crystallizes in the isometric system; common crystal forms are octahedra, usually twinned. It has no true cleavage, but shows an octahedral parting and a conchoidal fracture. Its hardness is 8, its specific gravity is 3.5–4.1, and it is transparent to opaque with a vitreous to dull luster. It may be colorless, but is usually various shades of red, lavender, blue, green, brown, black, or yellow. Some spinels are among the most famous gemstones; among them are the Black Prince's Ruby and the "Timur ruby" in the British Crown Jewels, and the "Côte de Bretagne", formerly from the French Crown jewels. The Samarian Spinel is the largest known spinel in the world, weighing . The transparent red spinels were called spinel-rubies or b ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sven Rinman
Sven Rinman (23 June (N.S)/12 June (O.S) 1720 – 20 December 1792) was a Swedish chemist and mineralogist who discovered the pigment A pigment is a colored material that is completely or nearly insoluble in water. In contrast, dyes are typically soluble, at least at some stage in their use. Generally dyes are often organic compounds whereas pigments are often inorganic compou ... cobalt green, sometimes also called Rinman's green. He had a great influence on mining and the steel production in Sweden. Life and work Rinman was born in Uppsala on 23 June(N.S) 1720. At the age of 20 he became an intern at the Royal Bureau of Mines. In the years 1746 and 1747 Rinman visited several European countries to improve his knowledge on mining and metallurgy. After his return he became inspector in several mines and metal works. His research on mining and metallurgy had a big influence on the Swedish industry. He was elected a member of the Royal Swedish Academy of Sciences in 1753. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Zinc Oxide
Zinc oxide is an inorganic compound with the formula . It is a white powder that is insoluble in water. ZnO is used as an additive in numerous materials and products including cosmetics, food supplements, rubbers, plastics, ceramics, glass, cement, lubricants, paints, ointments, adhesives, sealants, pigments, foods, batteries, ferrites, fire retardants, and first-aid tapes. Although it occurs naturally as the mineral zincite, most zinc oxide is produced synthetically. ZnO is a wide-band gap semiconductor of the II-VI semiconductor group. The native doping of the semiconductor due to oxygen vacancies or zinc interstitials is n-type. Other favorable properties include good transparency, high electron mobility, wide band gap, and strong room-temperature luminescence. Those properties make ZnO valuable for a variety of emerging applications: transparent electrodes in liquid crystal displays, energy-saving or heat-protecting windows, and electronics as thin-film transistors and lig ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chromium(III) Oxide
Chromium(III) oxide (or chromia) is an inorganic compound with the formula . It is one of the principal oxides of chromium and is used as a pigment. In nature, it occurs as the rare mineral eskolaite. Structure and properties has the corundum structure, consisting of a hexagonal close packed array of oxide anions with of the octahedral holes occupied by chromium. Similar to corundum, is a hard, brittle material (Mohs hardness 8 to 8.5). It is antiferromagnetic up to 307 K, the Néel temperature. It is not readily attacked by acids. Occurrence occurs naturally as the mineral eskolaite, which is found in chromium-rich tremolite skarns, metaquartzites, and chlorite veins. Eskolaite is also a rare component of chondrite meteorites. The mineral is named after Finnish geologist Pentti Eskola. Production The Parisians Pannetier and Binet first prepared the transparent hydrated form of in 1838 via a secret process, sold as a pigment. It is derived from the mineral chromite, . T ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Wurzite
Zinc sulfide (or zinc sulphide) is an inorganic compound with the chemical formula of ZnS. This is the main form of zinc found in nature, where it mainly occurs as the mineral sphalerite. Although this mineral is usually black because of various impurities, the pure material is white, and it is widely used as a pigment. In its dense synthetic form, zinc sulfide can be transparent, and it is used as a window for visible optics and infrared optics. Structure ZnS exists in two main crystalline forms. This dualism is an example of polymorphism. In each form, the coordination geometry at Zn and S is tetrahedral. The more stable cubic form is known also as zinc blende or sphalerite. The hexagonal form is known as the mineral wurtzite, although it also can be produced synthetically.. The transition from the sphalerite form to the wurtzite form occurs at around 1020 °C. A tetragonal form is also known as the very rare mineral called polhemusite, with the formula . Applicatio ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sodium Chloride
Sodium chloride , commonly known as salt (although sea salt also contains other chemical salts), is an ionic compound with the chemical formula NaCl, representing a 1:1 ratio of sodium and chloride ions. With molar masses of 22.99 and 35.45 g/mol respectively, 100 g of NaCl contains 39.34 g Na and 60.66 g Cl. Sodium chloride is the salt most responsible for the salinity of seawater and of the extracellular fluid of many multicellular organisms. In its edible form, salt (also known as ''table salt'') is commonly used as a condiment and food preservative. Large quantities of sodium chloride are used in many industrial processes, and it is a major source of sodium and chlorine compounds used as feedstocks for further chemical syntheses. Another major application of sodium chloride is de-icing of roadways in sub-freezing weather. Uses In addition to the familiar domestic uses of salt, more dominant applications of the approximately 250 million tonnes per year production (2008 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Spintronic
Spintronics (a portmanteau meaning spin transport electronics), also known as spin electronics, is the study of the intrinsic spin of the electron and its associated magnetic moment, in addition to its fundamental electronic charge, in solid-state devices. The field of spintronics concerns spin-charge coupling in metallic systems; the analogous effects in insulators fall into the field of multiferroics. Spintronics fundamentally differs from traditional electronics in that, in addition to charge state, electron spins are exploited as a further degree of freedom, with implications in the efficiency of data storage and transfer. Spintronic systems are most often realised in dilute magnetic semiconductors (DMS) and Heusler alloys and are of particular interest in the field of quantum computing and neuromorphic computing. History Spintronics emerged from discoveries in the 1980s concerning spin-dependent electron transport phenomena in solid-state devices. This includes the observa ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cobalt Blue
Cobalt blue is a blue pigment made by sintering cobalt(II) oxide with aluminum(III) oxide (alumina) at 1200 °C. Chemically, cobalt blue pigment is cobalt(II) oxide-aluminium oxide, or cobalt(II) aluminate, CoAl2O4. Cobalt blue is lighter and less intense than the (iron-cyanide based) pigment Prussian blue. It is extremely stable and historically has been used as a coloring agent in ceramics (especially Chinese porcelain), jewelry, and paint. Transparent glasses are tinted with the silica-based cobalt pigment smalt. Historical uses and production Cobalt blue in impure forms had long been used in Chinese porcelain. The first recorded use of ''cobalt blue'' as a color name in English was in 1777. It was independently discovered as a pure alumina-based pigment by Louis Jacques Thénard in 1802. Commercial production began in France in 1807. The leading world manufacturer of cobalt blue in the nineteenth century was Benjamin Wegner's Norwegian company Blaafarveværket ("blue c ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
List Of Inorganic Pigments
The following list includes commercially or artistically important inorganic pigments of natural and synthetic origin.. Purple pigments Aluminum pigments * Ultramarine violet: (PV15) - a synthetic or naturally occurring sulfur containing silicate mineral. Copper pigments *Han purple: BaCuSi2O6. Cobalt pigments *Cobalt violet: (PV14) Co3(PO4)2. Manganese pigments *Manganese violet: NH4MnP2O7 (PV16) manganic ammonium pyrophosphate. Gold pigments *Purple of Cassius: Gold nanoparticles suspended in tin dioxide - Aux • SnO2. Blue pigments Aluminum pigments *Ultramarine (PB29): a synthetic or naturally occurring sulfur containing silicate mineral - (generalized formula) *Persian blue: made by grinding up the mineral Lapis lazuli. The most important mineral component of lapis lazuli is lazurite (25% to 40%), a feldspathoid silicate mineral with the formula . Cobalt pigments *Cobalt blue (PB28): cobalt(II) aluminate. * Cerulean blue (PB35): cobalt(II) stannate. *Cerium ura ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Inorganic Pigments
In chemistry, an inorganic compound is typically a chemical compound that lacks carbon–hydrogen bonds, that is, a compound that is not an organic compound. The study of inorganic compounds is a subfield of chemistry known as '' inorganic chemistry''. Inorganic compounds comprise most of the Earth's crust, although the compositions of the deep mantle remain active areas of investigation. Some simple carbon compounds are often considered inorganic. Examples include the allotropes of carbon (graphite, diamond, buckminsterfullerene, etc.), carbon monoxide, carbon dioxide, carbides, and the following salts of inorganic anions: carbonates, cyanides, cyanates, and thiocyanates. Many of these are normal parts of mostly organic systems, including organisms; describing a chemical as inorganic does not necessarily mean that it does not occur within living things. History Friedrich Wöhler's conversion of ammonium cyanate into urea in 1828 is often cited as the starting point of modern ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cobalt Compounds
Cobalt compounds are chemical compounds formed by cobalt with other elements. In the compound, the most stable oxidation state of cobalt is the +2 oxidation state, and in the presence of specific ligands, there are also stable compounds with +3 valence. In addition, there are cobalt compounds in high oxidation states +4, +5 and low oxidation states -1, 0, +1. Inorganic compounds Halides Four halides of cobalt(II) are known, which are cobalt(II) fluoride (CoF2) which is a pink solid, cobalt(II) chloride (CoCl2) which is a blue solid, cobalt(II) bromide (CoBr2) which is a green solid, and cobalt(II) iodide (CoI2) which is a blue-black solid. In addition to the anhydrous forms, these cobalt halides also have hydrates. Anhydrous cobalt(II) chloride is blue, while the hexahydrate is red-purple. Because the color change of cobalt(II) chloride in different hydrates, it can be used to manufacture color-changing silica gel. Anhydrous cobalt halides react with nitric oxide at 70~120 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |





-chloride-hexahydrate-sample.jpg)