|
Carboxylic Acid Reduction
In organic chemistry, carbonyl reduction is the organic reduction of any carbonyl group by a reducing agent. Typical carbonyl compounds are ketones, aldehydes, carboxylic acids, esters, and acid halides. Carboxylic acids, esters, and acid halides can be reduced to either aldehydes or a step further to primary alcohols, depending on the strength of the reducing agent; aldehydes and ketones can be reduced respectively to primary and secondary alcohols. In deoxygenation, the alcohol can be further reduced and removed altogether. Metal hydrides based on boron and aluminum are common reducing agents; catalytic hydrogenation is also an important method of reducing carbonyls. Before the discovery of soluble hydride reagents, esters were reduced by the Bouveault–Blanc reduction, employing a mixture of sodium metal in the presence of alcohols. Carboxylic acid derivatives, aldehydes, and ketones to alcohols Hydride reduction Mechanism The reaction mechanism for metal hydride reduc ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Redox Ladder
A redox gradient is a series of reduction-oxidation (redox) reactions sorted according to redox potential. The redox ladder displays the order in which redox reactions occur based on the free energy gained from redox pairs. These redox gradients form both spatially and temporally as a result of differences in microbial processes, chemical composition of the environment, and oxidative potential. Common environments where redox gradients exist are coastal marshes, lakes, contaminant plumes, and soils. The Earth has a global redox gradient with an oxidizing environment at the surface and increasingly reducing conditions below the surface. Redox gradients are generally understood at the macro level, but characterization of redox reactions in heterogeneous environments at the micro-scale require further research and more sophisticated measurement techniques. Measuring Redox Conditions Redox conditions are measured according to the redox potential (Eh) in volts, which represents th ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Bull
A bull is an intact (i.e., not castrated) adult male of the species ''Bos taurus'' (cattle). More muscular and aggressive than the females of the same species (i.e., cows), bulls have long been an important symbol in many religions, including for sacrifices. These animals play a significant role in beef ranching, dairy farming, and a variety of sporting and cultural activities, including bullfighting and bull riding. Due to their temperament, handling requires precautions. Nomenclature The female counterpart to a bull is a cow, while a male of the species that has been castrated is a ''steer'', '' ox'', or ''bullock'', although in North America, this last term refers to a young bull. Use of these terms varies considerably with area and dialect. Colloquially, people unfamiliar with cattle may refer to both castrated and intact animals as "bulls". A wild, young, unmarked bull is known as a ''micky'' in Australia.Sheena Coupe (ed.), ''Frontier Country, Vol. 1'' (Weldon Ru ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Lithium Triethylborohydride
Lithium triethylborohydride is the organoboron compound with the formula Li Et3 BH. Commonly referred to as LiTEBH or Superhydride, it is a powerful reducing agent used in organometallic and organic chemistry. It is a colorless or white liquid but is typically marketed and used as a THF solution. The related reducing agent sodium triethylborohydride is commercially available as toluene solutions. LiBHEt3 is a stronger reducing agent than lithium borohydride and lithium aluminium hydride. Preparation LiBHEt3 is prepared by the reaction of lithium hydride (LiH) and triethylborane (Et3B) in tetrahydrofuran (THF): :LiH + Et3B → LiEt3BH Its THF solutions are stable indefinitely in the absence of moisture and air. Reactions Alkyl halides are reduced to the alkanes by LiBHEt3. LiBHEt3 reduces a wide range of functional groups, but so do many other hydride reagents. Instead, LiBHEt3 is reserved for difficult substrates, such as sterically hindered carbonyls, as illustrated by redu ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nitrile
In organic chemistry, a nitrile is any organic compound that has a functional group. The prefix '' cyano-'' is used interchangeably with the term ''nitrile'' in industrial literature. Nitriles are found in many useful compounds, including methyl cyanoacrylate, used in super glue, and nitrile rubber, a nitrile-containing polymer used in latex-free laboratory and medical gloves. Nitrile rubber is also widely used as automotive and other seals since it is resistant to fuels and oils. Organic compounds containing multiple nitrile groups are known as cyanocarbons. Inorganic compounds containing the group are not called nitriles, but cyanides instead. Though both nitriles and cyanides can be derived from cyanide salts, most nitriles are not nearly as toxic. Structure and basic properties The N−C−C geometry is linear in nitriles, reflecting the sp hybridization of the triply bonded carbon. The C−N distance is short at 1.16 Å, consistent with a triple bond. Nitr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Acetoxy Group
In organic chemistry, the acetoxy group (abbr. AcO or OAc; IUPAC name: acetyloxy), is a functional group with the formula and the structure . As the ''-oxy'' suffix implies, it differs from the acetyl group () by the presence of an additional oxygen atom. The name acetoxy is the short form of ''acetyl-oxy''. Functionality An acetoxy group may be used as a protection for an alcohol functionality in a synthetic route although the protecting group itself is called an acetyl group. Alcohol protection There are several options of introducing an acetoxy functionality in a molecule from an alcohol (in effect protecting the alcohol by acetylation): * Acetyl halide, such as acetyl chloride in the presence of a base like triethylamine * Activated ester form of acetic acid, such as a N-hydroxysuccinimide ester, although this is not advisable due to higher costs and difficulties. * Acetic anhydride in the presence of base with a catalyst such as pyridine with a bit of DMAP added. An a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sodium Triacetoxyborohydride
Sodium triacetoxyborohydride, also known as sodium triacetoxyhydroborate, commonly abbreviated STAB, is a chemical compound with the formula Na(CH3COO)3BH. Like other borohydrides, it is used as a reducing agent in organic synthesis. This colourless salt is prepared by protonolysis of sodium borohydride with acetic acid: :NaBH4 + 3 HO2CCH3 → NaBH(O2CCH3)3 + 3 H2 Comparison with related reagents Sodium triacetoxyborohydride is a milder reducing agent than sodium borohydride or even sodium cyanoborohydride. It reduces aldehydes but not most ketones. It is especially suitable for reductive aminations of aldehydes and ketones. However, unlike sodium cyanoborohydride, the triacetoxyborohydride hydrolyzes readily, nor is it compatible with methanol. It reacts only slowly with ethanol and isopropanol and can be used with these. NaBH(OAc)3 may also be used for reductive alkylation of secondary amines with aldehyde-bisulfite adducts. Monoacetoxyborohydride The combinatio ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
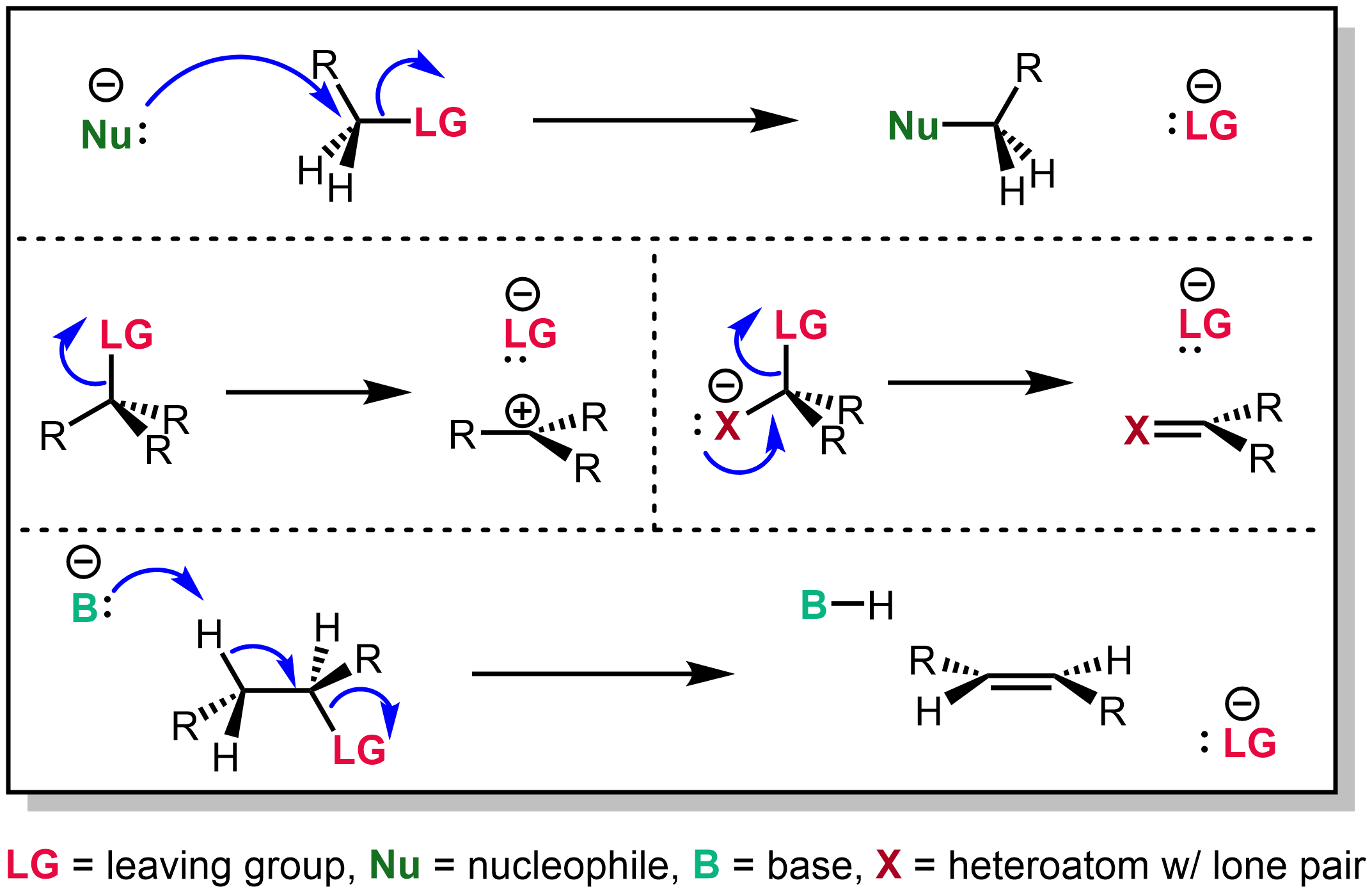
Leaving Group
In chemistry, a leaving group is defined by the IUPAC as an atom or group of atoms that detaches from the main or residual part of a substrate during a reaction or elementary step of a reaction. However, in common usage, the term is often limited to a fragment that departs with a pair of electrons in heterolytic bond cleavage. In this usage, a leaving group is a less formal but more commonly used synonym of the term '' nucleofuge''. In this context, leaving groups are generally anions or neutral species, departing from a neutral or cationic substrates, respectively, though in rare cases, cations leaving from a dicationic substrate are also known. A species' ability to serve as a leaving group depends on its ability to stabilize the additional electron density that results from bond heterolysis. Common anionic leaving groups are halides such as Cl−, Br−, and I−, and sulfonate esters such as tosylate (TsO−), while water (H2O), alcohols (HOR), and amines (R3N) are common neu ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electron Donor
In chemistry, an electron donor is a chemical entity that donates electrons to another compound. It is a reducing agent that, by virtue of its donating electrons, is itself oxidized in the process. Typical reducing agents undergo permanent chemical alteration through covalent or ionic reaction chemistry. This results in the complete and irreversible transfer of one or more electrons. In many chemical circumstances, however, the transfer of electronic charge to an electron acceptor may be only fractional, meaning an electron is not completely transferred, but results in an electron resonance between the donor and acceptor. This leads to the formation of charge transfer complexes in which the components largely retain their chemical identities. The electron donating power of a donor molecule is measured by its ionization potential which is the energy required to remove an electron from the highest occupied molecular orbital (HOMO). The overall energy balance (ΔE), i.e., energy ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Carbonyl Resonance
In organic chemistry, a carbonyl group is a functional group composed of a carbon atom double-bonded to an oxygen atom: C=O. It is common to several classes of organic compounds, as part of many larger functional groups. A compound containing a carbonyl group is often referred to as a carbonyl compound. The term carbonyl can also refer to carbon monoxide as a ligand in an inorganic or organometallic complex (a metal carbonyl, e.g. nickel carbonyl). The remainder of this article concerns itself with the organic chemistry definition of carbonyl, where carbon and oxygen share a double bond. Carbonyl compounds In organic chemistry, a carbonyl group characterizes the following types of compounds: Other organic carbonyls are urea and the carbamates, the derivatives of acyl chlorides chloroformates and phosgene, carbonate esters, thioesters, lactones, lactams, hydroxamates, and isocyanates. Examples of inorganic carbonyl compounds are carbon dioxide and carbonyl sulfide. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Aldehyde Reduction
In organic chemistry, carbonyl reduction is the organic reduction of any carbonyl group by a reducing agent. Typical carbonyl compounds are ketones, aldehydes, carboxylic acids, esters, and acid halides. Carboxylic acids, esters, and acid halides can be reduced to either aldehydes or a step further to primary alcohols, depending on the strength of the reducing agent; aldehydes and ketones can be reduced respectively to primary and secondary alcohols. In deoxygenation, the alcohol can be further reduced and removed altogether. Metal hydrides based on boron and aluminum are common reducing agents; catalytic hydrogenation is also an important method of reducing carbonyls. Before the discovery of soluble hydride reagents, esters were reduced by the Bouveault–Blanc reduction, employing a mixture of sodium metal in the presence of alcohols. Carboxylic acid derivatives, aldehydes, and ketones to alcohols Hydride reduction Mechanism The reaction mechanism for metal hydride reduc ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ester Reduction
In chemistry, an ester is a compound derived from an oxoacid (organic or inorganic) in which at least one hydroxyl group () is replaced by an alkoxy group (), as in the substitution reaction of a carboxylic acid and an alcohol. Glycerides are fatty acid esters of glycerol; they are important in biology, being one of the main classes of lipids and comprising the bulk of animal fats and vegetable oils. Esters typically have a pleasant smell; those of low molecular weight are commonly used as fragrances and are found in essential oils and pheromones. They perform as high-grade solvents for a broad array of plastics, plasticizers, resins, and lacquers, and are one of the largest classes of synthetic lubricants on the commercial market. Polyesters are important plastics, with monomers linked by ester moieties. Phosphoesters form the backbone of DNA molecules. Nitrate esters, such as nitroglycerin, are known for their explosive properties. '' Nomenclature Etymology Th ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electrophile
In chemistry, an electrophile is a chemical species that forms bonds with nucleophiles by accepting an electron pair. Because electrophiles accept electrons, they are Lewis acids. Most electrophiles are positively charged, have an atom that carries a partial positive charge, or have an atom that does not have an octet of electrons. Electrophiles mainly interact with nucleophiles through addition and substitution reactions. Frequently seen electrophiles in organic syntheses include cations such as H+ and NO+, polarized neutral molecules such as HCl, alkyl halides, acyl halides, and carbonyl compounds, polarizable neutral molecules such as Cl2 and Br2, oxidizing agents such as organic peracids, chemical species that do not satisfy the octet rule such as carbenes and radicals, and some Lewis acids such as BH3 and DIBAL. Organic chemistry Addition of halogens These occur between alkenes and electrophiles, often halogens as in halogen addition reactions. Common reactions ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |

.png)