|
Bath Salts
Bath salts are water-soluble, pulverized minerals that are added to water to be used for bathing. They are said to improve cleaning, enhance the enjoyment of bathing, and serve as a vehicle for cosmetic agents. Bath salts have been developed which mimic the properties of natural mineral baths or hot springs. Some bath salts contain glycerine so the product will act as an emollient, humectant, or lubricant. Fragrances and colors are often added to bath salts; the fragrances are used to increase the users' enjoyment of the bathing experience. Description Substances often labeled as bath salts include magnesium sulfate (Epsom salts), sodium chloride (table salt), sodium bicarbonate (baking soda), sodium hexametaphosphate (Calgon, amorphous/glassy sodium metaphosphate), sodium sesquicarbonate, borax, and sodium citrate. Glycerin, or liquid glycerin, is another common ingredient in bath salts. Depending on their properties, the additives can be classified as emollient, humect ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Diluent
A diluent (also referred to as a filler, dilutant or thinner) is a Concentration, diluting agent. Certain fluids are too Viscosity, viscous to be pumped easily or too density, dense to flow from one particular point to the other. This can be problematic, because it might not be economically feasible to transport such fluids in this state. To ease this restricted movement, diluents are added. This decreases the viscosity of the fluids, thereby also decreasing the pumping/transportation costs. One industrial application is the transport of crude oil via pipelines. Heavy crude oil/bitumen are fluids with high viscosity, especially at low temperatures. The addition of a diluent enables the diluted fluid (dilbit in the case of bitumen) to meet pipeline specifications in order for it to be efficiently transported. Typical diluent in this case is naphtha or Natural gas condensate, condensate. Types of diluents more familiar to the general public include paint thinner and nail polish ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Isolation Tank
An isolation tank, sensory deprivation tank, float tank, float pod, float cabin, flotation tank, or sensory attenuation tank is a water filled, pitch-black, light-proof, soundproof environment heated to the same temperature as the skin, developed in 1954 by John C. Lilly. Flotation tanks are widely advertised as a form of alternative medicine but beneficial health effects are unproven. Method The tank is filled with 10 inches of water which contains enough dissolved Epsom salt to create a specific gravity of approximately 1.25–1.26. This environment allows an individual to float effortlessly on the surface of the water. History The isolation tank was developed in 1954 by John C. Lilly, a medical practitioner and neuropsychiatrist. During his training in psychoanalysis at the US National Institute of Mental Health (NIMH), Lilly experimented with sensory deprivation. Widespread commercial interest and use of the isolation tank did not occur until 1972, when Glenn Perry, a com ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
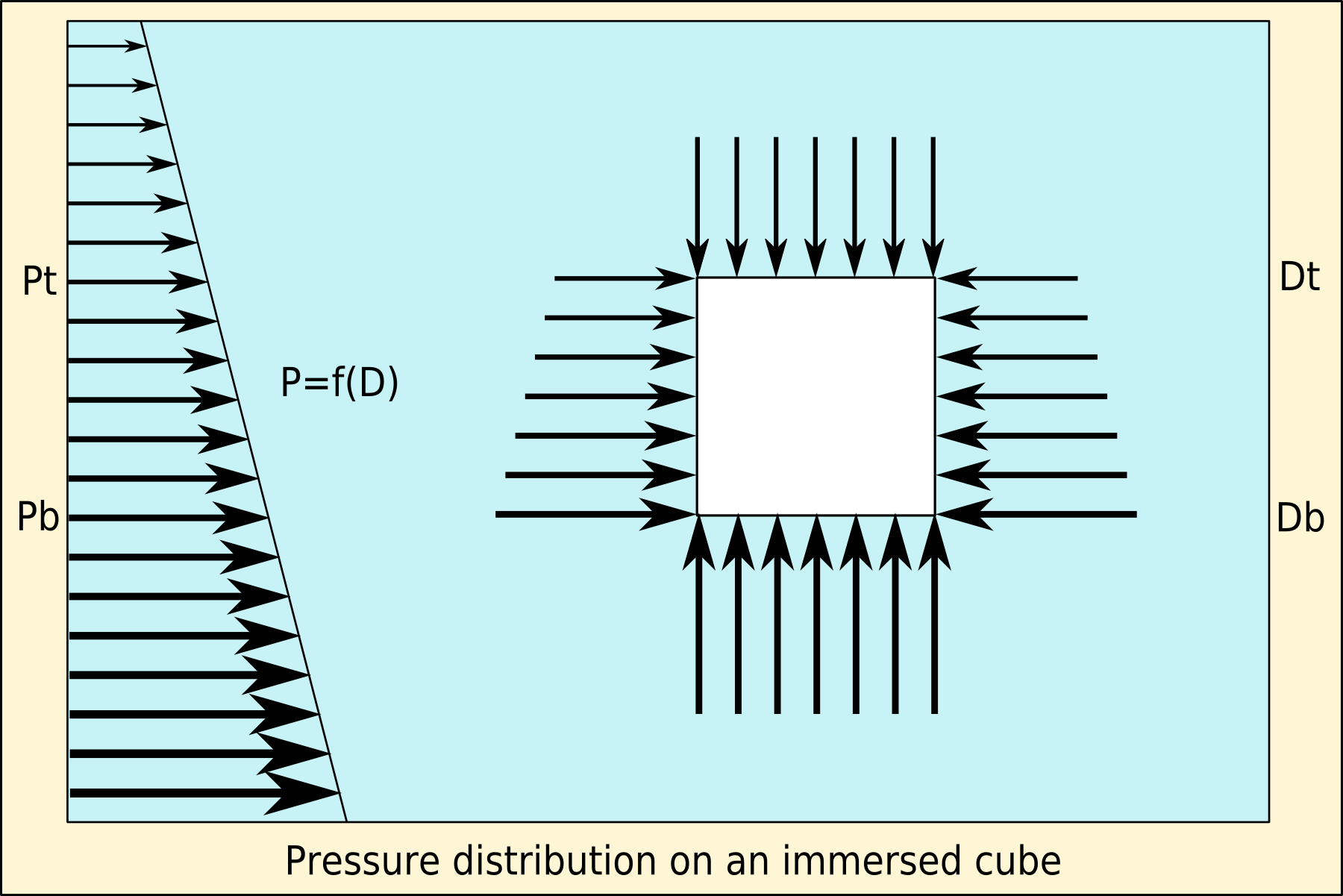
Buoyancy
Buoyancy (), or upthrust, is an upward force exerted by a fluid that opposes the weight of a partially or fully immersed object. In a column of fluid, pressure increases with depth as a result of the weight of the overlying fluid. Thus the pressure at the bottom of a column of fluid is greater than at the top of the column. Similarly, the pressure at the bottom of an object submerged in a fluid is greater than at the top of the object. The pressure difference results in a net upward force on the object. The magnitude of the force is proportional to the pressure difference, and (as explained by Archimedes' principle) is equivalent to the weight of the fluid that would otherwise occupy the submerged volume of the object, i.e. the displaced fluid. For this reason, an object whose average density is greater than that of the fluid in which it is submerged tends to sink. If the object is less dense than the liquid, the force can keep the object afloat. This can occur only in a no ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Density
Density (volumetric mass density or specific mass) is the substance's mass per unit of volume. The symbol most often used for density is ''ρ'' (the lower case Greek letter rho), although the Latin letter ''D'' can also be used. Mathematically, density is defined as mass divided by volume: : \rho = \frac where ''ρ'' is the density, ''m'' is the mass, and ''V'' is the volume. In some cases (for instance, in the United States oil and gas industry), density is loosely defined as its weight per unit volume, although this is scientifically inaccurate – this quantity is more specifically called specific weight. For a pure substance the density has the same numerical value as its mass concentration. Different materials usually have different densities, and density may be relevant to buoyancy, purity and packaging. Osmium and iridium are the densest known elements at standard conditions for temperature and pressure. To simplify comparisons of density across different s ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hard Water
Hard water is water that has high mineral content (in contrast with "soft water"). Hard water is formed when water percolates through deposits of limestone, chalk or gypsum, which are largely made up of calcium and magnesium carbonates, bicarbonates and sulfates. Hard drinking water may have moderate health benefits. It can pose critical problems in industrial settings, where water hardness is monitored to avoid costly breakdowns in boilers, cooling towers, and other equipment that handles water. In domestic settings, hard water is often indicated by a lack of foam formation when soap is agitated in water, and by the formation of limescale in kettles and water heaters.World Health OrganizatioHardness in Drinking-Water 2003 Wherever water hardness is a concern, water softening is commonly used to reduce hard water's adverse effects. Origins Natural rainwater, snow and other forms of precipitation typically have low concentrations of multivalent cations such as calcium and magnes ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Soft Water
Hard water is water that has high mineral content (in contrast with "soft water"). Hard water is formed when water percolates through deposits of limestone, chalk or gypsum, which are largely made up of calcium and magnesium carbonates, bicarbonates and sulfates. Hard drinking water may have moderate health benefits. It can pose critical problems in industrial settings, where water hardness is monitored to avoid costly breakdowns in boilers, cooling towers, and other equipment that handles water. In domestic settings, hard water is often indicated by a lack of foam formation when soap is agitated in water, and by the formation of limescale in kettles and water heaters.World Health OrganizatioHardness in Drinking-Water 2003 Wherever water hardness is a concern, water softening is commonly used to reduce hard water's adverse effects. Origins Natural rainwater, snow and other forms of precipitation typically have low concentrations of multivalent cations such as calcium and magnes ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Soap
Soap is a salt of a fatty acid used in a variety of cleansing and lubricating products. In a domestic setting, soaps are surfactants usually used for washing, bathing, and other types of housekeeping. In industrial settings, soaps are used as thickeners, components of some lubricants, and precursors to catalysts. When used for cleaning, soap solubilizes particles and grime, which can then be separated from the article being cleaned. In hand washing, as a surfactant, when lathered with a little water, soap kills microorganisms by disorganizing their membrane lipid bilayer and denaturing their proteins. It also emulsifies oils, enabling them to be carried away by running water. Soap is created by mixing fats and oils with a base. A similar process is used for making detergent which is also created by combining chemical compounds in a mixer. Humans have used soap for millennia. Evidence exists for the production of soap-like materials in ancient Babylon around 2800 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
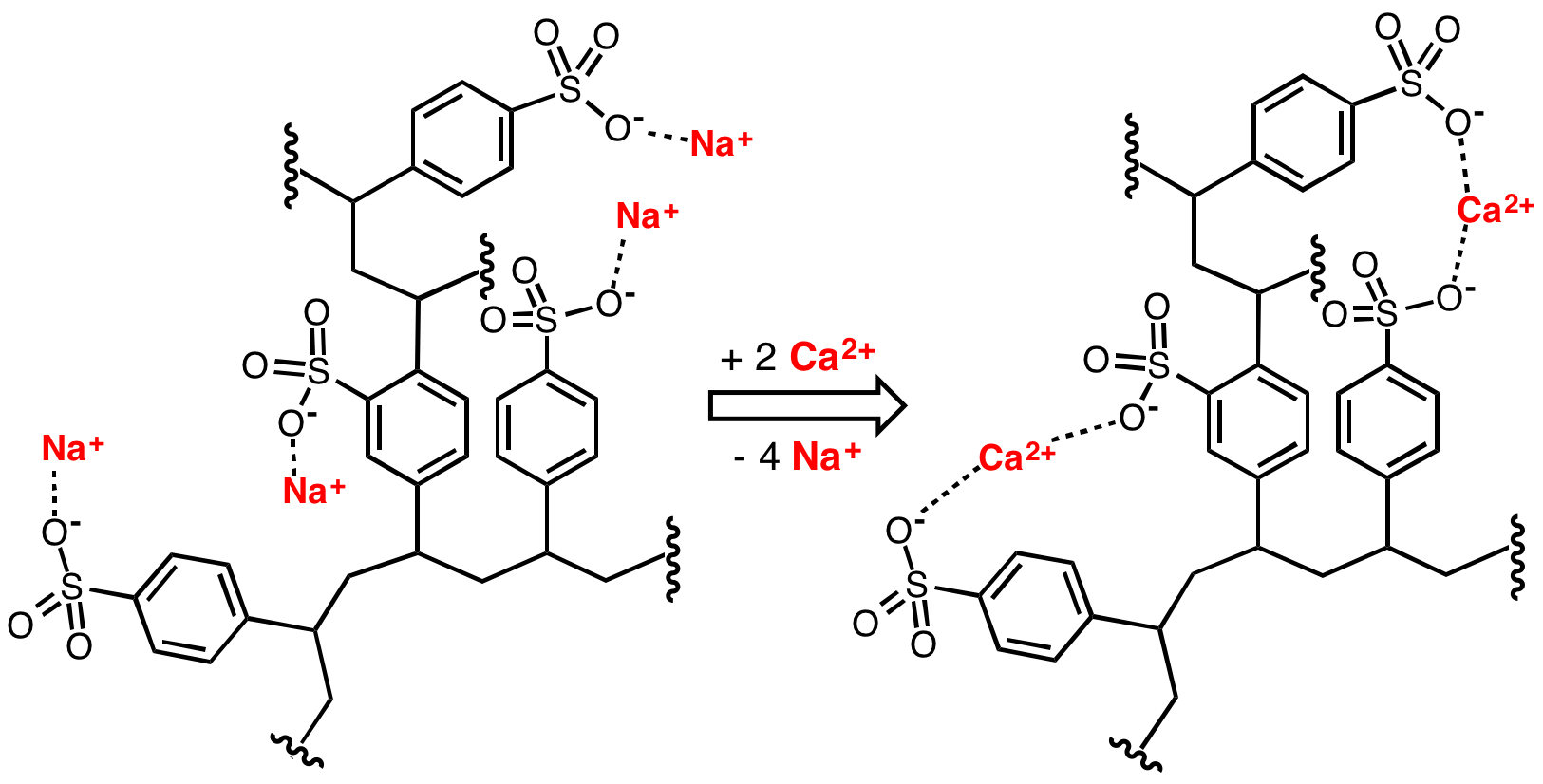
Water Softeners
Water softening is the removal of calcium, magnesium, and certain other metal cations in hard water. The resulting soft water requires less soap for the same cleaning effort, as soap is not wasted bonding with calcium ions. Soft water also extends the lifetime of plumbing by reducing or eliminating scale build-up in pipes and fittings. Water softening is usually achieved using lime softening or ion-exchange resins but is increasingly being accomplished using nanofiltration or reverse osmosis membranes. Rationale The presence of certain metal ions like calcium and magnesium, principally as bicarbonates, chlorides, and sulfates, in water causes a variety of problems. Hard water leads to the buildup of limescale, which can foul plumbing, and promote galvanic corrosion. In industrial scale water softening plants, the effluent flow from the re-generation process can precipitate scale that can interfere with sewage systems. The slippery feeling associated with washing in soft water ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Exfoliation (cosmetology)
Exfoliation is the removal of dead skin cells and built-up dirt from the skin's surface. The term comes from the Latin word ''exfoliare'' (to strip off leaves). This is a regular practice within the cosmetic industry, both for its outcome of promoting skin regeneration as well as providing a deep cleanse of the skin barrier. Being used in facials, this process can be achieved by mechanical or chemical means, such as microdermabrasion or chemical peels. Exfoliants are advertised as treatments that enhance beauty and promote a youthful and healthy appearance. History Exfoliation was first practiced among the ancient Egyptians. This was also used in Asia, specifically in China, during the Qing Dynasty (1644–1944). Mechanical Exfoliation Mechanical exfoliation methods involve physically scrubbing the skin with an abrasive material. These types of exfoliants include microfiber cloths, adhesive exfoliation sheets, micro-bead facial scrubs, crêpe paper, crushed apricot kernel or a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phosphates
In chemistry, a phosphate is an anion, salt, functional group or ester derived from a phosphoric acid. It most commonly means orthophosphate, a derivative of orthophosphoric acid . The phosphate or orthophosphate ion is derived from phosphoric acid by the removal of three protons . Removal of one or two protons gives the dihydrogen phosphate ion and the hydrogen phosphate ion ion, respectively. These names are also used for salts of those anions, such as ammonium dihydrogen phosphate and trisodium phosphate. File:3-phosphoric-acid-3D-balls.png, Phosphoricacid File:2-dihydrogenphosphate-3D-balls.png, Dihydrogenphosphate File:1-hydrogenphosphate-3D-balls.png, Hydrogenphosphate File:0-phosphate-3D-balls.png, Phosphate In organic chemistry, phosphate or orthophosphate is an organophosphate, an ester of orthophosphoric acid of the form where one or more hydrogen atoms are replaced by organic groups. An example is trimethyl phosphate, . The term also refers to the tri ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Seashell Bath Salt Tablet
A seashell or sea shell, also known simply as a shell, is a hard, protective outer layer usually created by an animal or organism that lives in the sea. The shell is part of the body of the animal. Empty seashells are often found washed up on beaches by beachcombers. The shells are empty because the animal has died and the soft parts have decomposed or been eaten by another animal. A seashell is usually the exoskeleton of an invertebrate (an animal without a backbone), and is typically composed of calcium carbonate or chitin. Most shells that are found on beaches are the shells of marine mollusks, partly because these shells are usually made of calcium carbonate, and endure better than shells made of chitin. Apart from mollusk shells, other shells that can be found on beaches are those of barnacles, horseshoe crabs and brachiopods. Marine annelid worms in the family Serpulidae create shells which are tubes made of calcium carbonate cemented onto other surfaces. Th ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |