|
Bromobenzene
Bromobenzene is an aryl halide, C6H5Br. It is a colourless liquid although older samples can appear yellow. It is a reagent in organic synthesis. Synthesis and reactions Bromobenzene is prepared by the action of bromine on benzene in the presence of Lewis acid catalysts such as aluminium chloride or ferric bromide. Bromobenzene is used to introduce a phenyl group into other compounds. One method involves its conversion to the Grignard reagent, phenylmagnesium bromide. This reagent can be used, e.g. in the reaction with carbon dioxide to prepare benzoic acid. Other methods involve palladium-catalyzed coupling reactions, such as the Suzuki reaction. Bromobenzene is used as a precursor in the manufacture of phencyclidine. Toxicity Animal tests indicate low toxicity. Little is known about chronic effects. For liver toxicity, the 3,4-epoxide are proposed intermediates. See also *Fluorobenzene *Chlorobenzene *Iodobenzene Iodobenzene is an organoiodine compound consisting ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Fluorobenzene
Fluorobenzene is the chemical compound with the formula C6H5F, often abbreviated PhF. A colorless liquid, it is a precursor to many fluorophenyl compounds. Preparation PhF was first reported in 1886 by O. Wallach at the University of Bonn, who prepared the compound in two steps. Phenyldiazonium chloride was first converted to a triazene using piperidine: : hN2l + 2 (CH2)5NH → PhN=N-N(CH2)5 + CH2)5NH2l The triazine was then cleaved with hydrofluoric acid: :PhN=N-N(CH2)5 + 2 HF → PhF + N2 + CH2)5NH2 Historical note: in Wallach's era, the element fluorine was symbolized with "Fl". Thus, his procedure is subtitled "Fluorbenzol, C6H5Fl". On the laboratory scale, PhF is prepared by the thermal decomposition of the benzenediazonium tetrafluoroborate: :PhN2BF4 → PhF + BF3 + N2 According to the procedure, solid hN2F4 is heated with a flame to initiate an exothermic reaction, which also affords boron trifluoride and nitrogen gas. Product PhF and BF3 are ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chlorobenzene
Chlorobenzene is an aromatic organic compound with the chemical formula C6H5Cl. This colorless, flammable liquid is a common solvent and a widely used intermediate in the manufacture of other chemicals. Uses Historical The major use of chlorobenzene is as an intermediate in the production of herbicides, dyestuffs, and rubber. Chlorobenzene is also used as a high-boiling solvent in industrial applications as well as in the laboratory. Chlorobenzene is nitrated on a large scale to give a mixture of 2-nitrochlorobenzene and 4-nitrochlorobenzene, which are separated. These mononitrochlorobenzenes are converted to related 2-nitrophenol, 2-nitroanisole, bis(2-nitrophenyl)disulfide, and 2-nitroaniline by nucleophilic displacement of the chloride, with respectively sodium hydroxide, sodium methoxide, sodium disulfide, and ammonia. The conversions of the 4-nitro derivative are similar. Chlorobenzene once was used in the manufacture of pesticides, most notably DDT, by reaction with ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Benzoic Acid
Benzoic acid is a white (or colorless) solid organic compound with the formula , whose structure consists of a benzene ring () with a carboxyl () substituent. It is the simplest aromatic carboxylic acid. The name is derived from gum benzoin, which was for a long time its only source. Benzoic acid occurs naturally in many plants and serves as an intermediate in the biosynthesis of many secondary metabolites. Salts of benzoic acid are used as food preservatives. Benzoic acid is an important precursor for the industrial synthesis of many other organic substances. The salts and esters of benzoic acid are known as benzoates . History Benzoic acid was discovered in the sixteenth century. The dry distillation of gum benzoin was first described by Nostradamus (1556), and then by Alexius Pedemontanus (1560) and Blaise de Vigenère (1596). Justus von Liebig and Friedrich Wöhler determined the composition of benzoic acid. These latter also investigated how hippuric acid is related ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Iodobenzene
Iodobenzene is an organoiodine compound consisting of a benzene ring substituted with one iodine atom. It is useful as a synthetic intermediate in organic chemistry. It is a volatile colorless liquid, although aged samples appear yellowish. Preparation Iodobenzene is commercially available, but it can be prepared in the laboratory from aniline via the Diazotization Reaction. In the first step, the amine functional group is diazotized with hydrochloric acid and sodium nitrite. Potassium iodide is added to the resultant phenyldiazonium chloride, causing nitrogen gas to evolve. The product is separated by steam distillation. : Alternatively, it can be produced by refluxing iodine and nitric acid with benzene. Reactions Since the C–I bond is weaker than C–Br or C–Cl, iodobenzene is more reactive than bromobenzene or chlorobenzene. Iodobenzene reacts readily with magnesium to form the Grignard reagent, phenylmagnesium iodide. Phenylmagnesium iodide, like the bromide analog ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Royal Society Of Chemistry
The Royal Society of Chemistry (RSC) is a learned society (professional association) in the United Kingdom with the goal of "advancing the chemistry, chemical sciences". It was formed in 1980 from the amalgamation of the Chemical Society, the Royal Institute of Chemistry, the Faraday Society, and the Society for Analytical Chemistry with a new Royal Charter and the dual role of learned society and professional body. At its inception, the Society had a combined membership of 34,000 in the UK and a further 8,000 abroad. The headquarters of the Society are at Burlington House, Piccadilly, London. It also has offices in Thomas Graham House in Cambridge (named after Thomas Graham (chemist), Thomas Graham, the first president of the Chemical Society) where ''RSC Publishing'' is based. The Society has offices in the United States, on the campuses of The University of Pennsylvania and Drexel University, at the University City Science Center in Philadelphia, Pennsylvania, in both Beijing a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ferric Bromide
Iron(III) bromide is the chemical compound with the formula FeBr3. Also known as ferric bromide, this red-brown odorless compound is used as a Lewis acid catalyst in the halogenation of aromatic compounds. It dissolves in water to give acidic solutions. Structure, synthesis and basic properties FeBr3 forms a polymeric structure featuring six-coordinate, octahedral Fe centers.Holleman, A. F.; Wiberg, E. "Inorganic Chemistry" Academic Press: San Diego, 2001. . Although inexpensively available commercially, FeBr3 can be prepared by treatment of iron metal with bromine: :2 Fe + 3 Br2 → 2 FeBr3 Above 200 °C, FeBr3 decomposes to ferrous bromide: :2FeBr3 → 2FeBr2 + Br2 Iron(III) chloride is considerably more stable, reflecting the greater oxidizing power of chlorine. FeI3 is not stable, as iron(III) will oxidize iodide ions. Uses Ferric bromide is occasionally used as an oxidant in organic chemistry, e.g. for the conversion of alcohols to ketones. It is used as ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phencyclidine
Phencyclidine or phenylcyclohexyl piperidine (PCP), also known as angel dust among other names, is a dissociative anesthetic mainly used recreationally for its significant mind-altering effects. PCP may cause hallucinations, distorted perceptions of sounds, and violent behavior. As a recreational drug, it is typically smoked, but may be taken by mouth, snorted, or injected. It may also be mixed with cannabis or tobacco. Adverse effects may include seizures, coma, addiction, and an increased risk of suicide. Flashbacks may occur despite stopping usage. Chemically, PCP is a member of the arylcyclohexylamine class, and pharmacologically, it is a dissociative anesthetic. PCP works primarily as an NMDA receptor antagonist. PCP is most commonly used in the United States. While usage peaked in the US in the 1970s, between 2005 and 2011 an increase in visits to emergency departments as a result of the drug occurred. As of 2017 in the United States, about 1% of people in Twelfth ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Suzuki Reaction
The Suzuki reaction is an organic reaction, classified as a cross-coupling reaction, where the coupling partners are a boronic acid and an organohalide and the catalyst is a palladium(0) complex. It was first published in 1979 by Akira Suzuki, and he shared the 2010 Nobel Prize in Chemistry with Richard F. Heck and Ei-ichi Negishi for their contribution to the discovery and development of palladium-catalyzed cross-couplings in organic synthesis. This reaction is also known as the Suzuki–Miyaura reaction or simply as the Suzuki coupling. It is widely used to synthesize poly olefins, styrenes, and substituted biphenyls. Several reviews have been published describing advancements and the development of the Suzuki reaction. The general scheme for the Suzuki reaction is shown below, where a carbon-carbon single bond is formed by coupling a halide (R1-X) with an organoboron species (R2-BY2) using a palladium catalyst and a base. Reaction mechanism The mechanism of the Suzuki r ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
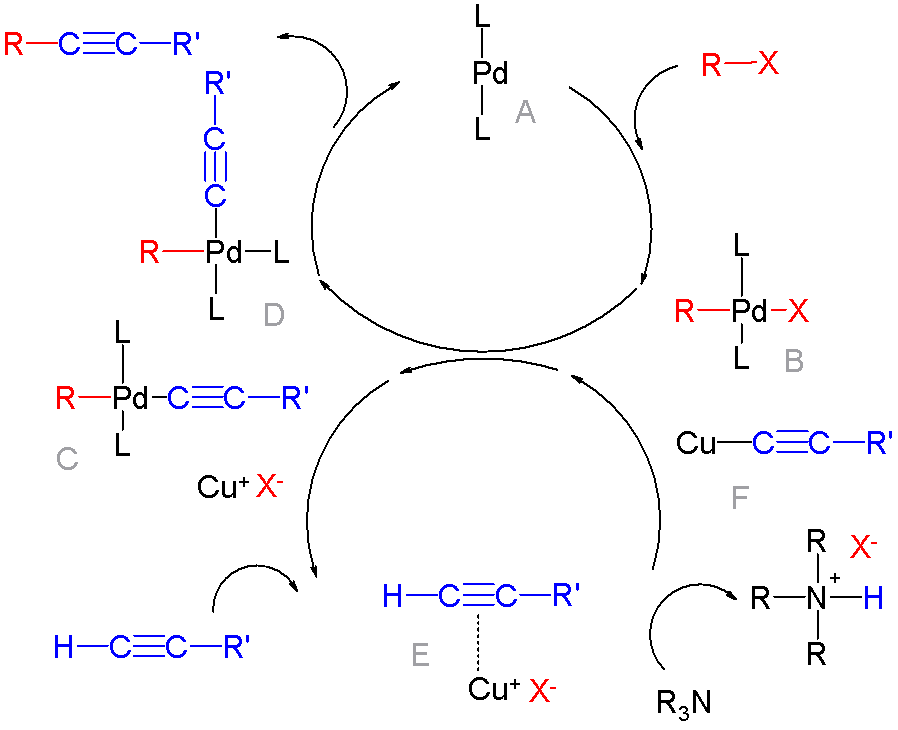
Palladium-catalyzed Coupling Reactions
In organic chemistry, a cross-coupling reaction is a reaction where two fragments are joined together with the aid of a metal catalyst. In one important reaction type, a main group organometallic compound of the type R-M (R = organic fragment, M = main group center) reacts with an organic halide of the type R'-X with formation of a new carbon–carbon bond in the product R-R'. Cross-coupling reaction are a subset of coupling reactions. It is often used in arylations. Richard F. Heck, Ei-ichi Negishi, and Akira Suzuki were awarded the 2010 Nobel Prize in Chemistry for developing palladium-catalyzed coupling reactions. Mechanism The mechanism generally involves reductive elimination of the organic substituents R and R' on a metal complex of the type LnMR(R') (where L is some arbitrary spectator ligand). The crucial intermediate LnMR(R') is formed in a two step process from a low valence precursor Ln. The oxidative addition of an organic halide (RX) to LnM gives LnMR(X). Subseq ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Carbon Dioxide
Carbon dioxide (chemical formula ) is a chemical compound made up of molecules that each have one carbon atom covalently double bonded to two oxygen atoms. It is found in the gas state at room temperature. In the air, carbon dioxide is transparent to visible light but absorbs infrared radiation, acting as a greenhouse gas. It is a trace gas in Earth's atmosphere at 421 parts per million (ppm), or about 0.04% by volume (as of May 2022), having risen from pre-industrial levels of 280 ppm. Burning fossil fuels is the primary cause of these increased CO2 concentrations and also the primary cause of climate change.IPCC (2022Summary for policy makersiClimate Change 2022: Mitigation of Climate Change. Contribution of Working Group III to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change Cambridge University Press, Cambridge, United Kingdom and New York, NY, USA Carbon dioxide is soluble in water and is found in groundwater, lakes, ice caps, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phenylmagnesium Bromide
Phenylmagnesium bromide, with the simplified formula , is a magnesium-containing organometallic compound. It is commercially available as a solution in diethyl ether or tetrahydrofuran (THF). Phenylmagnesium bromide is a Grignard reagent. It is often used as a synthetic equivalent for the phenyl "Ph−" synthon. Preparation Phenylmagnesium bromide is commercially available as solutions of diethyl ether or THF. Laboratory preparation involves treating bromobenzene with magnesium metal, usually in the form of turnings. A small amount of iodine may be used to activate the magnesium to initiate the reaction. Coordinating solvents such as ether or THF, are required to solvate (complex) the magnesium(II) center. The solvent must be aprotic since alcohols and water contain an acidic proton and thus react with phenylmagnesium bromide to give benzene. Carbonyl-containing solvents, such as acetone and ethyl acetate, are also incompatible with the reagent. Structure Although phenylmagn ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Grignard Reaction
The Grignard reaction () is an organometallic chemical reaction in which alkyl, allyl, vinyl, or aryl-magnesium halides ( Grignard reagent) is added to a carbonyl group in an aldehyde or ketone. This reaction is important for the formation of carbon–carbon bonds. The reaction of an organic halide with magnesium is ''not'' a Grignard reaction, but provides a Grignard reagent. : Grignard reactions and reagents were discovered by and are named after the French chemist François Auguste Victor Grignard (University of Nancy, France), who published it in 1900 and was awarded the 1912 Nobel Prize in Chemistry for this work. Reaction mechanism Because carbon is more electronegative than magnesium, the carbon attached to magnesium functions as a nucleophile and attacks the electrophilic carbon atom that is present within the polar bond of a carbonyl group. The addition of the Grignard reagent to the carbonyl typically proceeds through a six-membered ring transition state. Based on ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |

.jpg)