|
Bioproduction
Bioproduction is the production of biologics-based therapeutic drugs including protein-based therapeutics, vaccines, gene therapies as well as cell therapies; drugs so complex they can only be made in living systems or indeed are a living system (cell therapies). In practice, ‘bioproduction’ has become loosely synonymous with ‘bioprocessing’ as a way to describe the manufacturing process using, cell culture, chromatography, formulation and related analytical testing for large molecule drugs, vaccines and cellular therapies. Many combinations of reactor types and culture modes are now available for use in bioproduction: e.g., pharming, rocking wave-agitated bag batch, stirred-tank or air-lift fed-batch, and hollow-fiber or spin-filter perfusion. No single production format is inherently superior; that determination depends on many manufacturing capabilities, requirements, and goals.Whitford, William G., {{cite web , url=http://www.iclickmedia.com/bioprocessors/4_4_Whitfor ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Biotechnology
Biotechnology is the integration of natural sciences and engineering sciences in order to achieve the application of organisms, cells, parts thereof and molecular analogues for products and services. The term ''biotechnology'' was first used by Károly Ereky in 1919, meaning the production of products from raw materials with the aid of living organisms. Definition The concept of biotechnology encompasses a wide range of procedures for modifying living organisms according to human purposes, going back to domestication of animals, cultivation of the plants, and "improvements" to these through breeding programs that employ artificial selection and hybridization. Modern usage also includes genetic engineering as well as cell and tissue culture technologies. The American Chemical Society defines biotechnology as the application of biological organisms, systems, or processes by various industries to learning about the science of life and the improvement of the value of materials ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Vaccines
A vaccine is a biological preparation that provides active acquired immunity The adaptive immune system, also known as the acquired immune system, is a subsystem of the immune system that is composed of specialized, systemic cells and processes that eliminate pathogens or prevent their growth. The acquired immune system ... to a particular infectious disease, infectious or cancer, malignant disease. The safety and effectiveness of vaccines has been widely studied and verified. A vaccine typically contains an agent that resembles a disease-causing microorganism and is often made from weakened or Antigen, killed forms of the microbe, its toxins, or one of its surface proteins. The agent stimulates the body's immune system to recognize the agent as a threat, destroy it, and to further recognize and destroy any of the microorganisms associated with that agent that it may encounter in the future. Vaccines can be prophylaxis, prophylact ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Gene Therapy
Gene therapy is a medical field which focuses on the genetic modification of cells to produce a therapeutic effect or the treatment of disease by repairing or reconstructing defective genetic material. The first attempt at modifying human DNA was performed in 1980, by Martin Cline, but the first successful nuclear gene transfer in humans, approved by the National Institutes of Health, was performed in May 1989. The first therapeutic use of gene transfer as well as the first direct insertion of human DNA into the nuclear genome was performed by French Anderson in a trial starting in September 1990. It is thought to be able to cure many genetic disorders or treat them over time. Between 1989 and December 2018, over 2,900 clinical trials were conducted, with more than half of them in phase I.Gene Therapy Cli ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cell Therapy
Cell therapy (also called cellular therapy, cell transplantation, or cytotherapy) is a therapy in which viable cells are injected, grafted or implanted into a patient in order to effectuate a medicinal effect, for example, by transplanting T-cells capable of fighting cancer cells via cell-mediated immunity in the course of immunotherapy, or grafting stem cells to regenerate diseased tissues. Cell therapy originated in the nineteenth century when scientists experimented by injecting animal material in an attempt to prevent and treat illness. Although such attempts produced no positive benefit, further research found in the mid twentieth century that human cells could be used to help prevent the human body rejecting transplanted organs, leading in time to successful bone marrow transplantation as has become common practice in treatment for patients that have compromised bone marrow after disease, infection, radiation or chemotherapy. In recent decades, however, stem cell and cel ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Bioprocessing
Bioprocess engineering, also biochemical engineering, is a specialization of chemical engineering or biological engineering. It deals with the design and development of equipment and processes for the manufacturing of products such as agriculture, food, feed, pharmaceuticals, nutraceuticals, chemicals, and polymers and paper from biological materials & treatment of waste water. Bioprocess engineering is a conglomerate of mathematics, biology and industrial design, and consists of various spectrums like the design and study of bioreactors (operational mode, instrumentation, and physical layout) to the creation of kinetic models. It also deals with studying various biotechnological processes used in industries for large scale production of biological product for optimization of yield in the end product and the quality of end product. Bioprocess engineering may include the work of mechanical, electrical, and industrial engineers to apply principles of their disciplines to processes ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cell Culture
Cell culture or tissue culture is the process by which cells are grown under controlled conditions, generally outside of their natural environment. The term "tissue culture" was coined by American pathologist Montrose Thomas Burrows. This technique is also called micropropagation. After the cells of interest have been isolated from living tissue, they can subsequently be maintained under carefully controlled conditions the need to be kept at body temperature (37 °C) in an incubator. These conditions vary for each cell type, but generally consist of a suitable vessel with a substrate or rich medium that supplies the essential nutrients (amino acids, carbohydrates, vitamins, minerals), growth factors, hormones, and gases ( CO2, O2), and regulates the physio-chemical environment (pH buffer, osmotic pressure, temperature). Most cells require a surface or an artificial substrate to form an adherent culture as a monolayer (one single-cell thick), whereas others can be grown ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chromatography
In chemical analysis, chromatography is a laboratory technique for the separation of a mixture into its components. The mixture is dissolved in a fluid solvent (gas or liquid) called the ''mobile phase'', which carries it through a system (a column, a capillary tube, a plate, or a sheet) on which a material called the ''stationary phase'' is fixed. Because the different constituents of the mixture tend to have different affinities for the stationary phase and are retained for different lengths of time depending on their interactions with its surface sites, the constituents travel at different apparent velocities in the mobile fluid, causing them to separate. The separation is based on the differential partitioning between the mobile and the stationary phases. Subtle differences in a compound's partition coefficient result in differential retention on the stationary phase and thus affect the separation. Chromatography may be preparative or analytical. The purpose of preparativ ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pharming (genetics)
Pharming, a portmanteau of "farming" and "pharmaceutical", refers to the use of genetic engineering to insert genes that code for useful pharmaceuticals into host animals or plants that would otherwise not express those genes, thus creating a genetically modified organism (GMO). Pharming is also known as molecular farming, molecular pharming or biopharming. The products of pharming are recombinant proteins or their metabolic products. Recombinant proteins are most commonly produced using bacteria or yeast in a bioreactor, but pharming offers the advantage to the producer that it does not require expensive infrastructure, and production capacity can be quickly scaled to meet demand, at greatly reduced cost. History The first recombinant plant-derived protein (PDP) was human serum albumin, initially produced in 1990 in transgenic tobacco and potato plants. Open field growing trials of these crops began in the United States in 1992 and have taken place every year since. While th ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Fed-batch
Fed-batch culture is, in the broadest sense, defined as an operational technique in biotechnological processes where one or more nutrients (substrates) are fed (supplied) to the bioreactor during cultivation and in which the product(s) remain in the bioreactor until the end of the run. An alternative description of the method is that of a culture in which "a base medium supports initial cell culture and a feed medium is added to prevent nutrient depletion". It is also a type of semi-batch culture. In some cases, all the nutrients are fed into the bioreactor. The advantage of the fed-batch culture is that one can control concentration of fed-substrate in the culture liquid at arbitrarily desired levels (in many cases, at low levels). Generally speaking, fed-batch culture is superior to conventional batch culture when controlling concentrations of a nutrient (or nutrients) affects the yield or productivity of the desired metabolite. Types of bioprocesses The types of bioprocess ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
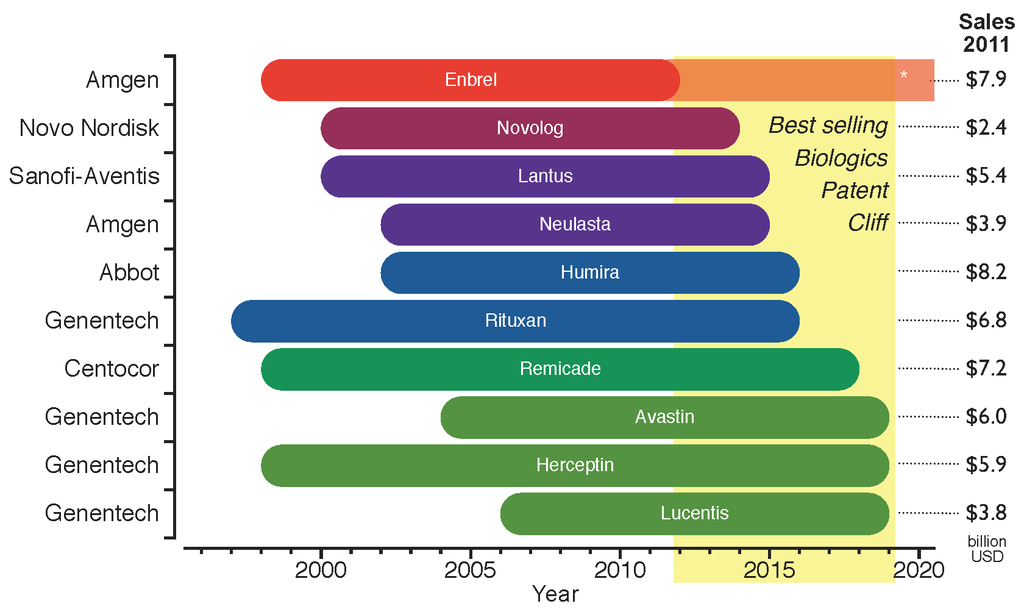
Biosimilars
A biosimilar (also known as follow-on biologic or subsequent entry biologic) is a biologic medical product that is almost an identical copy of an original product that is manufactured by a different company. Biosimilars are officially approved versions of original "innovator" products and can be manufactured when the original product's patent expires. Reference to the innovator product is an integral component of the approval. Unlike with generic drugs of the more common small-molecule type, biologics generally exhibit high molecular complexity and may be quite sensitive to changes in manufacturing processes. Despite that heterogeneity, all biopharmaceuticals, including biosimilars, must maintain consistent quality and clinical performance throughout their lifecycle. Drug-related authorities such as the European Medicines Agency (EMA) of the European Union, the United States Food and Drug Administration (FDA), and the Health Products and Food Branch of Health Canada hold their o ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Biomanufacturing
Biomanufacturing is a type of manufacturing or biotechnology that utilizes biological systems to produce commercially important biomaterials and biomolecules for use in medicines, food and beverage processing, and industrial applications. Biomanufacturing products are recovered from natural sources, such as blood, or from cultures of microbes, animal cells, or plant cells grown in specialized equipment. The cells used during the production may have been naturally occurring or derived using genetic engineering techniques. Products There are thousands of biomanufacturing products on the market today. Some examples of general classes are listed below: Medicine * Amino acids * Biopharmaceuticals * Cytokines * Fusion proteins * Growth factors * Monoclonal antibodies * Vaccines Food and beverage * Amino acids * Enzymes * Protein supplements Industrial applications that employ cells and/or enzymes * Biocementation * Bioremediation * Detergents * Plastics Unit operations A p ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Biopharmaceutical
A biopharmaceutical, also known as a biological medical product, or biologic, is any pharmaceutical drug product manufactured in, extracted from, or semisynthesized from biological sources. Different from totally synthesized pharmaceuticals, they include vaccines, whole blood, blood components, allergenics, somatic cells, gene therapies, tissues, recombinant therapeutic protein, and living medicines used in cell therapy. Biologics can be composed of sugars, proteins, nucleic acids, or complex combinations of these substances, or may be living cells or tissues. They (or their precursors or components) are isolated from living sources—human, animal, plant, fungal, or microbial. They can be used in both human and animal medicine. Terminology surrounding biopharmaceuticals varies between groups and entities, with different terms referring to different subsets of therapeutics within the general biopharmaceutical category. Some regulatory agencies use the terms ''biological ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |