|
Aggregation-induced Emission
Aggregation-induced emission (AIE) is a phenomenon that is observed with certain organic luminophores (fluorescent dyes). The photoemission efficiencies of most organic compounds is higher in solution than in the solid state. Photoemission from some organic compounds follows the reverse pattern, being greater in the solid than in solution. The effect is attributed to the decreased flexibility in the solid. Aggregation-induced emission enhancement The phenomenon in which organic luminophores show higher photoluminescence efficiency in the aggregated state than in solution is called aggregation-induced emission enhancement (AIEE). Some luminophores, e.g., diketopyrrolopyrrole-based and sulfonamide In organic chemistry, the sulfonamide functional group (also spelled sulphonamide) is an organosulfur group with the structure . It consists of a sulfonyl group () connected to an amine group (). Relatively speaking this group is unreactive. ...-based luminophores, only displa ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Organic Compound
In chemistry, organic compounds are generally any chemical compounds that contain carbon-hydrogen or carbon-carbon bonds. Due to carbon's ability to catenate (form chains with other carbon atoms), millions of organic compounds are known. The study of the properties, reactions, and syntheses of organic compounds comprise the discipline known as organic chemistry. For historical reasons, a few classes of carbon-containing compounds (e.g., carbonate salts and cyanide salts), along with a few other exceptions (e.g., carbon dioxide, hydrogen cyanide), are not classified as organic compounds and are considered inorganic. Other than those just named, little consensus exists among chemists on precisely which carbon-containing compounds are excluded, making any rigorous definition of an organic compound elusive. Although organic compounds make up only a small percentage of Earth's crust, they are of central importance because all known life is based on organic compounds. Living t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Luminophore
In chemistry, a luminophore (sometimes shortened to lumophore) is an atom or functional group in a chemical compound that is responsible for its luminescent properties. Luminophores can be either organic or inorganic. Luminophores can be further classified as fluorophores or phosphors, depending on the nature of the excited state responsible for the emission of photons. However, some luminophores cannot be classified as being exclusively fluorophores or phosphors. Examples include transition-metal complexes such as tris(bipyridine)ruthenium(II) chloride, whose luminescence comes from an excited (nominally triplet) metal-to-ligand charge-transfer (MLCT) state, which is not a true triplet state in the strict sense of the definition; and colloidal quantum dots, whose emissive state does not have either a purely singlet or triplet spin. Most luminophores consist of conjugated π systems or transition-metal complexes. There are also purely inorganic luminophores, such as zinc ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Photoemission
The photoelectric effect is the emission of electrons when electromagnetic radiation, such as light, hits a material. Electrons emitted in this manner are called photoelectrons. The phenomenon is studied in condensed matter physics, and solid state and quantum chemistry to draw inferences about the properties of atoms, molecules and solids. The effect has found use in electronic devices specialized for light detection and precisely timed electron emission. The experimental results disagree with classical electromagnetism, which predicts that continuous light waves transfer energy to electrons, which would then be emitted when they accumulate enough energy. An alteration in the intensity of light would theoretically change the kinetic energy of the emitted electrons, with sufficiently dim light resulting in a delayed emission. The experimental results instead show that electrons are dislodged only when the light exceeds a certain frequency—regardless of the light's intensity or d ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Organic Compounds
In chemistry, organic compounds are generally any chemical compounds that contain carbon-hydrogen or carbon-carbon bonds. Due to carbon's ability to catenate (form chains with other carbon atoms), millions of organic compounds are known. The study of the properties, reactions, and syntheses of organic compounds comprise the discipline known as organic chemistry. For historical reasons, a few classes of carbon-containing compounds (e.g., carbonate salts and cyanide salts), along with a few other exceptions (e.g., carbon dioxide, hydrogen cyanide), are not classified as organic compounds and are considered inorganic. Other than those just named, little consensus exists among chemists on precisely which carbon-containing compounds are excluded, making any rigorous definition of an organic compound elusive. Although organic compounds make up only a small percentage of Earth's crust, they are of central importance because all known life is based on organic compounds. Living thin ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Solution (chemistry)
In chemistry, a solution is a special type of homogeneous mixture composed of two or more substances. In such a mixture, a solute is a substance dissolved in another substance, known as a solvent. If the attractive forces between the solvent and solute particles are greater than the attractive forces holding the solute particles together, the solvent particles pull the solute particles apart and surround them. These surrounded solute particles then move away from the solid solute and out into the solution. The mixing process of a solution happens at a scale where the effects of chemical polarity are involved, resulting in interactions that are specific to solvation. The solution usually has the state of the solvent when the solvent is the larger fraction of the mixture, as is commonly the case. One important parameter of a solution is the concentration, which is a measure of the amount of solute in a given amount of solution or solvent. The term "aqueous solution" is used when ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Solid (state Of Matter)
Solid is one of the four fundamental states of matter (the others being liquid, gas, and plasma). The molecules in a solid are closely packed together and contain the least amount of kinetic energy. A solid is characterized by structural rigidity and resistance to a force applied to the surface. Unlike a liquid, a solid object does not flow to take on the shape of its container, nor does it expand to fill the entire available volume like a gas. The atoms in a solid are bound to each other, either in a regular geometric lattice (crystalline solids, which include metals and ordinary ice), or irregularly (an amorphous solid such as common window glass). Solids cannot be compressed with little pressure whereas gases can be compressed with little pressure because the molecules in a gas are loosely packed. The branch of physics that deals with solids is called solid-state physics, and is the main branch of condensed matter physics (which also includes liquids). Materials sc ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Diketopyrrolopyrrole Dye
Diketopyrrolopyrroles (DPPs) are organic dyes and pigments based on the heterocyclic dilactam 2,5-dihydropyrrolo ,4-''c''yrrole-1,4-dione, widely used in optoelectronics. DPPs were initially used as pigments in the painting industry (e.g. in automotive paints) due to their high resistance to photodegradation. More recently, DPP derivatives have been also investigated as promising fluorescent dyes for bioimaging applications, as well as components of materials for use in organic electronics. Structure DPP dyes are based on the bicyclic heterocyclic compound diketopyrrolopyrrole. 2,5-Dihydropyrrolo ,4-cyrrol-1,4-dion is a basic body of Diketopyrrolopyrrole dye. DPP pigments are an important class of high-performance pigments used in inks, paints and plastic. More recently their optical-electronic performance was discovered for photovoltaic applications and related uses. Optical properties DPP has high stability to heat but not very well to acid and base. The alkylation of am ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
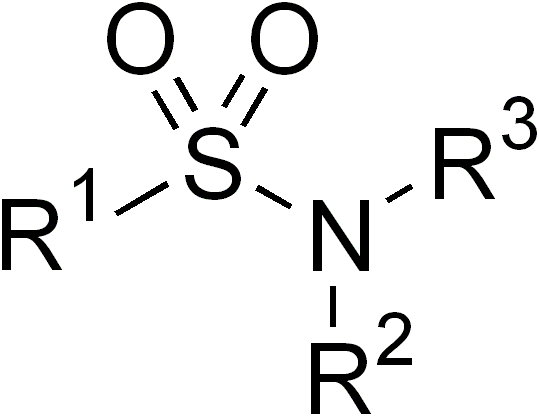
Sulfonamide
In organic chemistry, the sulfonamide functional group (also spelled sulphonamide) is an organosulfur group with the structure . It consists of a sulfonyl group () connected to an amine group (). Relatively speaking this group is unreactive. Because of the rigidity of the functional group, sulfonamides are typically crystalline; for this reason, the formation of a sulfonamide is a classic method to convert an amine into a crystalline derivative which can be identified by its melting point. Many important drugs contain the sulfonamide group. A sulfonamide (compound) is a chemical compound that contains this group. The general formula is or , where each R is some organic group; for example, "methanesulfonamide" (where R = methane, R' = R" = hydrogen) is . Any sulfonamide can be considered as derived from a sulfonic acid by replacing a hydroxyl group () with an amine group. In medicine, the term "sulfonamide" is sometimes used as a synonym for sulfa drug, a derivative or var ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Luminescence
Luminescence is spontaneous emission of light by a substance not resulting from heat; or "cold light". It is thus a form of cold-body radiation. It can be caused by chemical reactions, electrical energy, subatomic motions or stress on a crystal. This distinguishes luminescence from incandescence, which is light emitted by a substance as a result of heating. Historically, radioactivity was thought of as a form of "radio-luminescence", although it is today considered to be separate since it involves more than electromagnetic radiation. The dials, hands, scales, and signs of aviation and navigational instruments and markings are often coated with luminescent materials in a process known as "luminising". Types The following are types of luminescence: * Chemiluminescence, the emission of light as a result of a chemical reaction **Bioluminescence, a result of biochemical reactions in a living organism **Electrochemiluminescence, a result of an electrochemical reaction **Lyolumine ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Materials
Material is a substance or mixture of substances that constitutes an object. Materials can be pure or impure, living or non-living matter. Materials can be classified on the basis of their physical and chemical properties, or on their geological origin or biological function. Materials science is the study of materials, their properties and their applications. Raw materials can be processed in different ways to influence their properties, by purification, shaping or the introduction of other materials. New materials can be produced from raw materials by synthesis. In industry, materials are inputs to manufacturing processes to produce products or more complex materials. Historical elements Materials chart the history of humanity. The system of the three prehistoric ages (Stone Age, Bronze Age, Iron Age) were succeeded by historical ages: steel age in the 19th century, polymer age in the middle of the following century (plastic age) and silicon age in the second half of the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Organic Chemistry
Organic chemistry is a subdiscipline within chemistry involving the scientific study of the structure, properties, and reactions of organic compounds and organic materials, i.e., matter in its various forms that contain carbon atoms.Clayden, J.; Greeves, N. and Warren, S. (2012) ''Organic Chemistry''. Oxford University Press. pp. 1–15. . Study of structure determines their structural formula. Study of properties includes physical and chemical properties, and evaluation of chemical reactivity to understand their behavior. The study of organic reactions includes the chemical synthesis of natural products, drugs, and polymers, and study of individual organic molecules in the laboratory and via theoretical ( in silico) study. The range of chemicals studied in organic chemistry includes hydrocarbons (compounds containing only carbon and hydrogen) as well as compounds based on carbon, but also containing other elements, especially oxygen, nitrogen, sulfur, phosphorus (included in ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |