|
Anaerobic Corrosion
Anaerobic corrosion (also known as hydrogen corrosion) is a form of metal corrosion occurring in anoxic water. Typically following aerobic corrosion, anaerobic corrosion involves a redox reaction that reduces hydrogen ions and oxidizes a solid metal. This process can occur in either abiotic conditions through a thermodynamically spontaneous reaction or biotic conditions through a process known as bacterial anaerobic corrosion. Along with other forms of corrosion, anaerobic corrosion is significant when considering the safe, permanent storage of chemical waste. Chemical mechanisms The overall process of corrosion can be represented by a bimodal function, where the type of corrosion varies with time, including both oxygen-driven and anaerobic mechanisms. The dominant process will depend on the given conditions. During oxygen-driven corrosion, layers of rust form, creating various non-homogenous anoxic niches throughout the metal's surface. Within the niches the diffusion of oxy ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Bacterial Anaerobic Corrosion-en
Bacteria (; singular: bacterium) are ubiquitous, mostly free-living organisms often consisting of one biological cell. They constitute a large domain of prokaryotic microorganisms. Typically a few micrometres in length, bacteria were among the first life forms to appear on Earth, and are present in most of its habitats. Bacteria inhabit soil, water, acidic hot springs, radioactive waste, and the deep biosphere of Earth's crust. Bacteria are vital in many stages of the nutrient cycle by recycling nutrients such as the fixation of nitrogen from the atmosphere. The nutrient cycle includes the decomposition of dead bodies; bacteria are responsible for the putrefaction stage in this process. In the biological communities surrounding hydrothermal vents and cold seeps, extremophile bacteria provide the nutrients needed to sustain life by converting dissolved compounds, such as hydrogen sulphide and methane, to energy. Bacteria also live in symbiotic and parasitic relationships wi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Anode
An anode is an electrode of a polarized electrical device through which conventional current enters the device. This contrasts with a cathode, an electrode of the device through which conventional current leaves the device. A common mnemonic is ACID, for "anode current into device". The direction of conventional current (the flow of positive charges) in a circuit is opposite to the direction of electron flow, so (negatively charged) electrons flow out the anode of a galvanic cell, into an outside or external circuit connected to the cell. For example, the end of a household battery marked with a "-" (minus) is the anode. In both a galvanic cell and an electrolytic cell, the anode is the electrode at which the oxidation reaction occurs. In a galvanic cell the anode is the wire or plate having excess negative charge as a result of the oxidation reaction. In an electrolytic cell, the anode is the wire or plate upon which excess positive charge is imposed. As a result of this, anion ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electrochemistry
Electrochemistry is the branch of physical chemistry concerned with the relationship between electrical potential difference, as a measurable and quantitative phenomenon, and identifiable chemical change, with the potential difference as an outcome of a particular chemical change, or vice versa. These reactions involve electrons moving via an electronically-conducting phase (typically an external electrical circuit, but not necessarily, as in electroless plating) between electrodes separated by an ionically conducting and electronically insulating electrolyte (or ionic species in a solution). When a chemical reaction is driven by an electrical potential difference, as in electrolysis, or if a potential difference results from a chemical reaction as in an electric battery or fuel cell, it is called an ''electrochemical'' reaction. Unlike in other chemical reactions, in electrochemical reactions electrons are not transferred directly between atoms, ions, or molecules, but via the af ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
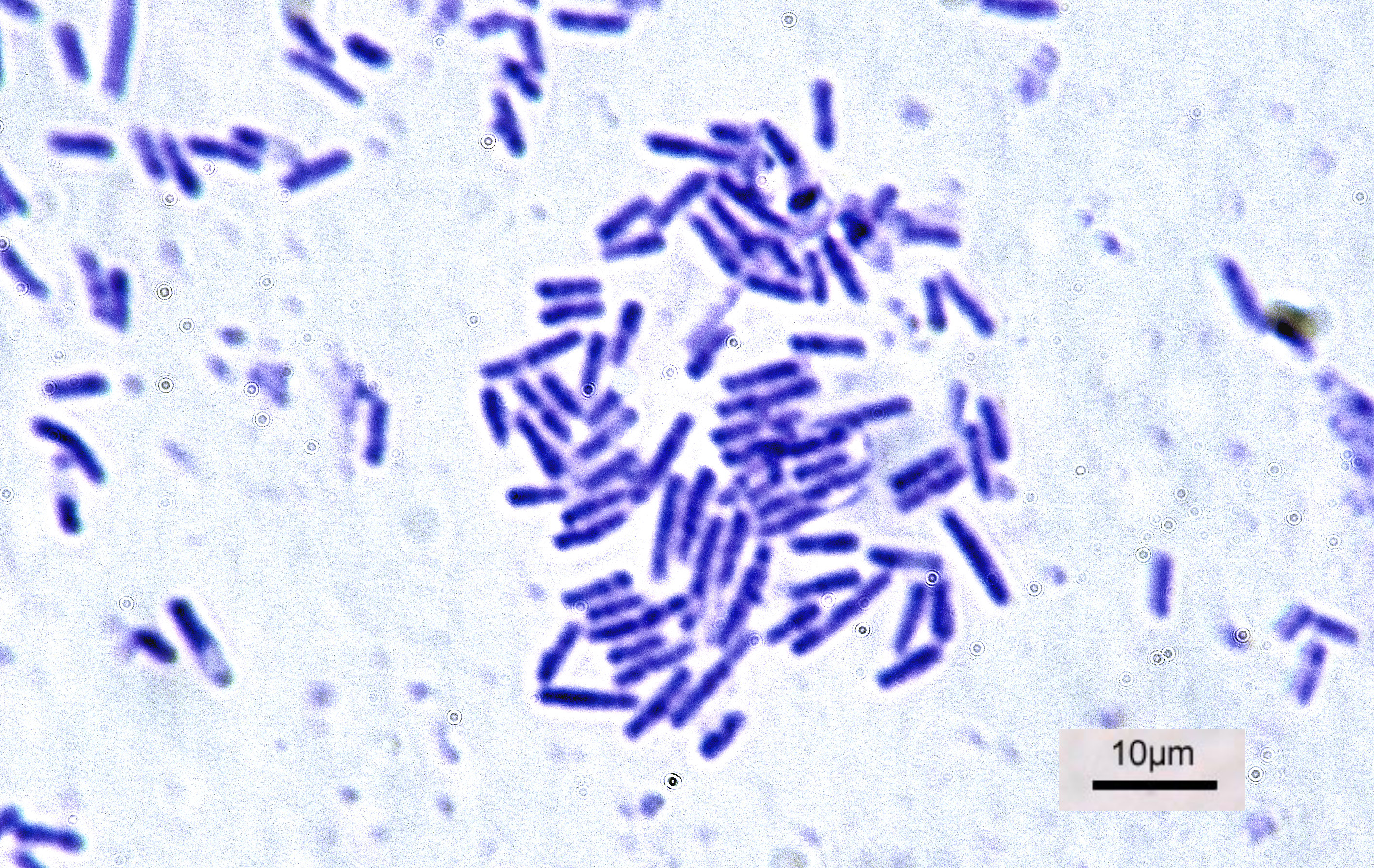
Bacterial Anaerobic Corrosion
Bacterial anaerobic corrosion is the bacterially-induced oxidation of metals. Corrosion of metals typically alters the metal to a form that is more stable. Thus, bacterial anaerobic corrosion typically occurs in conditions favorable to the corrosion of the underlying substrate. In humid, anoxic conditions the corrosion of metals occurs as a result of a redox reaction. This redox reaction generates molecular hydrogen from local hydrogen ions. Conversely, anaerobic corrosion occurs spontaneously. Anaerobic corrosion primarily occurs on metallic substrates but may also occur on concrete. Details Bacterial anaerobic corrosion typically impacts metallic substrates but may also occur in concrete. Corrosion of concrete mediums leads to considerable losses in industrial settings. When considering the corrosion of concrete there is significant documentation of structural degradation in concrete wastewater infrastructure where wastewater is collected or treated. Similarly, biofilms are i ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Corrosion
Corrosion is a natural process that converts a refined metal into a more chemically stable oxide. It is the gradual deterioration of materials (usually a metal) by chemical or electrochemical reaction with their environment. Corrosion engineering is the field dedicated to controlling and preventing corrosion. In the most common use of the word, this means electrochemical oxidation of metal in reaction with an oxidant such as oxygen, hydrogen or hydroxide. Rusting, the formation of iron oxides, is a well-known example of electrochemical corrosion. This type of damage typically produces oxide(s) or salt(s) of the original metal and results in a distinctive orange colouration. Corrosion can also occur in materials other than metals, such as ceramics or polymers, although in this context, the term "degradation" is more common. Corrosion degrades the useful properties of materials and structures including strength, appearance and permeability to liquids and gases. Many structural ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Deep Geological Repository
A deep geological repository is a way of storing hazardous or radioactive waste within a stable geologic environment (typically 200–1000 m deep). It entails a combination of waste form, waste package, engineered seals and geology that is suited to provide a high level of long-term isolation and containment without future maintenance. This will prevent any radioactive dangers. A number of mercury, cyanide and arsenic waste repositories are operating worldwide including Canada (Giant Mine) and Germany (potash mines in Herfa-Neurode and Zielitz) and a number of radioactive waste storages are under construction with the Onkalo in Finland being the most advanced. Principles and background Highly toxic waste that cannot be further recycled must be stored in isolation to avoid contamination of air, ground and underground water. Deep geological repository is a type of long-term storage that isolates waste in geological structures that are expected to be stable for millions of years, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sulfate-reducing Microorganism
Sulfate-reducing microorganisms (SRM) or sulfate-reducing prokaryotes (SRP) are a group composed of sulfate-reducing bacteria (SRB) and sulfate-reducing archaea (SRA), both of which can perform anaerobic respiration utilizing sulfate () as terminal electron acceptor, reducing it to hydrogen sulfide (H2S). Therefore, these sulfidogenic microorganisms "breathe" sulfate rather than molecular oxygen (O2), which is the terminal electron acceptor reduced to water (H2O) in aerobic respiration. Most sulfate-reducing microorganisms can also reduce some other oxidized inorganic sulfur compounds, such as sulfite (), dithionite (), thiosulfate (), trithionate (), tetrathionate (), elemental sulfur (S8), and polysulfides (). Depending on the context, "sulfate-reducing microorganisms" can be used in a broader sense (including all species that can reduce any of these sulfur compounds) or in a narrower sense (including only species that reduce sulfate, and excluding strict thiosulfate and sulfu ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sulfide
Sulfide (British English also sulphide) is an inorganic anion of sulfur with the chemical formula S2− or a compound containing one or more S2− ions. Solutions of sulfide salts are corrosive. ''Sulfide'' also refers to chemical compounds large families of inorganic and organic compounds, e.g. lead sulfide and dimethyl sulfide. Hydrogen sulfide (H2S) and bisulfide (SH−) are the conjugate acids of sulfide. Chemical properties The sulfide ion, S2−, does not exist in aqueous alkaline solutions of Na2S. Instead sulfide converts to hydrosulfide: :S2− + H2O → SH− + OH− Upon treatment with an acid, sulfide salts convert to hydrogen sulfide: :S2− + H+ → SH− :SH− + H+ → H2S Oxidation of sulfide is a complicated process. Depending on the conditions, the oxidation can produce elemental sulfur, polysulfides, polythionates, sulfite, or sulfate. Metal sulfides react with halogens, forming sulfur and metal salts. :8 MgS + 8 I2 → S8 + 8 M ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Schikorr Reaction
The Schikorr reaction formally describes the conversion of the iron(II) hydroxide (Fe(OH)2) into iron(II,III) oxide (Fe3O4). This transformation reaction was first studied by Gerhard Schikorr. The global reaction follows: :\underset -> \underset + \underset + \underset It is of special interest in the context of the serpentinization, the formation of hydrogen by the action of water on a common mineral. Reaction mechanism The ''Schikorr reaction'' can be viewed as two distinct processes: * the wikt:anaerobic, anaerobic oxidation of two Fe(II) (Fe2+) into Fe(III) (Fe3+) by the protons of water. The redox, reduction of two water protons is accompanied by the production of molecular hydrogen (H2), and; * the loss of two water molecules from the iron(II) and iron(III) hydroxides giving rise to its dehydration and to the formation of a thermodynamically more stable phase iron(II,III) oxide. The global reaction can thus be decomposed in half redox reactions as follows: :2 (Fe2+ → Fe3 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Magnetite
Magnetite is a mineral and one of the main iron ores, with the chemical formula Fe2+Fe3+2O4. It is one of the oxides of iron, and is ferrimagnetic; it is attracted to a magnet and can be magnetized to become a permanent magnet itself. With the exception of extremely rare native iron deposits, it is the most magnetic of all the naturally occurring minerals on Earth. Naturally magnetized pieces of magnetite, called lodestone, will attract small pieces of iron, which is how ancient peoples first discovered the property of magnetism. Magnetite is black or brownish-black with a metallic luster, has a Mohs hardness of 5–6 and leaves a black streak. Small grains of magnetite are very common in igneous and metamorphic rocks. The chemical IUPAC name is iron(II,III) oxide and the common chemical name is ''ferrous-ferric oxide''. Properties In addition to igneous rocks, magnetite also occurs in sedimentary rocks, including banded iron formations and in lake and marine sediments ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Iron(II) Hydroxide
Iron(II) hydroxide or ferrous hydroxide is an inorganic compound with the formula Fe(OH)2. It is produced when iron(II) salts, from a compound such as iron(II) sulfate, are treated with hydroxide ions. Iron(II) hydroxide is a white solid, but even traces of oxygen impart a greenish tinge. The air-oxidised solid is sometimes known as " green rust". Preparation and reactions Iron(II) hydroxide is poorly soluble in water (1.43 × 10−3 g/ L), or 1.59 × 10−5 mol/L. It precipitates from the reaction of iron(II) and hydroxide salts:H. Lux "Iron(II) Hydroxide" in Handbook of Preparative Inorganic Chemistry, 2nd Ed. Edited by G. Brauer, Academic Press, 1963, NY. Vol. 1. p. 1498. :FeSO4 + 2 NaOH → Fe(OH)2 + Na2SO4 If the solution is not deoxygenated and iron not totally reduced in Fe(II), the precipitate can vary in colour starting from green to reddish brown depending on the iron(III) content. Iron(II) ions are easily substituted by iron(III) ions produced by its progr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Metal Hydroxide
Metal hydroxides are hydroxides of metals. They are often strong bases. They consist of hydroxide anions and metallic cations. Some metal hydroxides, such as alkali metal hydroxides, ionize completely when dissolved. Certain metal hydroxides are weak electrolytes and dissolve only partially in aqueous solution. Examples * Aluminium hydroxide * Beryllium hydroxide * Cobalt(II) hydroxide * Copper(II) hydroxide * Curium hydroxide * Gold(III) hydroxide * Iron(II) hydroxide * Mercury(II) hydroxide * Nickel(II) hydroxide * Tin(II) hydroxide * Uranyl hydroxide * Zinc hydroxide * Zirconium(IV) hydroxide * Lithium hydroxide * Rubidium hydroxide * Cesium hydroxide * Sodium hydroxide * Potassium hydroxide Alkali metal hydroxides Other metal hydroxides * Gallium(III) hydroxide * Lead(II) hydroxide * Thallium(I) hydroxide * Thallium(III) hydroxide Role in soils In soils, it is assumed that larger amounts of natural phenols are released from decomposing plant litter ra ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |