|
Aminolysis
In chemistry, aminolysis (/am·i·nol·y·sis/) is any chemical reaction in which a molecule is lysed (split into two parts) by reacting with ammonia () or an amine (any molecule containing a nitrogen atom with a lone pair, :N). The case where the reaction involves ammonia may be more specifically referred to as ammonolysis. Reactions Alkyl group An example of an aminolysis reaction is the replacement of a halogen in an alkyl group () by an amine () and the elimination of hydrogen halide (HX). :R-X + R'-NH2 -> R-NH-R' + HX Synthesis of peptides Another common example is the reaction of a primary amine or secondary amine with a carboxylic acid or with a carboxylic acid derivative to form an amide. This reaction is widely used, especially in the synthesis of peptides. On the simple addition of an amine to a carboxylic acid, a salt of the organic acid and base is obtained. To overcome this, the carboxylic acid first needs to be "activated". This is usually done by converting the ac ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Solvolysis
In chemistry, solvolysis is a type of nucleophilic substitution (S1/S2) or elimination where the nucleophile is a solvent molecule. Characteristic of S1 reactions, solvolysis of a chiral reactant affords the racemate. Sometimes however, the stereochemical course is complicated by intimate ion pairs, whereby the leaving anion remains close to the carbocation, effectively shielding it from an attack by the nucleophile. Particularly fast reactions can occur by neighbour group participation, with nonclassical ions as intermediates or transition states. Examples For certain nucleophiles, solvolysis reactions are classified. Solvolysis involving water is called hydrolysis. Related terms are alcoholysis (alcohols) and specifically methanolysis (methanol), acetolysis, ammonolysis (ammonia), and aminolysis (alkyl amines). Glycolysis is however an older term for the multistep conversion of glucose to pyruvate. Hydrolysis While solvolysis often refers to an organic chemistry context, hydr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chemistry
Chemistry is the science, scientific study of the properties and behavior of matter. It is a natural science that covers the Chemical element, elements that make up matter to the chemical compound, compounds made of atoms, molecules and ions: their composition, structure, properties, behavior and the changes they undergo during a Chemical reaction, reaction with other Chemical substance, substances. Chemistry also addresses the nature of chemical bonds in chemical compounds. In the scope of its subject, chemistry occupies an intermediate position between physics and biology. It is sometimes called the central science because it provides a foundation for understanding both Basic research, basic and Applied science, applied scientific disciplines at a fundamental level. For example, chemistry explains aspects of plant growth (botany), the formation of igneous rocks (geology), how atmospheric ozone is formed and how environmental pollutants are degraded (ecology), the properties ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Acid Halide
In organic chemistry, an acyl halide (also known as an acid halide) is a chemical compound derived from an oxoacid by replacing a hydroxyl group () with a halide group (, where X is a halogen). If the acid is a carboxylic acid (), the compound contains a functional group, which consists of a carbonyl group () singly bonded to a halogen atom. The general formula for such an acyl halide can be written RCOX, where R may be, for example, an alkyl group, CO is the carbonyl group, and X represents the halide, such as chloride. Acyl chlorides are the most commonly encountered acyl halides, but acetyl iodide is the one produced (transiently) on the largest scale. Billions of kilograms are generated annually in the production of acetic acid. Preparation Aliphatic acyl halides On an industrial scale, the reaction of acetic anhydride with hydrogen chloride produces a mixture of acetyl chloride and acetic acid: :(CH3CO)2O + HCl -> CH3COCl + CH3CO2H Common syntheses of acyl chlorides als ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
PET Degradation With Polyamines Through Aminolysis Route
A pet, or companion animal, is an animal kept primarily for a person's company or entertainment rather than as a working animal, livestock, or a laboratory animal. Popular pets are often considered to have attractive appearances, intelligence, and relatable personalities, but some pets may be taken in on an altruistic basis (such as a stray animal) and accepted by the owner regardless of these characteristics. Two of the most popular pets are dogs and cats. Other animals commonly kept include rabbits; ferrets; pigs; rodents such as gerbils, hamsters, chinchillas, rats, mice, and guinea pigs; birds such as parrots, passerines, and fowls; reptiles such as turtles, lizards, snakes, and iguanas; aquatic pets such as fish, freshwater snails, and saltwater snails; amphibians such as frogs and salamanders; and arthropod pets such as tarantulas and hermit crabs. Small pets may be grouped together as pocket pets, while the equine and bovine group include the largest companion animals. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dehydrate Ammonium Acetate
In physiology, dehydration is a lack of total body water, with an accompanying disruption of metabolic processes. It occurs when free water loss exceeds free water intake, usually due to exercise, disease, or high environmental temperature. Mild dehydration can also be caused by immersion diuresis, which may increase risk of decompression sickness in divers. Most people can tolerate a 3-4% decrease in total body water without difficulty or adverse health effects. A 5-8% decrease can cause fatigue and dizziness. Loss of over ten percent of total body water can cause physical and mental deterioration, accompanied by severe thirst. Death occurs at a loss of between fifteen and twenty-five percent of the body water.Ashcroft F, Life Without Water in Life at the Extremes. Berkeley and Los Angeles, 2000, 134-138. Mild dehydration is characterized by thirst and general discomfort and is usually resolved with oral rehydration. Dehydration can cause hypernatremia (high levels of sodium i ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Acetamide
Acetamide (systematic name: ethanamide) is an organic compound with the formula CH3CONH2. It is the simplest amide derived from acetic acid. It finds some use as a plasticizer and as an industrial solvent. The related compound ''N'',''N''-dimethylacetamide (DMA) is more widely used, but it is not prepared from acetamide. Acetamide can be considered an intermediate between acetone, which has two methyl (CH3) groups either side of the carbonyl (CO), and urea which has two amide (NH2) groups in those locations. Acetamide is also a naturally occurring mineral with the IMA symbol: Ace. Production Laboratory scale Acetamide can be produced in the laboratory from ammonium acetate by dehydration: : H4CH3CO2] → CH3C(O)NH2 + H2O Alternatively acetamide can be obtained in excellent yield via ammonolysis of acetylacetone under conditions commonly used in reductive amination. It can also be made from anhydrous acetic acid, acetonitrile and very well dried hydrogen chloride gas, using ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
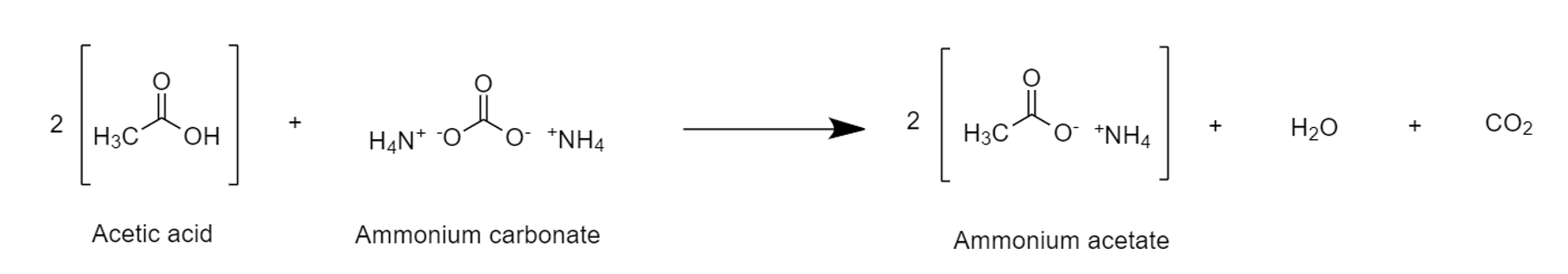
Carboxylic Acid React With Ammonium Carbonate
In organic chemistry, a carboxylic acid is an organic acid that contains a carboxyl group () attached to an R-group. The general formula of a carboxylic acid is or , with R referring to the alkyl, alkenyl, aryl, or other group. Carboxylic acids occur widely. Important examples include the amino acids and fatty acids. Deprotonation of a carboxylic acid gives a carboxylate anion. Examples and nomenclature Carboxylic acids are commonly identified by their trivial names. They at oftentimes have the suffix ''-ic acid''. IUPAC-recommended names also exist; in this system, carboxylic acids have an ''-oic acid'' suffix. For example, butyric acid (C3H7CO2H) is butanoic acid by IUPAC guidelines. For nomenclature of complex molecules containing a carboxylic acid, the carboxyl can be considered position one of the parent chain even if there are other substituents, such as 3-chloropropanoic acid. Alternately, it can be named as a "carboxy" or "carboxylic acid" substituent on another pa ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ammonium Acetate
Ammonium acetate, also known as spirit of Mindererus in aqueous solution, is a chemical compound with the formula NH4CH3CO2. It is a white, hygroscopic solid and can be derived from the reaction of ammonia and acetic acid. It is available commercially. Uses It is the main precursor to acetamide: :NH4CH3CO2 → CH3C(O)NH2 + H2O It is also used as a diuretic. Buffer As the salt of a weak acid and a weak base, ammonium acetate is often used with acetic acid to create a buffer solution. Ammonium acetate is volatile at low pressures. Because of this, it has been used to replace cell buffers that contain non-volatile salts in preparing samples for mass spectrometry. It is also popular as a buffer for mobile phases for HPLC with ELSD detection for this reason. Other volatile salts that have been used for this include ammonium formate. When dissolving ammonium acetate in pure water, the resulting solution typically has a pH of 7, because the equal amounts of acetate and ammonium ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Acetic Acid
Acetic acid , systematically named ethanoic acid , is an acidic, colourless liquid and organic compound with the chemical formula (also written as , , or ). Vinegar is at least 4% acetic acid by volume, making acetic acid the main component of vinegar apart from water and other trace elements. Acetic acid is the second simplest carboxylic acid (after formic acid). It is an important Reagent, chemical reagent and industrial chemical, used primarily in the production of cellulose acetate for photographic film, polyvinyl acetate for wood Adhesive, glue, and synthetic fibres and fabrics. In households, diluted acetic acid is often used in descaling agents. In the food industry, acetic acid is controlled by the E number, food additive code E260 as an acidity regulator and as a condiment. In biochemistry, the acetyl group, derived from acetic acid, is fundamental to all forms of life. When bound to coenzyme A, it is central to the metabolism of carbohydrates and fats. The global ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ammonium Carbonate
Ammonium carbonate is a salt with the chemical formula (NH4)2CO3. Since it readily degrades to gaseous ammonia and carbon dioxide upon heating, it is used as a leavening agent and also as smelling salt. It is also known as baker's ammonia and is a predecessor to the more modern leavening agents baking soda and baking powder. It is a component of what was formerly known as sal volatile and salt of hartshorn, and produces a pungent smell when baked. Production Ammonium carbonate is produced by combining carbon dioxide and aqueous ammonia. About 80,000 tons/year were produced as of 1997. An orthorhombic monohydrate is known. It crystallizes in an ammonia solution exposed in a carbon dioxide-rich atmosphere. Decomposition Ammonium carbonate slowly decomposes at standard temperature and pressure through two pathways. Thus any initially pure sample of ammonium carbonate will soon become a mixture including various byproducts. Ammonium carbonate can spontaneously decompose into ammo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Racemization
In chemistry, racemization is a conversion, by heat or by chemical reaction, of an optically active compound into a racemic (optically inactive) form. This creates a 1:1 molar ratio of enantiomers and is referred too as a racemic mixture (i.e. contain equal amount of (+) and (−) forms). Plus and minus forms are called Dextrorotation and levorotation. The D and L enantiomers are present in equal quantities, the resulting sample is described as a racemic mixture or a racemate. Racemization can proceed through a number of different mechanisms, and it has particular significance in pharmacology as different enantiomers may have different pharmaceutical effects. Stereochemistry Chiral molecules have two forms (at each point of asymmetry), which differ in their optical characteristics: The ''levorotatory form'' (the ''(−)-form'') will rotate counter-clockwise on the plane of polarization of a beam of light, whereas the ''dextrorotatory'' form (the ''(+)-form'') will rotate clockw ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ester
In chemistry, an ester is a compound derived from an oxoacid (organic or inorganic) in which at least one hydroxyl group () is replaced by an alkoxy group (), as in the substitution reaction of a carboxylic acid and an alcohol. Glycerides are fatty acid esters of glycerol; they are important in biology, being one of the main classes of lipids and comprising the bulk of animal fats and vegetable oils. Esters typically have a pleasant smell; those of low molecular weight are commonly used as fragrances and are found in essential oils and pheromones. They perform as high-grade solvents for a broad array of plastics, plasticizers, resins, and lacquers, and are one of the largest classes of synthetic lubricants on the commercial market. Polyesters are important plastics, with monomers linked by ester moieties. Phosphoesters form the backbone of DNA molecules. Nitrate esters, such as nitroglycerin, are known for their explosive properties. '' Nomenclature Etymology Th ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |