|
Aminocarb
Animocarb (Matacil) is an organic chemical compound with the molecular formula C11 H16 N2 O2. It has a colorless or white crystal-like appearance and is most commonly used as an insecticide. History Aminocarb has been extensively used in eastern Canada since 1976 in order to control the spruce budworm. The fate of this chemical in the ecosystem and detection of aminocarb was studied by the use of two-dimensional thin-layer chromatography. The use of thin-layer chromatography helped isolate and identify the methyl amino, amino and hydroxymethyl analogues from the in vitro metabolism of aminocarb by liver homogenates from humans and rats. Production and uses Aminocarb is a carbamate insecticide widely used to protect cotton fields, crop fields, and forests from insect infestation. It helps in the control of aphids, soil mollusks, lepidopterous larvae, and other types of chewing insects. It is most commonly administered as an aerosol spray. Reactions Aminocarb can be ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Organic Compound
In chemistry, organic compounds are generally any chemical compounds that contain carbon-hydrogen or carbon-carbon bonds. Due to carbon's ability to catenate (form chains with other carbon atoms), millions of organic compounds are known. The study of the properties, reactions, and syntheses of organic compounds comprise the discipline known as organic chemistry. For historical reasons, a few classes of carbon-containing compounds (e.g., carbonate salts and cyanide salts), along with a few other exceptions (e.g., carbon dioxide, hydrogen cyanide), are not classified as organic compounds and are considered inorganic. Other than those just named, little consensus exists among chemists on precisely which carbon-containing compounds are excluded, making any rigorous definition of an organic compound elusive. Although organic compounds make up only a small percentage of Earth's crust, they are of central importance because all known life is based on organic compounds. Living t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
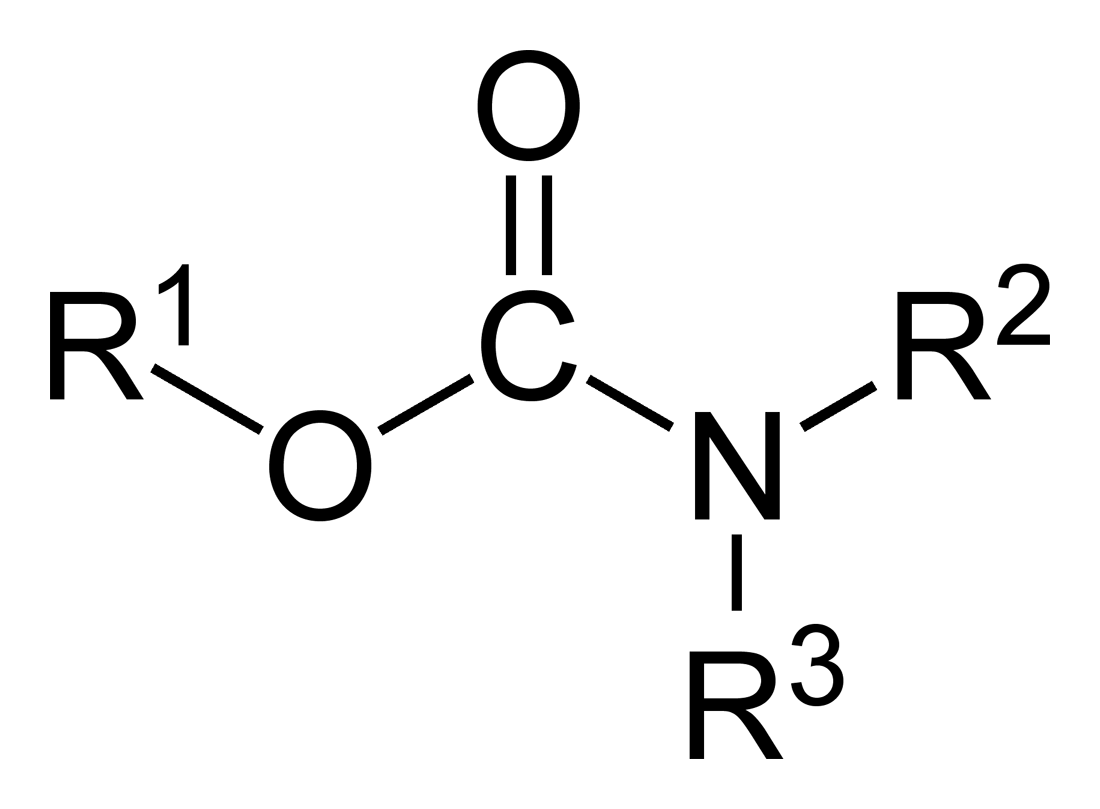
Carbamate
In organic chemistry, a carbamate is a category of organic compounds with the general formula and structure , which are formally derived from carbamic acid (). The term includes organic compounds (e.g., the ester ethyl carbamate), formally obtained by replacing one or more of the hydrogen atoms by other organic functional groups; as well as salts with the carbamate anion (e.g. ammonium carbamate). Polymers whose units are joined by carbamate groups are an important family of plastics, the polyurethanes. Properties While carbamic acids are unstable, many carbamate esters or ionic) are stable and well known. Equilibrium with carbonate and bicarbonate In water solutions, the carbamate anion slowly equilibrates with the ammonium cation and the carbonate or bicarbonate anions: : : Calcium carbamate is soluble in water, whereas calcium carbonate is not. Adding a calcium salt to an ammonium carbamate/carbonate solution will precipitate some calcium carbonate immediatel ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
High Pressure
In science and engineering the study of high pressure examines its effects on materials and the design and construction of devices, such as a diamond anvil cell, which can create high pressure. By ''high pressure'' is usually meant pressures of thousands (kilobars) or millions (megabars) of times atmospheric pressure (about 1 bar or 100,000 Pa). History and overview Percy Williams Bridgman received a Nobel Prize in 1946 for advancing this area of physics by several magnitudes of pressure (400 MPa to 40,000 MPa). The list of founding fathers of this field includes also the names of Harry George Drickamer, Tracy Hall, Francis P. Bundy, Leonid F. Vereschagin, and Sergey M. Stishov. It was by applying high pressure as well as high temperature to carbon that man-made diamonds were first produced as well as many other interesting discoveries. Almost any material when subjected to high pressure will compact itself into a denser form, for example, quartz, also called silica or silicon di ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Irradiation
Irradiation is the process by which an object is exposed to radiation. The exposure can originate from various sources, including natural sources. Most frequently the term refers to ionizing radiation, and to a level of radiation that will serve a specific purpose, rather than radiation exposure to normal levels of background radiation. The term irradiation usually excludes the exposure to non-ionizing radiation, such as infrared, visible light, microwaves from cellular phones or electromagnetic waves emitted by radio and TV receivers and power supplies. Applications Sterilization If administered at appropriate levels, all forms of ionizing radiation can sterilize objects, including medical instruments, disposables such as syringes, and sterilize food. Ionizing radiation (electron beams, X-rays and gamma rays) may be used to kill bacteria in food or other organic material, including blood. Food irradiation, while effective, is seldom used due to problems with public accepta ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ultraviolet Radiation
Ultraviolet (UV) is a form of electromagnetic radiation with wavelength from 10 nm (with a corresponding frequency around 30 PHz) to 400 nm (750 THz), shorter than that of visible light, but longer than X-rays. UV radiation is present in sunlight, and constitutes about 10% of the total electromagnetic radiation output from the Sun. It is also produced by electric arcs and specialized lights, such as mercury-vapor lamps, tanning lamps, and black lights. Although long-wavelength ultraviolet is not considered an ionizing radiation because its photons lack the energy to ionize atoms, it can cause chemical reactions and causes many substances to glow or fluoresce. Consequently, the chemical and biological effects of UV are greater than simple heating effects, and many practical applications of UV radiation derive from its interactions with organic molecules. Short-wave ultraviolet light damages DNA and sterilizes surfaces with which it comes into contact. For hum ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Short-wave
Shortwave radio is radio transmission using shortwave (SW) radio frequencies. There is no official definition of the band, but the range always includes all of the high frequency band (HF), which extends from 3 to 30 MHz (100 to 10 metres); above the medium frequency band (MF), to the bottom of the VHF band. Radio waves in the shortwave band can be reflected or refracted from a layer of electrically charged atoms in the atmosphere called the ionosphere. Therefore, short waves directed at an angle into the sky can be reflected back to Earth at great distances, beyond the horizon. This is called skywave or "skip" propagation. Thus shortwave radio can be used for communication over very long distances, in contrast to radio waves of higher frequency, which travel in straight lines (line-of-sight propagation) and are limited by the visual horizon, about 64 km (40 miles). Shortwave broadcasts of radio programs played an important role in the early days of radio ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrolysis Of Aminocarb
Hydrolysis (; ) is any chemical reaction in which a molecule of water breaks one or more chemical bonds. The term is used broadly for substitution, elimination, and solvation reactions in which water is the nucleophile. Biological hydrolysis is the cleavage of biomolecules where a water molecule is consumed to effect the separation of a larger molecule into component parts. When a carbohydrate is broken into its component sugar molecules by hydrolysis (e.g., sucrose being broken down into glucose and fructose), this is recognized as saccharification. Hydrolysis reactions can be the reverse of a condensation reaction in which two molecules join into a larger one and eject a water molecule. Thus hydrolysis adds water to break down, whereas condensation builds up by removing water. Types Usually hydrolysis is a chemical process in which a molecule of water is added to a substance. Sometimes this addition causes both the substance and water molecule to split into two parts. In ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |