|
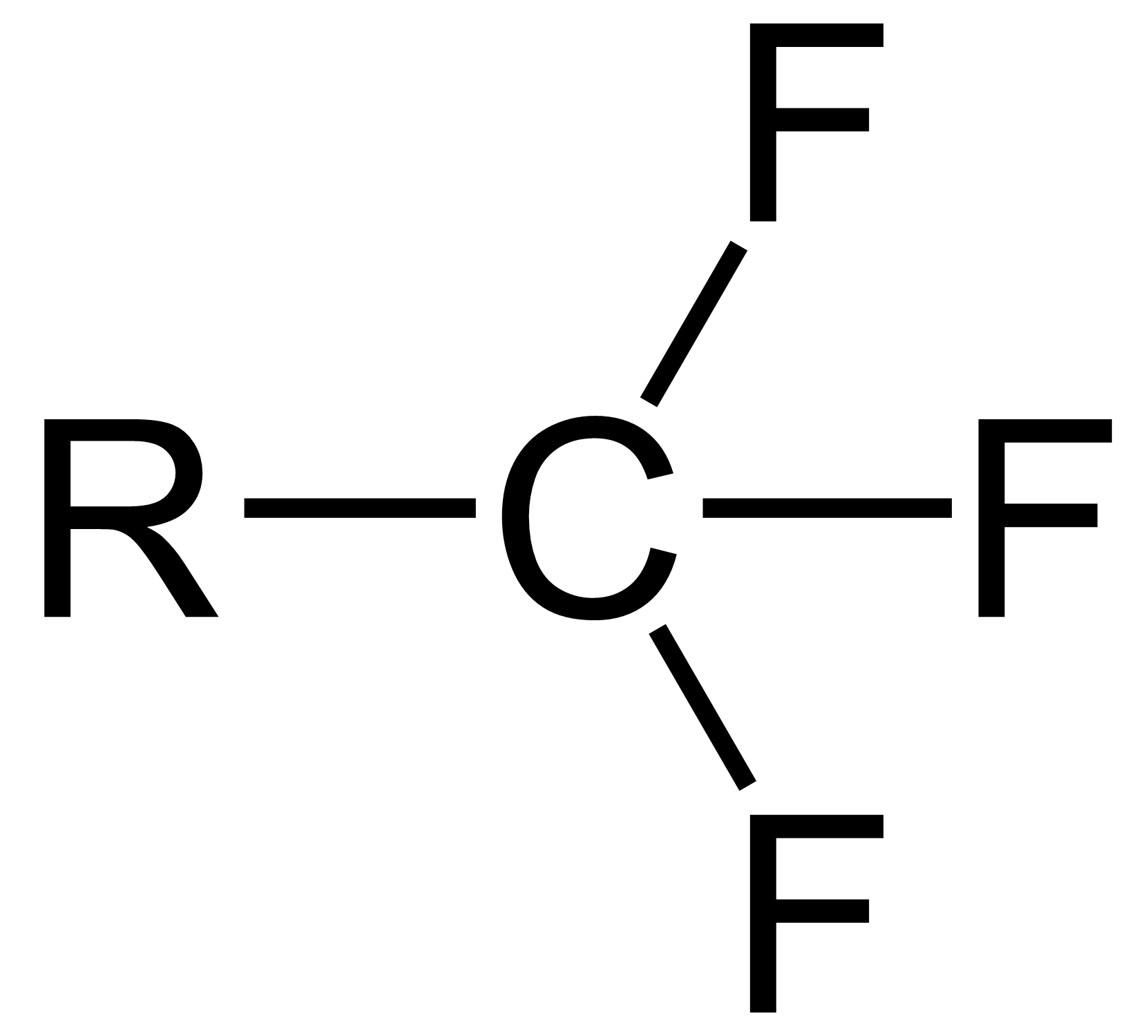
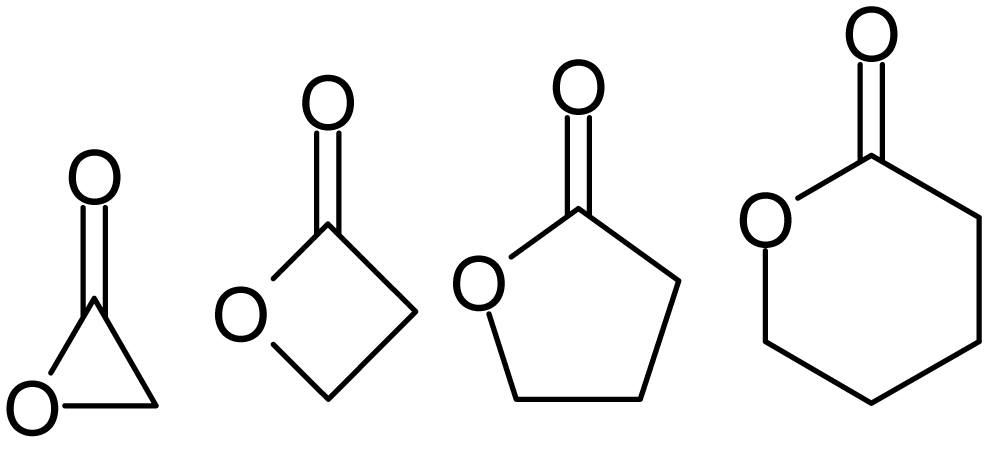
Alpha-acetolactone
Acetolactone (α-acetolactone) is an organic compound with formula . It is the smallest member of the lactone family but can also be described as the epoxide of ethenone. The compound was described in 1997 as a transient species in mass spectrometry experiments. Although acetolactone itself has not been isolated in bulk, the related species bis(trifluoromethyl)acetolactone ((), which has increased electronic stabilisation due to its two trifluoromethyl groups, is known and has a half-life of 8 hours at 25 °C. This compound is prepared by photolysis of bis(trifluoromethyl)malonyl peroxide.Waldemar Adam, Ju-Chao Liu, Oswaldo Rodriguez (1973), ''Bis(trifluoromethyl)acetolactone, a Stable α-Lactone''. J. Org. Chem., volume 38, pages 2269–2270 See also * α-Propiolactone *Oxalic anhydride Oxalic anhydride or ethanedioic anhydride, also called oxiranedione, is a hypothetical organic compound, one of several isomers having the formula C2O3 that have been studied computati ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Organic Compound
In chemistry, organic compounds are generally any chemical compounds that contain carbon-hydrogen or carbon-carbon bonds. Due to carbon's ability to catenate (form chains with other carbon atoms), millions of organic compounds are known. The study of the properties, reactions, and syntheses of organic compounds comprise the discipline known as organic chemistry. For historical reasons, a few classes of carbon-containing compounds (e.g., carbonate salts and cyanide salts), along with a few other exceptions (e.g., carbon dioxide, hydrogen cyanide), are not classified as organic compounds and are considered inorganic. Other than those just named, little consensus exists among chemists on precisely which carbon-containing compounds are excluded, making any rigorous definition of an organic compound elusive. Although organic compounds make up only a small percentage of Earth's crust, they are of central importance because all known life is based on organic compounds. Living t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Lactone
Lactones are cyclic carboxylic esters, containing a 1-oxacycloalkan-2-one structure (), or analogues having unsaturation or heteroatoms replacing one or more carbon atoms of the ring. Lactones are formed by intramolecular esterification of the corresponding hydroxycarboxylic acids, which takes place spontaneously when the ring that is formed is five- or six-membered. Lactones with three- or four-membered rings (α-lactones and β-lactones) are very reactive, making their isolation difficult. Special methods are normally required for the laboratory synthesis of small-ring lactones as well as those that contain rings larger than six-membered. Nomenclature Lactones are usually named according to the precursor acid molecule (''aceto'' = 2 carbon atoms, ''propio'' = 3, ''butyro'' = 4, ''valero'' = 5, ''capro'' = 6, etc.), with a ''-lactone'' suffix and a Greek letter prefix that specifies the number of carbon atoms in the heterocycle — that is, the distance between the relevant -OH ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ethenone
In organic chemistry, ethenone is the formal name for ketene, an organic compound with formula or . It is the simplest member of the ketene class. It is an important reagent for acetylations. Properties Ethenone is a highly reactive gas (at standard conditions) and has a sharp irritating odour. It is only reasonably stable at low temperatures (−80 °C). It must therefore always be prepared for each use and processed immediately, otherwise a dimerization to diketene occurs or it reacts to polymers that are difficult to handle. The polymer content formed during the preparation is reduced, for example, by adding sulfur dioxide to the ketene gas. Because of its cumulative double bonds, ethenone is highly reactive and reacts in an addition reaction H-acidic compounds to the corresponding acetic acid derivatives. It does for example react with water to acetic acid or with primary or secondary amines to the corresponding acetamides. Preparation In industrial chemistry, k ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Mass Spectrometry
Mass spectrometry (MS) is an analytical technique that is used to measure the mass-to-charge ratio of ions. The results are presented as a ''mass spectrum'', a plot of intensity as a function of the mass-to-charge ratio. Mass spectrometry is used in many different fields and is applied to pure samples as well as complex mixtures. A mass spectrum is a type of plot of the ion signal as a function of the mass-to-charge ratio. These spectra are used to determine the elemental or isotopic signature of a sample, the masses of particles and of molecules, and to elucidate the chemical identity or structure of molecules and other chemical compounds. In a typical MS procedure, a sample, which may be solid, liquid, or gaseous, is ionized, for example by bombarding it with a beam of electrons. This may cause some of the sample's molecules to break up into positively charged fragments or simply become positively charged without fragmenting. These ions (fragments) are then separated accordin ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Helmut Schwarz
Helmut Schwarz (born 6 August 1943) is a German organic chemist. He has been a professor of chemistry at the Technische Universität Berlin since 1978. In 2018, he was elected a foreign associate of the U.S. National Academy of Sciences. Career Helmut Schwarz first learned to be a chemical technician and then went on to study chemistry at the TU Berlin. He completed his studies in 1971 and obtained his PhD in 1972 and his Habilitation in 1974 under Ferdinand Bohlmann. He pursued post-doctoral work at the Massachusetts Institute of Technology (MIT) and in the UK, after which he became a professor at the TU Berlin in 1978. Schwarz studies chemical reactions, specifically gas phase chemistry of ionic and radical organic species. He is furthermore working on advancing the analytical capabilities of mass spectrometry. He was president of the Alexander von Humboldt Foundation from 2008 to 2018. From 2010 to 2015 he was president of the German Academy of Researchers Leopoldina an ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Trifluoromethyl
The trifluoromethyl group is a functional group that has the formula -CF3. The naming of is group is derived from the methyl group (which has the formula -CH3), by replacing each hydrogen atom by a fluorine atom. Some common examples are trifluoromethane H–, 1,1,1-trifluoroethane –, and hexafluoroacetone –CO–. Compounds with this group are a subclass of the organofluorines. Properties The trifluoromethyl group has a significant electronegativity that is often described as being intermediate between the electronegativities of fluorine and chlorine. For this reason, trifluoromethyl-substituted compounds are often strong acids, such as trifluoromethanesulfonic acid and trifluoroacetic acid Trifluoroacetic acid (TFA) is an organofluorine compound with the chemical formula CF3CO2H. It is a structural analogue of acetic acid with all three of the acetyl group's hydrogen atoms replaced by fluorine atoms and is a colorless liquid with a .... Conversely, the trifluoromethyl group ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Photodissociation
Photodissociation, photolysis, photodecomposition, or photofragmentation is a chemical reaction in which molecules of a chemical compound are broken down by photons. It is defined as the interaction of one or more photons with one target molecule. Photodissociation is not limited to visible light. Any photon with sufficient energy can affect the chemical bonds of a chemical compound. Since a photon's energy is inversely proportional to its wavelength, electromagnetic radiations with the energy of visible light or higher, such as ultraviolet light, x-rays, and gamma rays can induce such reactions. Photolysis in photosynthesis Photolysis is part of the light-dependent reaction or light phase or photochemical phase or Hill reaction of photosynthesis. The general reaction of photosynthetic photolysis can be given in terms of photons as: :\ce + 2 \text \longrightarrow \ce The chemical nature of "A" depends on the type of organism. Purple sulfur bacteria oxidize hydrogen sulfide () ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
α-Propiolactone
Methyloxiranone or α-propiolactone is a chemical compound of the lactone Lactones are cyclic carboxylic esters, containing a 1-oxacycloalkan-2-one structure (), or analogues having unsaturation or heteroatoms replacing one or more carbon atoms of the ring. Lactones are formed by intramolecular esterification of the co ... family, with a three-membered ring. It is a stable product which can be obtained from the 2-bromopropionate anion. It is an intermediate in the decomposition of 2-chloropropionic acid in the gas phase.Vicent S. Safont, Vicente Moliner, Juan Andrés, Luís R. Domingo (1997)''Theoretical Study of the Elimination Kinetics of Carboxylic Acid Derivatives in the Gas Phase. Decomposition of 2-Chloropropionic Acid'' J. Physical Chemistry series A, volume 101, issue 10, pp. 1859–1865. See also * β-Propiolactone * Acetolactone References Alpha-lactones {{heterocyclic-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Oxalic Anhydride
Oxalic anhydride or ethanedioic anhydride, also called oxiranedione, is a hypothetical organic compound, one of several isomers having the formula C2O3 that have been studied computationally. It can be viewed as the anhydride of oxalic acid or the two-fold ketone of ethylene oxide. It is an oxide of carbon (an oxocarbon). The simple compound apparently has yet to be observed (as of 2009). In 1998, however, Paolo Strazzolini and others have claimed the synthesis of dioxane tetraketone (C4O6), which can be viewed as the cyclic dimer of oxalic anhydride. It has been conjectured to be a fleeting intermediate in the thermal decomposition of certain oxalates and certain chemoluminescent reactions of oxalyl chloride Oxalyl chloride is an organic chemical compound with the formula (COCl)2. This colorless, sharp-smelling liquid, the diacyl chloride of oxalic acid, is a useful reagent in organic synthesis. Preparation Oxalyl chloride was first prepared in 1892 .... See also * 1,2-dio ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |

.jpg)