|
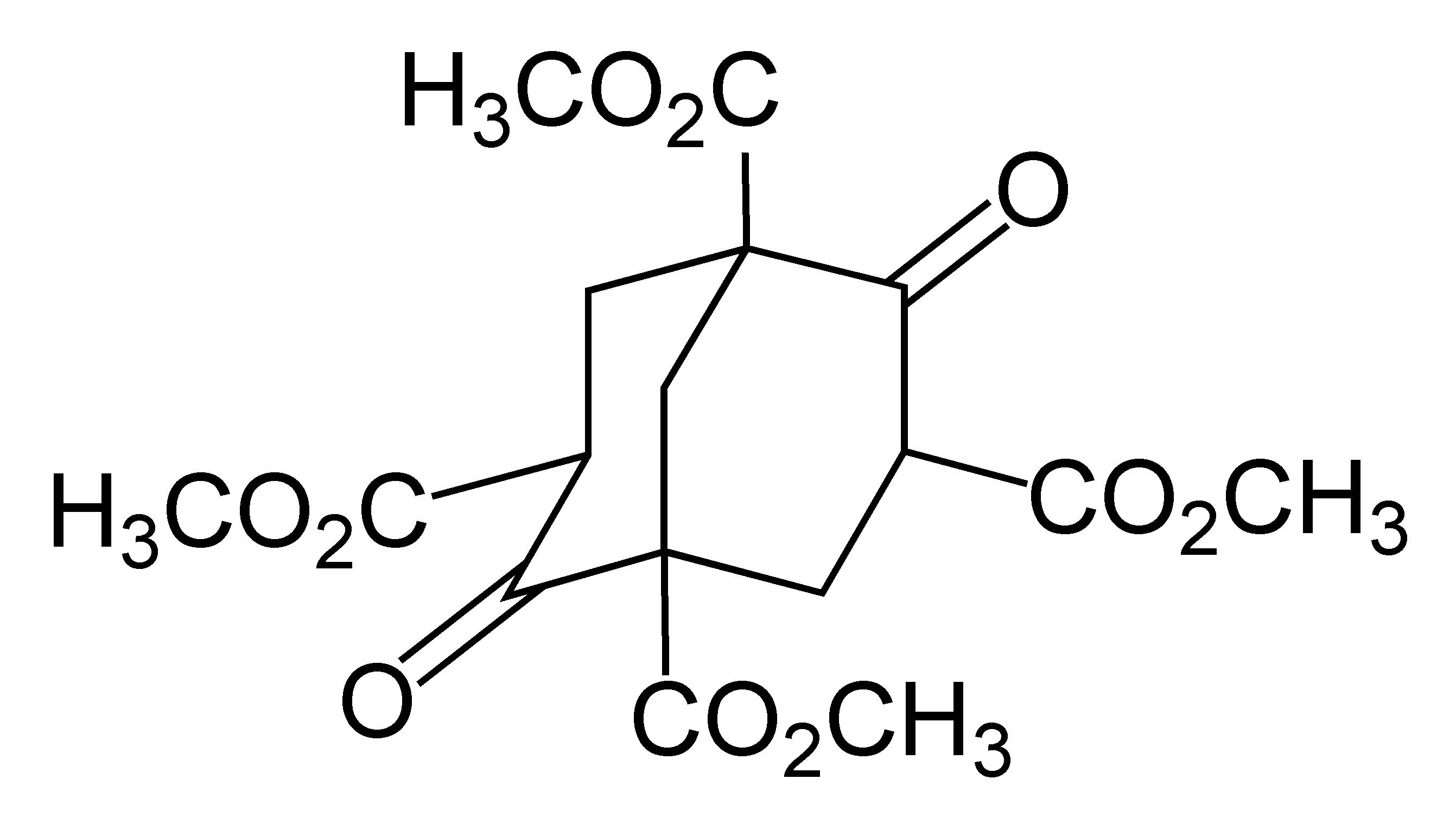
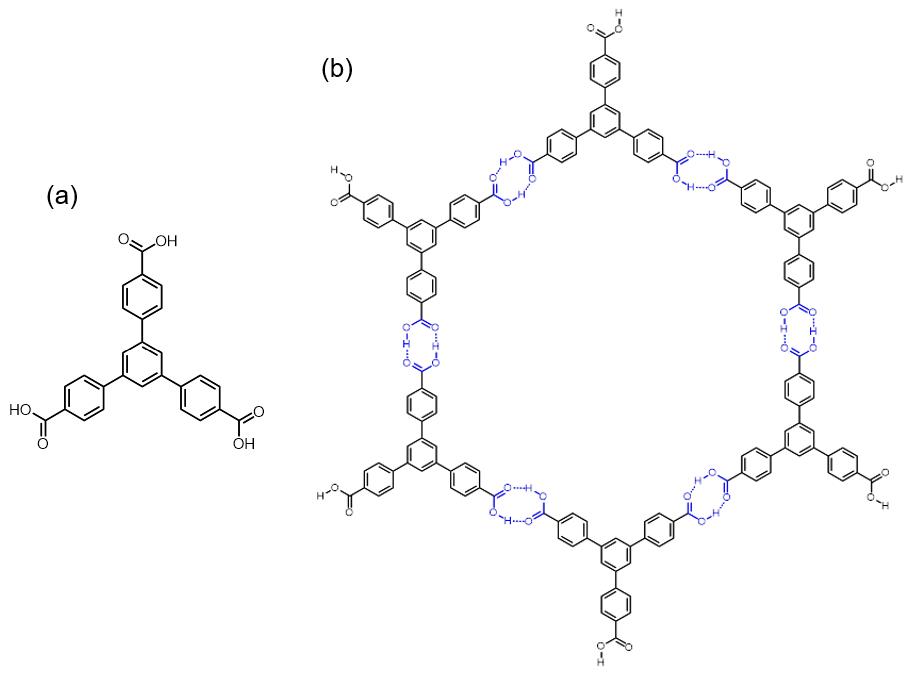
Adamantane-1,3,5,7-tetracarboxylic Acid
1,3,5,7-Adamantanetetracarboxylic acid is an adamantane derivative containing four carboxylic acid groups bonded to each of its four tetrahedral carbon centers. Its tetrahedral symmetry provides applications as a hydrogen-bonded organic framework linker and as a dendrimer core. Preparation In Ermer's 1988 first application of 1,3,5,7-adamantanetetracarboxylic acid as a monomer for a hydrogen-bonded organic framework, it was prepared by alkaline hydrolysis of 1,3,5,7-adamantanetetracarboxamide. Uses 1,3,5,7-Adamantanetetracarboxylic acid (abbreviated as the ADTA linker in reticular chemistry Omar M. Yaghi ( ar, عمر مونّس ياغي; born February 9, 1965) is the James and Neeltje Tretter Chair Professor of Chemistry at the University of California, Berkeley, the Founding Director of the Berkeley Global Science Institute, and an ...) was used in 1988 as one of the first linkers in hydrogen-bonded organic frameworks. A tetrahedral linker, it crystallizes into a five-fold i ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
PubChem
PubChem is a database of chemical molecules and their activities against biological assays. The system is maintained by the National Center for Biotechnology Information (NCBI), a component of the National Library of Medicine, which is part of the United States National Institutes of Health (NIH). PubChem can be accessed for free through a web user interface. Millions of compound structures and descriptive datasets can be freely downloaded via FTP. PubChem contains multiple substance descriptions and small molecules with fewer than 100 atoms and 1,000 bonds. More than 80 database vendors contribute to the growing PubChem database. History PubChem was released in 2004 as a component of the Molecular Libraries Program (MLP) of the NIH. As of November 2015, PubChem contains more than 150 million depositor-provided substance descriptions, 60 million unique chemical structures, and 225 million biological activity test results (from over 1 million assay experiments performed on more t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
ChemSpider
ChemSpider is a database of chemicals. ChemSpider is owned by the Royal Society of Chemistry. Database The database contains information on more than 100 million molecules from over 270 data sources including: * EPA DSSTox * U.S. Food and Drug Administration (FDA) * Human Metabolome Database * Journal of Heterocyclic Chemistry * KEGG * KUMGM * LeadScope * LipidMAPS * Marinlit * MDPI * MICAD * MLSMR * MMDB * MOLI * MTDP * Nanogen * Nature Chemical Biology * NCGC * NIAID * National Institutes of Health (NIH) * NINDS Approved Drug Screening Program * NIST * NIST Chemistry WebBook * NMMLSC * NMRShiftDB * PANACHE * PCMD * PDSP * Peptides * Prous Science Drugs of the Future * QSAR * R&D Chemicals * San Diego Center for Chemical Genomics * SGCOxCompounds, SGCStoCompounds * SMID * Specs * Structural Genomics Consortium * SureChem * Synthon-Lab * Thomson Pharma * Total TOSLab Building-Blocks * UM-BBD * UPCMLD * UsefulChem * Web of Science * xPharm Each chemical is given a unique i ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Journal Of Organic Chemistry
''The Journal of Organic Chemistry'', colloquially known as ''JOC'', is a peer-reviewed scientific journal for original contributions of fundamental research in all branches of theory and practice in organic and bioorganic chemistry. It is published by the publishing arm of the American Chemical Society, with 24 issues per year. According to the ''Journal Citation Reports'', the journal had a 2017 impact factor of 4.805 and it is the journal that received the most cites (100,091 in 2017) in the field of organic chemistry. According to Web of Knowledge (and as December 2012), eleven papers from the journal have received more than 1,000 citations, with the most cited paper having received 7,967 citations. The current editor-in-chief is Scott J. Miller from Yale University. Indexing ''J. Org. Chem.'' is currently indexed in: See also *Organic Letters *Organometallics ''Organometallics'' is a biweekly journal published by the American Chemical Society. Its area of focus is ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
1-Adamantanecarboxylic Acid
1-Adamantanecarboxylic acid is an organic compound with the formula . A white solid, it is the simplest carboxylic acid derivative of adamantane. The compound is notable for its synthesis by carboxylation of adamantane. 1-Adamantanecarboxylic acid is unusual in forming mononuclear transition metal carboxylate complex, tris(carboxylate) coordination complexes of the formula [M(O2CR)3]− (M = Mn, Ni, Co, Zn). References [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Adamantane
Adamantane is an organic compound with a formula C10H16 or, more descriptively, (CH)4(CH2)6. Adamantane molecules can be described as the fusion of three cyclohexane rings. The molecule is both rigid and virtually stress-free. Adamantane is the most stable isomer of C10H16. The spatial arrangement of carbon atoms in the adamantane molecule is the same as in the diamond crystal. This similarity led to the name ''adamantane'', which is derived from the Greek ''adamantinos'' (relating to steel or diamond). It is a white solid with a camphor-like odor. It is the simplest diamondoid. The discovery of adamantane in petroleum in 1933 launched a new field of chemistry dedicated to the synthesis and properties of polyhedral organic compounds. Adamantane derivatives have found practical application as drugs, polymeric materials, and thermally stable lubricants. History and synthesis In 1924, H. Decker suggested the existence of adamantane, which he called decaterpene. The first attempted ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Carboxylic Acid
In organic chemistry, a carboxylic acid is an organic acid that contains a carboxyl group () attached to an R-group. The general formula of a carboxylic acid is or , with R referring to the alkyl, alkenyl, aryl, or other group. Carboxylic acids occur widely. Important examples include the amino acids and fatty acids. Deprotonation of a carboxylic acid gives a carboxylate anion. Examples and nomenclature Carboxylic acids are commonly identified by their trivial names. They at oftentimes have the suffix ''-ic acid''. IUPAC-recommended names also exist; in this system, carboxylic acids have an ''-oic acid'' suffix. For example, butyric acid (C3H7CO2H) is butanoic acid by IUPAC guidelines. For nomenclature of complex molecules containing a carboxylic acid, the carboxyl can be considered position one of the parent chain even if there are other substituents, such as 3-chloropropanoic acid. Alternately, it can be named as a "carboxy" or "carboxylic acid" substituent on another ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrogen-bonded Organic Framework
Hydrogen-bonded organic frameworks (HOFs) are a class of porous polymers formed by hydrogen bonds among molecular monomer units to afford porosity and structural flexibility. There are diverse hydrogen bonding pair choices that could be used in HOFs construction, including identical or nonidentical hydrogen bonding donors and acceptors. For organic groups acting as hydrogen bonding units, species like carboxylic acid, amide, 2,4-diaminotriazine, and imidazole, etc., are commonly used for the formation of hydrogen bonding interaction. Compared with other organic frameworks, like Covalent organic framework, COF and Metal–organic framework, MOF, the binding force of HOFs is relatively weaker, and the activation of HOFs is more difficult than other frameworks, while the reversibility of hydrogen bonds guarantees a high crystallinity of the materials. Though the stability and pore size expansion of HOFs has potential problems, HOFs still show strong potential for applications in differen ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Accounts Of Materials Research .
{{disambig ...
Account may refer to: * Account (bookkeeping) * A report * A bank account ** Deposit account ** Personal account ** Sweep account ** Transaction account * User account, the means by which a user can access a computer system * Customer of a company, used in B2B business. See account manager or account executive Account executive is a role in sales, advertising, marketing, and finance involving intimate understanding of a client company's objectives and products and a professional capability to provide effective advice toward creation of successful promot ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dendrimer
Dendrimers are highly ordered, branched polymeric molecules. Synonymous terms for dendrimer include arborols and cascade molecules. Typically, dendrimers are symmetric about the core, and often adopt a spherical three-dimensional morphology. The word dendron is also encountered frequently. A dendron usually contains a single chemically addressable group called the focal point or core. The difference between dendrons and dendrimers is illustrated in the top figure, but the terms are typically encountered interchangeably. The first dendrimers were made by divergent synthesis approaches by Fritz Vögtle in 1978, R.G. Denkewalter at Allied Corporation in 1981, Donald Tomalia at Dow Chemical in 1983 and in 1985, and by George R. Newkome in 1985. In 1990 a convergent synthetic approach was introduced by Craig Hawker and Jean Fréchet. Dendrimer popularity then greatly increased, resulting in more than 5,000 scientific papers and patents by the year 2005. Properties Dendritic ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Alkaline Hydrolysis
Alkaline hydrolysis, in organic chemistry, usually refers to types of nucleophilic substitution reactions in which the attacking nucleophile is a hydroxide ion. Example In the alkaline hydrolysis of esters and amides the hydroxide ion nucleophile attacks the carbonyl carbon in a nucleophilic acyl substitution reaction. This mechanism is supported by isotope labeling experiments. For example, when ethyl propionate with an oxygen-18 labeled ethoxy group is treated with sodium hydroxide (NaOH), the oxygen-18 is completely absent from the sodium propionate product and is found exclusively in the ethanol formed. Uses The reaction is often used to turn solid organic matter into a liquid form for easier disposal. Drain cleaners take advantage of this method to dissolve hair and fat in pipes. The reaction is also used to dispose of human and other animal remains as an alternative to traditional burial or cremation. See also * Acid hydrolysis * Enzymatic hydrolysis * Saponificati ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Journal Of The American Chemical Society
The ''Journal of the American Chemical Society'' is a weekly peer-reviewed scientific journal that was established in 1879 by the American Chemical Society. The journal has absorbed two other publications in its history, the ''Journal of Analytical and Applied Chemistry'' (July 1893) and the ''American Chemical Journal'' (January 1914). It covers all fields of chemistry. Since 2021, the editor-in-chief is Erick M. Carreira (ETH Zurich). In 2014, the journal moved to a hybrid open access publishing model. Abstracting and indexing The journal is abstracted and indexed in Chemical Abstracts Service, Scopus, EBSCO databases, ProQuest databases, Index Medicus/MEDLINE/PubMed, and the Science Citation Index Expanded. According to the ''Journal Citation Reports'', the journal has a 2021 impact factor of 16.383. Editors-in-chief The following people are or have been editor-in-chief: * 1879–1880 – Hermann Endemann * 1880–1881 – Gideon E. Moore * 1881–1882 – Hermann Endemann ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Reticular Chemistry
Omar M. Yaghi ( ar, عمر مونّس ياغي; born February 9, 1965) is the James and Neeltje Tretter Chair Professor of Chemistry at the University of California, Berkeley, the Founding Director of the Berkeley Global Science Institute, and an elected member of the US National Academy of Sciences. Early life and education Yaghi was born in Amman, Jordan in 1965 to a refugee family, originally from Mandatory Palestine. He grew up in a household with many children, but only had limited access to clean water and without electricity. At the age of 15, he moved to the United States at the encouragement of his father. Although he knew little English, he began classes at Hudson Valley Community College, and later transferred to the University at Albany, SUNY to finish his college degree. He began his graduate studies at University of Illinois, Urbana-Champaign and received his PhD in 1990 under the guidance of Walter G. Klemperer. He was a National Science Foundation Postdoctora ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |