|
Acetoacetylate
Diketene is an organic compound with the molecular formula , and which is sometimes written as . It is formed by dimerization of ketene, . Diketene is a member of the oxetane family. It is used as a reagent in organic chemistry. It is a colorless liquid. Production Ketene is generated by dehydrating acetic acid at 700–750 °C in the presence of triethyl phosphate as a catalyst or by the thermolysis of acetone at in the presence of carbon disulfide as a catalyst. :\begin\\ \ce & \ce & \ce & & \Delta H = +147 \text\\\\ \ce & \ce & \ce\\ \end The dimerization to diketene proceeds spontaneously at room temperature: Reactions Heating or irradiation with UV light regenerates the ketene monomer: :(C2H2O)2 2 CH2CO Alkylated ketenes also dimerize with ease and form substituted diketenes. Diketene readily hydrolyzes in water forming acetoacetic acid. Its half-life in water is approximately 45 min. a 25 °C at . Certain diketenes with two aliphatic chains, such as alkyl ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Organic Compound
In chemistry, organic compounds are generally any chemical compounds that contain carbon-hydrogen or carbon-carbon bonds. Due to carbon's ability to catenate (form chains with other carbon atoms), millions of organic compounds are known. The study of the properties, reactions, and syntheses of organic compounds comprise the discipline known as organic chemistry. For historical reasons, a few classes of carbon-containing compounds (e.g., carbonate salts and cyanide salts), along with a few other exceptions (e.g., carbon dioxide, hydrogen cyanide), are not classified as organic compounds and are considered inorganic. Other than those just named, little consensus exists among chemists on precisely which carbon-containing compounds are excluded, making any rigorous definition of an organic compound elusive. Although organic compounds make up only a small percentage of Earth's crust, they are of central importance because all known life is based on organic compounds. Living t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
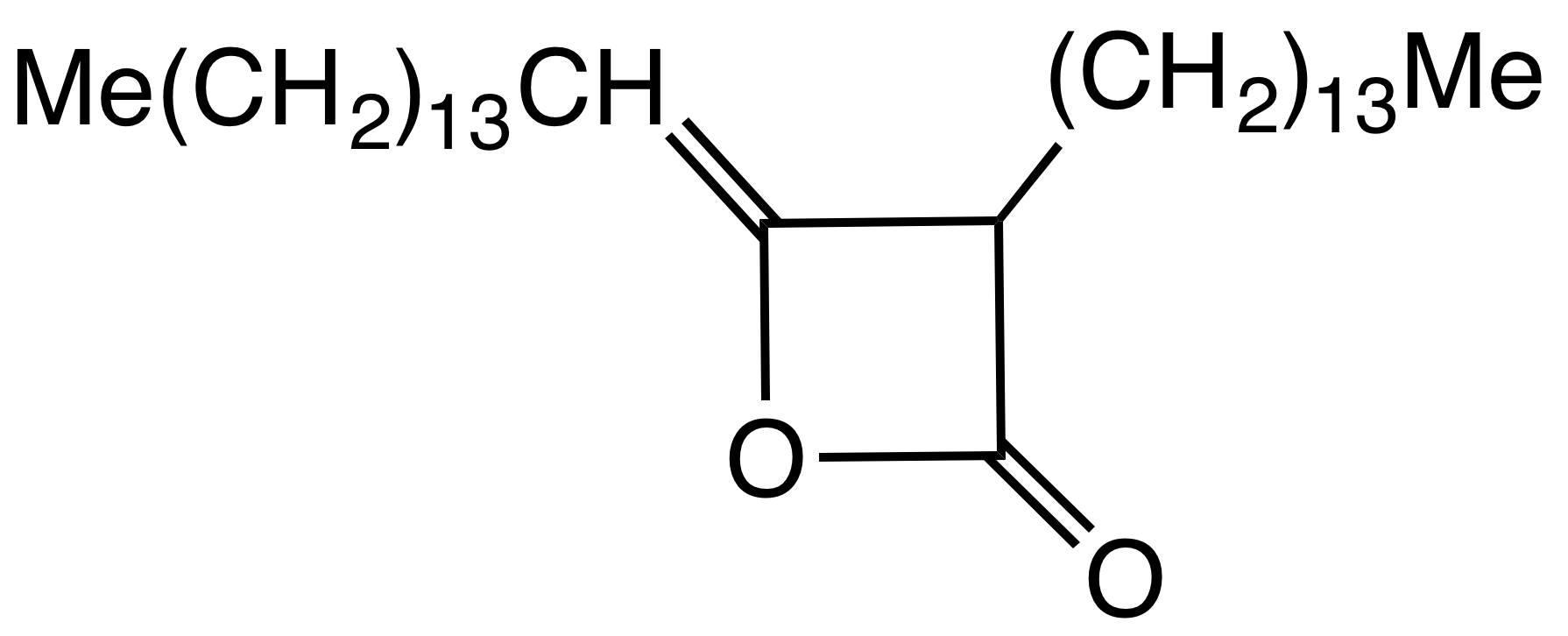
Alkyl Ketene Dimer
Alkyl ketene dimers (AKDs) are a family of organic compounds based on the 4-membered ring system of oxetan-2-one, which is also the central structural element of propiolactone and diketene. Attached to the oxetane ring of technically relevant alkyl ketene dimers there is a C12 – C16 alkyl group in the 3-position and a C13 – C17 alkylidene group in the 4-position. The main application of alkylated ketene dimers is in the sizing of paper and cardboard, as well as in the hydrophobation of cellulose fibers. The products thus modified are distinguished by higher mechanical strengths and less penetration of water, inks or printing inks. AKD's feature hydrophobic alkyl groups extending from a beta-propiolactone ring. A specific example is derived from the dimerization of the ketene of stearic acid. This ketene is generated by pyrolysis of stearoyl chloride. AKD's react with the hydroxyl groups on the cellulose via esterification reaction. The esterification is competitive with hy ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Stearic Acid
Stearic acid ( , ) is a saturated fatty acid with an 18-carbon chain. The IUPAC name is octadecanoic acid. It is a waxy solid and its chemical formula is C17H35CO2H. Its name comes from the Greek word στέαρ "''stéar''", which means tallow. The salts and esters of stearic acid are called stearates. As its ester, stearic acid is one of the most common saturated fatty acids found in nature following palmitic acid.Gunstone, F. D., John L. Harwood, and Albert J. Dijkstra "The Lipid Handbook with Cd-Rom. 3rd ed. Boca Raton: CRC Press, 2007. , The triglyceride derived from three molecules of stearic acid is called stearin. Occurrence and production Stearic acid is more abundant in animal fat (up to 30%) than in vegetable fat (typically <5%). The important exceptions are the foods (34%) and [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sizing
Sizing or size is a substance that is applied to, or incorporated into, other materials—especially papers and textiles—to act as a protective filler or glaze. Sizing is used in papermaking and textile manufacturing to change the absorption and wear characteristics of those materials. Sizing is used for oil-based surface preparation for gilding (sometimes called ''mordant'' in this context). It is used by painters and artists to prepare paper and textile surfaces for some art techniques. Sizing is used in photography to increase the sharpness of a print, to change the glossiness of a print, or for other purposes depending on the type of paper and printing technique. Fibers used in composite materials are treated with various sizing agents to promote adhesion with the matrix material. Sizing is used during paper manufacture to reduce the paper's tendency when dry to absorb liquid, with the goal of allowing inks and paints to remain on the surface of the paper and to dry the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ullmann's Encyclopedia Of Industrial Chemistry
''Ullmann's Encyclopedia of Industrial Chemistry'' is a major reference work related to industrial chemistry by Chemist Fritz Ullmann, first published in 1914, and exclusively in German as "Enzyklopädie der Technischen Chemie" until 1984. History Ullmann's Encyclopedia of Industrial Chemistry is a major reference work related to industrial chemistry by chemist Fritz Ullmann. Its 1st edition was published in German by Fritz Ullmann in 1914. The 4th edition, published 1972 to 1984, already contained 25 volumes. The 5th edition, published 1985 to 1996, was the first version available in English. In 1997, the first online version was published. 2014 marked its centenary. As of 2016, Ullmann's Encyclopedia was in its 7th edition, in 40 volumes including one index volume and more than 1,050 articles (200 more than the 6th edition), approx. 30,000 pages, 22,000 images, 8,000 tables, 19,000 references and 85,000 indices. Editions * 1914–1922: 1st edition in 12 volumes, which can ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
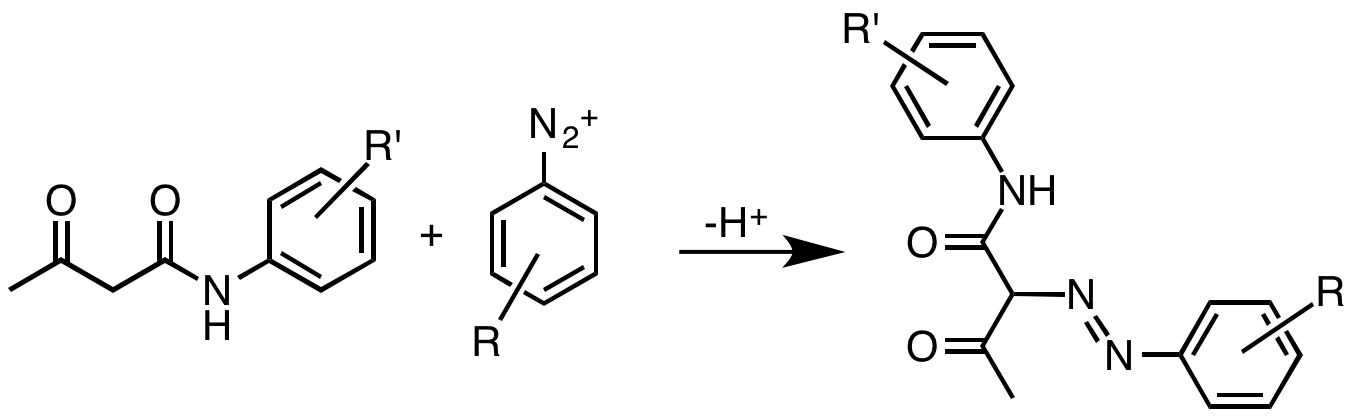
Diarylide Pigment
Diarylide pigments are organic compounds that are used as pigments in inks and related materials. They often are yellow or yellow-green. To some extent, these organic compounds have displaced cadmium sulfide from the market. They exist as yellow powders of low solubility in water.K. Hunger. W. Herbst "Pigments, Organic" in ''Ullmann's Encyclopedia of Industrial Chemistry'', Wiley-VCH, Weinheim, 2012. Production The formation of these pigments involves the reaction of diazotized aromatic diamines (derivatives of benzidine) with coupling components, typically derivatives of acetoacetanilide. By varying both of these components, a wide variety of pigments have been produced. A related family of organic pigments are the simpler arylides, which arise from the coupling of monodiazonium salts with the same coupling partners. The molecular structures of these molecules is more conjugated than represented in the images shown below. The pigments' colors can range from yellow to ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Arylide Yellow
Arylide yellow, also known as Hansa yellow and monoazo yellow, is a family of organic compounds used as pigments. They are primarily used as industrial colorants including plastics, building paints and inks. They are also used in artistic oil paints, acrylics and watercolors. These pigments are usually semi-transparent and range from orange-yellow to yellow-greens. Related organic pigments are the diarylide pigments. Overall, these pigments have partially displaced the toxic cadmium yellow in the marketplace. Painters such as Alexander Calder and Jackson Pollock are known to have employed arylide yellow in their artworks. Colourlex Production The compound is obtained by of |
Indane
Indane or indan is an organic compound with the formula C6H4(CH2)3. It is a colorless liquid hydrocarbon. It is a petrochemical, a bicyclic compound. It occurs at the level of about 0.1% in coal tar. It is usually produced by hydrogenation of indene.Karl Griesbaum, Arno Behr, Dieter Biedenkapp, Heinz-Werner Voges, Dorothea Garbe, Christian Paetz, Gerd Collin, Dieter Mayer, Hartmut Höke "Hydrocarbons" in Ullmann's Encyclopedia of Industrial Chemistry 2002 Wiley-VCH, Weinheim. Reactions Indane can also be converted in a catalytic reactor to other aromatics such as xylene. Indane is used in the synthesis of sulofenur. Derivatives Derivatives include compounds such as 1-methylindane and 2-methylindane (where one methyl group is attached to the five carbon ring), 4-methylindane and 5-methylindane (where one methyl group is attached to the benzene ring), and various dimethylindanes. Other derivatives can be obtained indirectly, e.g. the reaction of diethyl phthalate with ethyl ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Amine
In chemistry, amines (, ) are compounds and functional groups that contain a basic nitrogen atom with a lone pair. Amines are formally derivatives of ammonia (), wherein one or more hydrogen atoms have been replaced by a substituent such as an alkyl or aryl group (these may respectively be called alkylamines and arylamines; amines in which both types of substituent are attached to one nitrogen atom may be called alkylarylamines). Important amines include amino acids, biogenic amines, trimethylamine, and aniline; Inorganic derivatives of ammonia are also called amines, such as monochloramine (). The substituent is called an amino group. Compounds with a nitrogen atom attached to a carbonyl group, thus having the structure , are called amides and have different chemical properties from amines. Classification of amines Amines can be classified according to the nature and number of substituents on nitrogen. Aliphatic amines contain only H and alkyl substituents. A ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Alcohol (chemistry)
In chemistry, an alcohol is a type of organic compound that carries at least one hydroxyl () functional group bound to a saturated carbon atom. The term ''alcohol'' originally referred to the primary alcohol ethanol (ethyl alcohol), which is used as a drug and is the main alcohol present in alcoholic drinks. An important class of alcohols, of which methanol and ethanol are the simplest examples, includes all compounds which conform to the general formula . Simple monoalcohols that are the subject of this article include primary (), secondary () and tertiary () alcohols. The suffix ''-ol'' appears in the IUPAC chemical name of all substances where the hydroxyl group is the functional group with the highest priority. When a higher priority group is present in the compound, the prefix ''hydroxy-'' is used in its IUPAC name. The suffix ''-ol'' in non-IUPAC names (such as paracetamol or cholesterol) also typically indicates that the substance is an alcohol. However, some compou ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |