|
Water Softening
Water softening is the removal of calcium, magnesium, and certain other metal cations in hard water. The resulting soft water requires less soap for the same cleaning effort, as soap is not wasted bonding with calcium ions. Soft water also extends the lifetime of plumbing by reducing or eliminating scale build-up in pipes and fittings. Water softening is usually achieved using lime softening or ion-exchange resins but is increasingly being accomplished using nanofiltration or reverse osmosis membranes. Rationale The presence of certain metal ions like calcium and magnesium, principally as bicarbonates, chlorides, and sulfates, in water causes a variety of problems. Hard water leads to the buildup of limescale, which can foul plumbing, and promote galvanic corrosion. In industrial scale water softening plants, the effluent flow from the re-generation process can precipitate scale that can interfere with sewage systems. The slippery feeling associated with washing in soft water ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Limescale
Limescale is a hard, chalky deposit, consisting mainly of calcium carbonate (CaCO3). It often builds up inside kettles, boilers, and pipework, especially that for hot water. It is also often found as a similar deposit on the inner surfaces of old pipes and other surfaces where "hard water" has flown. Limescale also forms as travertine or tufa in hard water springs. The colour varies from off-white through a range of greys and pink or reddish browns, depending on the other minerals present. Iron compounds give the reddish-browns. In addition to being unsightly and hard to clean, limescale can seriously damage or impair the operation of various plumbing and heating components.Hermann Weingärtner, "Water" in ''Ullmann's Encyclopedia of Industrial Chemistry'', December 2006, Wiley–VCH, Weinheim. Descaling agents are commonly used to remove limescale. Prevention of fouling by scale build-up relies on the technologies of water softening or other water treatment. Chemical compo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
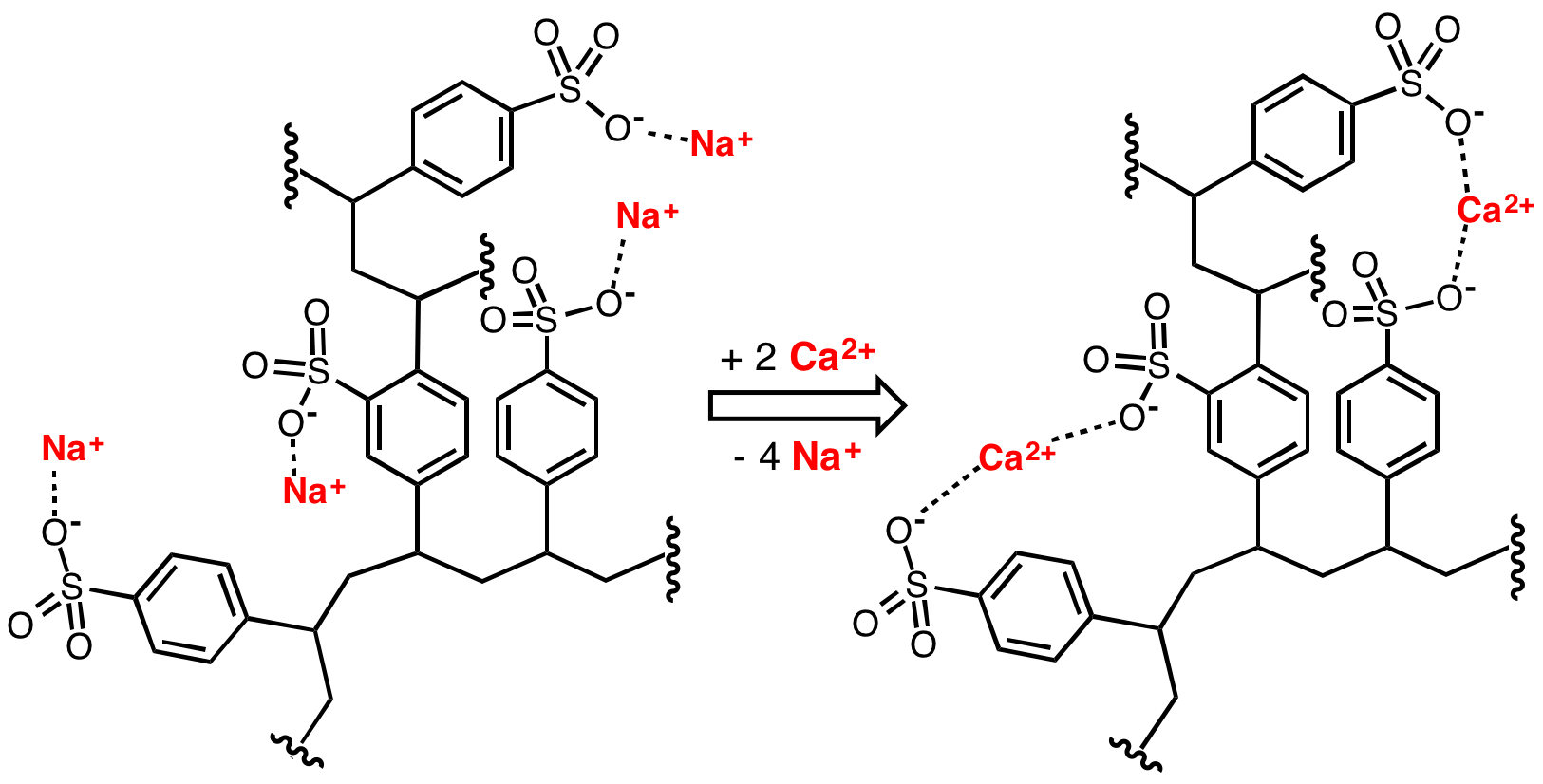
Zeolites
Zeolites are microporous, crystalline aluminosilicate materials commonly used as commercial adsorbents and catalysts. They mainly consist of silicon, aluminium, oxygen, and have the general formula ・y where is either a metal ion or H+. These positive ions can be exchanged for others in a contacting electrolyte solution. exchanged zeolites are particularly useful as solid acid catalysts. The term ''zeolite'' was originally coined in 1756 by Swedish mineralogist Axel Fredrik Cronstedt, who observed that rapidly heating a material, believed to have been stilbite, produced large amounts of steam from water that had been adsorbed by the material. Based on this, he called the material ''zeolite'', from the Greek , meaning "to boil" and , meaning "stone". Zeolites occur naturally but are also produced industrially on a large scale. , 253 unique zeolite frameworks have been identified, and over 40 naturally occurring zeolite frameworks are known. Every new zeolite structure that is obt ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Anionic
An ion () is an atom or molecule with a net electrical charge. The charge of an electron is considered to be negative by convention and this charge is equal and opposite to the charge of a proton, which is considered to be positive by convention. The net charge of an ion is not zero because its total number of electrons is unequal to its total number of protons. A cation is a positively charged ion with fewer electrons than protons while an anion is a negatively charged ion with more electrons than protons. Opposite electric charges are pulled towards one another by electrostatic force, so cations and anions attract each other and readily form ionic compounds. Ions consisting of only a single atom are termed atomic or monatomic ions, while two or more atoms form molecular ions or polyatomic ions. In the case of physical ionization in a fluid (gas or liquid), "ion pairs" are created by spontaneous molecule collisions, where each generated pair consists of a free electron and ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Organic Polymer
A polymer (; Greek '' poly-'', "many" + ''-mer'', "part") is a substance or material consisting of very large molecules called macromolecules, composed of many repeating subunits. Due to their broad spectrum of properties, both synthetic and natural polymers play essential and ubiquitous roles in everyday life. Polymers range from familiar synthetic plastics such as polystyrene to natural biopolymers such as DNA and proteins that are fundamental to biological structure and function. Polymers, both natural and synthetic, are created via polymerization of many small molecules, known as monomers. Their consequently large molecular mass, relative to small molecule compounds, produces unique physical properties including toughness, high elasticity, viscoelasticity, and a tendency to form amorphous and semicrystalline structures rather than crystals. The term "polymer" derives from the Greek word πολύς (''polus'', meaning "many, much") and μέρος (''meros'', meani ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ion-exchange Resin
An ion-exchange resin or ion-exchange polymer is a resin or polymer that acts as a medium for ion exchange. It is an insoluble matrix (or support structure) normally in the form of small (0.25–1.43 mm radius) microbeads, usually white or yellowish, fabricated from an organic polymer substrate. The beads are typically porous, providing a large surface area on and inside them where the trapping of ions occurs along with the accompanying release of other ions, and thus the process is called ion exchange. There are multiple types of ion-exchange resin. Most commercial resins are made of polystyrene sulfonate.François Dardel and Thomas V. Arden "Ion Exchangers" in Ullmann's Encyclopedia of Industrial Chemistry, 2008, Wiley-VCH, Weinheim. . Ion-exchange resins are widely used in different separation, purification, and decontamination processes. The most common examples are water softening and water purification. In many cases ion-exchange resins were introduced in such proce ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ion Exchange Resin Beads
An ion () is an atom or molecule with a net electric charge, electrical charge. The charge of an electron is considered to be negative by convention and this charge is equal and opposite to the charge of a proton, which is considered to be positive by convention. The net charge of an ion is not zero because its total number of electrons is unequal to its total number of protons. A cation is a positively charged ion with fewer electrons than protons while an anion is a negatively charged ion with more electrons than protons. Opposite electric charges are pulled towards one another by electrostatic force, so cations and anions attract each other and readily form ionic compounds. Ions consisting of only a single atom are termed atomic or monatomic ions, while two or more atoms form molecular ions or polyatomic ions. In the case of physical ionization in a fluid (gas or liquid), "ion pairs" are created by spontaneous molecule collisions, where each generated pair consists of a fr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
American National Standards Institute
The American National Standards Institute (ANSI ) is a private non-profit organization that oversees the development of voluntary consensus standards for products, services, processes, systems, and personnel in the United States. The organization also coordinates U.S. standards with international standards so that American products can be used worldwide. ANSI accredits standards that are developed by representatives of other standards organizations, government agencies, consumer groups, companies, and others. These standards ensure that the characteristics and performance of products are consistent, that people use the same definitions and terms, and that products are tested the same way. ANSI also accredits organizations that carry out product or personnel certification in accordance with requirements defined in international standards. The organization's headquarters are in Washington, D.C. ANSI's operations office is located in New York City. The ANSI annual operating b ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sodium
Sodium is a chemical element with the symbol Na (from Latin ''natrium'') and atomic number 11. It is a soft, silvery-white, highly reactive metal. Sodium is an alkali metal, being in group 1 of the periodic table. Its only stable isotope is 23Na. The free metal does not occur in nature, and must be prepared from compounds. Sodium is the sixth most abundant element in the Earth's crust and exists in numerous minerals such as feldspars, sodalite, and halite (NaCl). Many salts of sodium are highly water-soluble: sodium ions have been leached by the action of water from the Earth's minerals over eons, and thus sodium and chlorine are the most common dissolved elements by weight in the oceans. Sodium was first isolated by Humphry Davy in 1807 by the electrolysis of sodium hydroxide. Among many other useful sodium compounds, sodium hydroxide (lye) is used in soap manufacture, and sodium chloride (edible salt) is a de-icing agent and a nutrient for animals including h ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chelation
Chelation is a type of bonding of ions and molecules to metal ions. It involves the formation or presence of two or more separate coordinate bonds between a Denticity, polydentate (multiple bonded) ligand and a single central metal atom. These ligands are called chelants, chelators, chelating agents, or sequestering agents. They are usually organic compounds, but this is not a necessity, as in the case of zinc and its use as a maintenance therapy to prevent the absorption of copper in people with Wilson's disease. Chelation is useful in applications such as providing nutritional supplements, in chelation therapy to remove toxic metals from the body, as contrast medium, contrast agents in MRI, MRI scanning, in manufacturing using homogeneous catalysts, in chemical water treatment to assist in the removal of metals, and in fertilizers. Chelate effect The chelate effect is the greater affinity of chelating ligands for a metal ion than that of similar nonchelating (monodentate ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Soap Scum
Soap scum or lime soap is the white solid composed of calcium stearate, magnesium stearate, and similar alkaline earth metal derivatives of fatty acids. These materials result from the addition of soap and other anionic surfactants to hard water. Hard water contains calcium and magnesium ions, which react with the surfactant anion to give these metallic or lime soaps.{{Ullmann, author=Angelo Nora, Alfred Szczepanek, Gunther Koenen, title=Metallic Soaps, year=2005, doi=10.1002/14356007.a16_361 :2 C17H35COO−Na+ + Ca2+ → (C17H35COO)2Ca + 2 Na+ In this reaction, the sodium cation in soap is replaced by calcium to form calcium stearate. Lime soaps build deposits on fibres, washing machines, and sinks. Synthetic surfactants are less susceptible to the effects of hard water. Most detergents contain builders that prevent the formation of lime soaps. See also * Water softening * Tadelakt, a form of lime-soap-based waterproof plaster * Qadad ''Qadad'' ( ''qaḍāḍ'') or '' ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |