|
Vinyl Propionate
Vinyl propionate is the organic compound with the formula CH3CH2CO2CH=CH2. This colorless liquid is the ester of propionic acid and vinyl alcohol. It is used to produce poly(vinyl propionate) as well as copolymers with acrylate esters, vinyl chloride, and vinyl acetate, some of which are used in paints. The compound resembles vinyl acetate Vinyl acetate is an organic compound with the formula CH3CO2CH=CH2. This colorless liquid is the precursor to polyvinyl acetate and ethene-vinyl acetate copolymers, important industrial polymers. Production The worldwide production capacity of v ....{{cite encyclopedia, author=G. Roscher, title=Ullmann's Encyclopedia of Industrial Chemistry, chapter=Vinyl Esters, encyclopedia=Ullmann's Encyclopedia of Chemical Technology, year=2007, publisher=Wiley-VCH, location=Weinheim, doi=10.1002/14356007.a27_419, isbn=978-3527306732 Since vinyl alcohol is not available, vinyl propionate is produced by the addition of propionic acid to acetylene. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Organic Compound
In chemistry, organic compounds are generally any chemical compounds that contain carbon-hydrogen or carbon-carbon bonds. Due to carbon's ability to catenate (form chains with other carbon atoms), millions of organic compounds are known. The study of the properties, reactions, and syntheses of organic compounds comprise the discipline known as organic chemistry. For historical reasons, a few classes of carbon-containing compounds (e.g., carbonate salts and cyanide salts), along with a few other exceptions (e.g., carbon dioxide, hydrogen cyanide), are not classified as organic compounds and are considered inorganic. Other than those just named, little consensus exists among chemists on precisely which carbon-containing compounds are excluded, making any rigorous definition of an organic compound elusive. Although organic compounds make up only a small percentage of Earth's crust, they are of central importance because all known life is based on organic compounds. Living t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
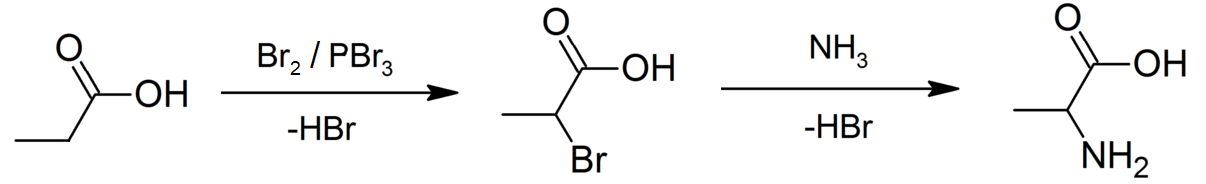
Propionic Acid
Propionic acid (, from the Greek words πρῶτος : ''prōtos'', meaning "first", and πίων : ''píōn'', meaning "fat"; also known as propanoic acid) is a naturally occurring carboxylic acid with chemical formula CH3CH2CO2H. It is a liquid with a pungent and unpleasant smell somewhat resembling body odor. The anion CH3CH2CO2− as well as the salts and esters of propionic acid are known as propionates or propanoates. History Propionic acid was first described in 1844 by Johann Gottlieb, who found it among the degradation products of sugar. Over the next few years, other chemists produced propionic acid by different means, none of them realizing they were producing the same substance. In 1847, French chemist Jean-Baptiste Dumas established all the acids to be the same compound, which he called propionic acid, from the Greek words πρῶτος (prōtos), meaning ''first'', and πίων (piōn), meaning ''fat'', because it is the smallest H(CH2)''n''COOH acid that exhib ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Vinyl Alcohol
Vinyl alcohol, also called ethenol (IUPAC name; not ethanol), is the simplest enol. With the formula , it is a labile compound that converts to acetaldehyde. It is not a precursor to polyvinyl alcohol. Synthesis Vinyl alcohol can be formed by the pyrolytic elimination of water from ethylene glycol at a temperature of 900 °C and low pressure. Tautomerization of vinyl alcohol to acetaldehyde Under normal conditions, vinyl alcohol converts (tautomerizes) to acetaldehyde: : At room temperature, acetaldehyde () is more stable than vinyl alcohol () by 42.7 kJ/mol: : Δ''H''298,g = −42.7 kJ/mol The uncatalyzed keto–enol tautomerism by a 1,3-hydrogen migration is forbidden by the Woodward–Hoffmann rules and therefore has a high activation barrier and is not a significant pathway at or near room temperature. However, even trace amounts of acids or bases (including water) can catalyze the reaction. Even with rigorous precautions to minimize adventitious moisture ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Copolymer
In polymer chemistry, a copolymer is a polymer derived from more than one species of monomer. The polymerization of monomers into copolymers is called copolymerization. Copolymers obtained from the copolymerization of two monomer species are sometimes called ''bipolymers''. Those obtained from three and four monomers are called ''terpolymers'' and ''quaterpolymers'', respectively. Copolymers can be characterized by a variety of techniques such as NMR spectroscopy and size-exclusion chromatography to determine the molecular size, weight, properties, and composition of the material. Commercial copolymers include acrylonitrile butadiene styrene (ABS), styrene/butadiene co-polymer (SBR), nitrile rubber, styrene-acrylonitrile, styrene-isoprene-styrene (SIS) and ethylene-vinyl acetate, all of which are formed by chain-growth polymerization. Another production mechanism is step-growth polymerization, which is used to produce the nylon-12/6/66 copolymer of nylon 12, nylon 6 and nylon ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Acrylate
Acrylates (IUPAC: prop-2-enoates) are the salts, esters, and conjugate bases of acrylic acid. The acrylate ion is the anion C H2=CHC OO−. Often, acrylate refers to esters of acrylic acid, the most common member being methyl acrylate. These acrylates contain vinyl groups. These compounds are of interest because they are bifunctional: the vinyl group is susceptible to polymerization and the carboxylate group carries myriad functionalities. Modified acrylates are also numerous, some examples being methacrylates (CH2=C(CH3)CO2R) and cyanoacrylates (CH2=C(CN)CO2R). Acrylate can also refer to polyacrylates prepared through the polymerization of the vinyl groups of acrylate monomers. File:Acrylate-anion.svg, The acrylate anion File:Trimethylolpropane triacrylate.svg, Trimethylolpropane triacrylate (TMPTA), a trifunctional acrylate ester File:Methylacrylat.svg, Methyl acrylate, an acrylic ester File:Hexandioldiacrylat.svg, Hexandiol diacrylate, a bifunctional acrylate File:Methyl-meth ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Vinyl Chloride
Vinyl chloride is an organochloride with the formula H2C=CHCl. It is also called vinyl chloride monomer (VCM) or chloroethene. This colorless compound is an important industrial chemical chiefly used to produce the polymer polyvinyl chloride (PVC). About 13 million metric tonne are produced annually. VCM is among the top twenty largest petrochemicals (petroleum-derived chemicals) in world production. The United States currently remains the largest VCM manufacturing region because of its low-production-cost position in chlorine and ethylene raw materials. China is also a large manufacturer and one of the largest consumers of VCM. Vinyl chloride is a gas with a sweet odor. It is highly toxic, flammable, and carcinogenic. It can be formed in the environment when soil organisms break down chlorinated solvents. Vinyl chloride that is released by industries or formed by the breakdown of other chlorinated chemicals can enter the air and drinking water supplies. Vinyl chloride is a common ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Vinyl Acetate
Vinyl acetate is an organic compound with the formula CH3CO2CH=CH2. This colorless liquid is the precursor to polyvinyl acetate and ethene-vinyl acetate copolymers, important industrial polymers. Production The worldwide production capacity of vinyl acetate was estimated at 6,969,000 tonnes/year in 2007, with most capacity concentrated in the United States (1,585,000 all in Texas), China (1,261,000), Japan (725,000) and Taiwan (650,000). The average list price for 2008 was US$1600/tonne. Celanese is the largest producer (ca 25% of the worldwide capacity), while other significant producers include China Petrochemical Corporation (7%), Chang Chun Group (6%), and LyondellBasell (5%). It is a key ingredient in furniture glue. Preparation Vinyl acetate is the acetate ester of vinyl alcohol. Since vinyl alcohol is highly unstable (with respect to acetaldehyde), the preparation of vinyl acetate is more complex than the synthesis of other acetate esters. The major industrial route i ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Acetylene
Acetylene (systematic name: ethyne) is the chemical compound with the formula and structure . It is a hydrocarbon and the simplest alkyne. This colorless gas is widely used as a fuel and a chemical building block. It is unstable in its pure form and thus is usually handled as a solution. Pure acetylene is odorless, but commercial grades usually have a marked odor due to impurities such as divinyl sulfide and phosphine.Compressed Gas Association (1995Material Safety and Data Sheet – Acetylene As an alkyne, acetylene is unsaturated because its two carbon atoms are bonded together in a triple bond. The carbon–carbon triple bond places all four atoms in the same straight line, with CCH bond angles of 180°. Discovery Acetylene was discovered in 1836 by Edmund Davy, who identified it as a "new carburet of hydrogen". It was an accidental discovery while attempting to isolate potassium metal. By heating potassium carbonate with carbon at very high temperatures, he produced a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Monomers
In chemistry, a monomer ( ; ''mono-'', "one" + '' -mer'', "part") is a molecule that can react together with other monomer molecules to form a larger polymer chain or three-dimensional network in a process called polymerization. Classification Monomers can be classified in many ways. They can be subdivided into two broad classes, depending on the kind of the polymer that they form. Monomers that participate in condensation polymerization have a different stoichiometry than monomers that participate in addition polymerization: : Other classifications include: *natural vs synthetic monomers, e.g. glycine vs caprolactam, respectively *polar vs nonpolar monomers, e.g. vinyl acetate vs ethylene, respectively *cyclic vs linear, e.g. ethylene oxide vs ethylene glycol, respectively The polymerization of one kind of monomer gives a homopolymer. Many polymers are copolymers, meaning that they are derived from two different monomers. In the case of condensation polymerizations, the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Esters
In chemistry, an ester is a compound derived from an oxoacid (organic or inorganic) in which at least one hydroxyl group () is replaced by an alkoxy group (), as in the substitution reaction of a carboxylic acid and an alcohol. Glycerides are fatty acid esters of glycerol; they are important in biology, being one of the main classes of lipids and comprising the bulk of animal fats and vegetable oils. Esters typically have a pleasant smell; those of low molecular weight are commonly used as fragrances and are found in essential oils and pheromones. They perform as high-grade solvents for a broad array of plastics, plasticizers, resins, and lacquers, and are one of the largest classes of synthetic lubricants on the commercial market. Polyesters are important plastics, with monomers linked by ester moieties. Phosphoesters form the backbone of DNA molecules. Nitrate esters, such as nitroglycerin, are known for their explosive properties. '' Nomenclature Etymology The ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Commodity Chemicals
Commodity chemicals (or bulk commodities or bulk chemicals) are a group of chemicals that are made on a very large scale to satisfy global markets. The average prices of commodity chemicals are regularly published in the chemical trade magazines and web sites such aChemical WeekanICIS There have been several studies of the scale and complexity of this market for example in the USA. Commodity chemicals are a sub-sector of the chemical industry (other sub sectors are fine chemicals, specialty chemicals, inorganic chemicals, petrochemicals, pharmaceuticals, renewable energy (e.g. biofuels) and materials (e.g. biopolymers)) Commodity chemicals are differentiated primarily by the bulk of their manufacture. Types Chemical compounds are often classified into two classes, inorganic and organic. Inorganic chemicals * aluminium sulfate * ammonia * ammonium nitrate * ammonium sulfate * carbon black * chlorine * diammonium phosphate * monoammonium phosphate * hydrochloric acid * hydrogen fl ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |