|
Trimethyl Aluminium
Trimethylaluminium is one of the simplest examples of an Organoaluminium chemistry, organoaluminium compound. Despite its name it has the Chemical formula, formula aluminum, Al2(methyl, CH3)6 (abbreviated as Al2Me6 or TMA), as it exists as a Dimer (chemistry), dimer. This colorless liquid is pyrophoric. It is an industrially important compound, closely related to triethylaluminium. Structure and bonding The structure and bonding in Al2R6 and diborane are analogous (R = alkyl). In Al2Me6, the Al-C(terminal) and Al-C(bridging) distances are 1.97 and 2.14 Å, respectively. The Al center is tetrahedral. The carbon atoms of the bridging methyl groups are each surrounded by five neighbors: three hydrogen atoms and two aluminium atoms. The methyl groups interchange readily intramolecularly. At higher temperatures, the dimer Cracking (chemistry), cracks into monomeric AlMe3. Synthesis TMA is prepared via a two-step process that can be summarized as follows: :2 Al + 6 Methyl chloride, CH ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dimer (chemistry)
A dimer () ('' di-'', "two" + ''-mer'', "parts") is an oligomer consisting of two monomers joined by bonds that can be either strong or weak, covalent or intermolecular. Dimers also have significant implications in polymer chemistry, inorganic chemistry, and biochemistry. The term ''homodimer'' is used when the two molecules are identical (e.g. A–A) and ''heterodimer'' when they are not (e.g. A–B). The reverse of dimerization is often called dissociation. When two oppositely charged ions associate into dimers, they are referred to as ''Bjerrum pairs'', after Niels Bjerrum. Noncovalent dimers Anhydrous carboxylic acids form dimers by hydrogen bonding of the acidic hydrogen and the carbonyl oxygen. For example, acetic acid forms a dimer in the gas phase, where the monomer units are held together by hydrogen bonds. Under special conditions, most OH-containing molecules form dimers, e.g. the water dimer. Excimers and exciplexes are excited structures with a short lifetime. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Methylaluminoxane
Methylaluminoxane, commonly called MAO, is a mixture of organoaluminium compounds with the approximate formula (Al(CH3)O)''n''. It is usually encountered as a solution in (aromatic) solvents, commonly toluene but also xylene, cumene, or mesitylene, Used in large excess, it activates precatalysts for alkene polymerization. Preparation and structure MAO is prepared by the incomplete hydrolysis of trimethylaluminium, as indicated by this idealized equation :''n'' Al(CH3)3 + ''n'' H2O → (Al(CH3)O)''n'' + 2''n'' CH4 Diverse mechanisms have been proposed for the formation of MAO. Well defined analogues of MAO can be generated with tert-butyl substituents. Uses MAO is well known as catalyst activator for olefin polymerizations by homogeneous catalysis. In traditional Ziegler–Natta catalysis, supported titanium trichloride is activated by treatment with trimethylaluminium (TMA). TMA only weakly activates homogeneous precatalysts, such as zirconacene dichloride. In the mid- ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
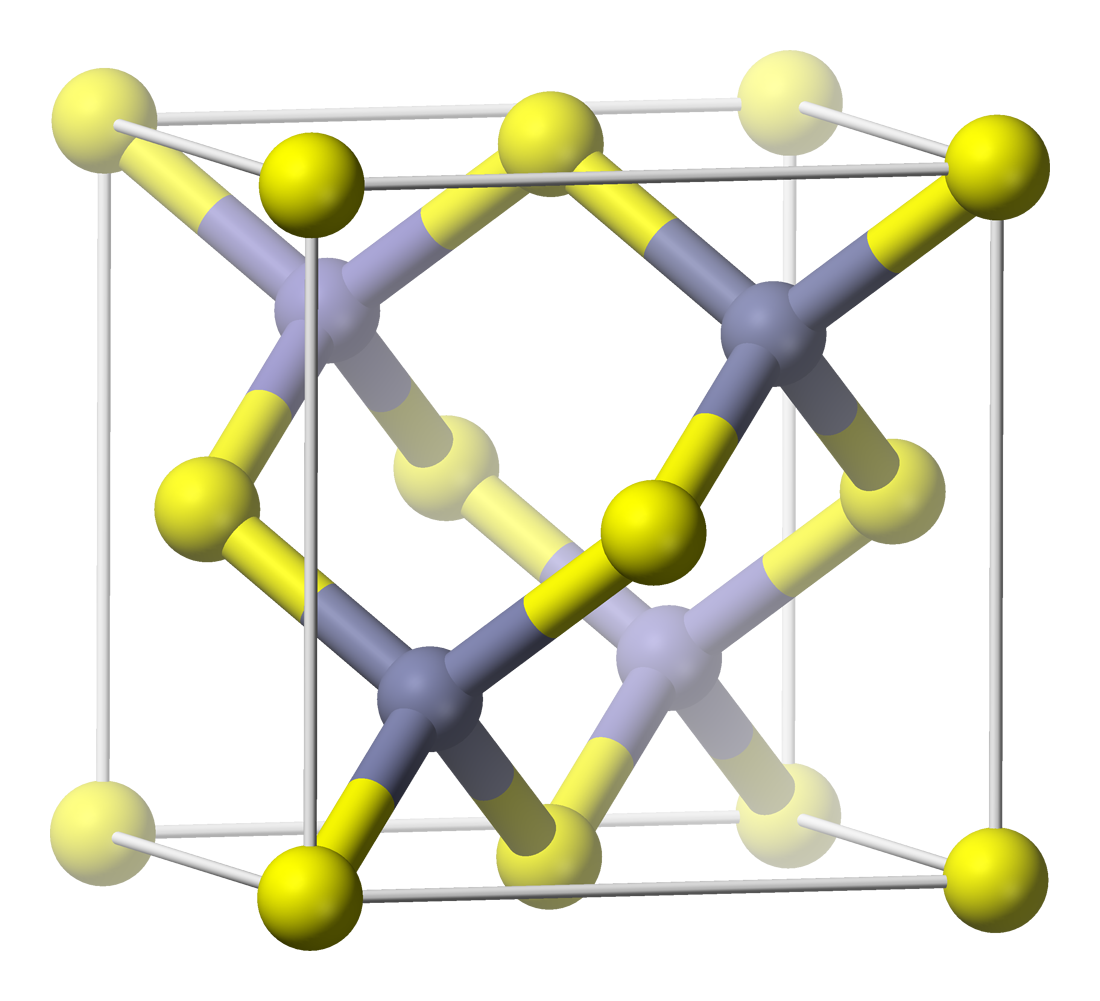
Aluminium Indium Gallium Phosphide
Aluminium (aluminum in American and Canadian English) is a chemical element with the symbol Al and atomic number 13. Aluminium has a density lower than those of other common metals, at approximately one third that of steel. It has a great affinity towards oxygen, and forms a protective layer of oxide on the surface when exposed to air. Aluminium visually resembles silver, both in its color and in its great ability to reflect light. It is soft, non-magnetic and ductile. It has one stable isotope, 27Al; this isotope is very common, making aluminium the twelfth most common element in the Universe. The radioactivity of 26Al is used in radiodating. Chemically, aluminium is a post-transition metal in the boron group; as is common for the group, aluminium forms compounds primarily in the +3 oxidation state. The aluminium cation Al3+ is small and highly charged; as such, it is polarizing, and Chemical bond, bonds aluminium forms tend towards Covalent bond, covalency. The ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Aluminium Indium Gallium Arsenide
Aluminium (aluminum in American and Canadian English) is a chemical element with the symbol Al and atomic number 13. Aluminium has a density lower than those of other common metals, at approximately one third that of steel. It has a great affinity towards oxygen, and forms a protective layer of oxide on the surface when exposed to air. Aluminium visually resembles silver, both in its color and in its great ability to reflect light. It is soft, non-magnetic and ductile. It has one stable isotope, 27Al; this isotope is very common, making aluminium the twelfth most common element in the Universe. The radioactivity of 26Al is used in radiodating. Chemically, aluminium is a post-transition metal in the boron group; as is common for the group, aluminium forms compounds primarily in the +3 oxidation state. The aluminium cation Al3+ is small and highly charged; as such, it is polarizing, and bonds aluminium forms tend towards covalency. The strong affinity towards oxyg ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Aluminium Gallium Arsenide
Aluminium gallium arsenide (also gallium aluminium arsenide) ( Alx Ga1−x As) is a semiconductor material with very nearly the same lattice constant as GaAs, but a larger bandgap. The ''x'' in the formula above is a number between 0 and 1 - this indicates an arbitrary alloy between GaAs and AlAs. The chemical formula ''AlGaAs'' should be considered an abbreviated form of the above, rather than any particular ratio. The bandgap varies between 1.42 eV (GaAs) and 2.16 eV (AlAs). For x < 0.4, the bandgap is direct. The is related with the bandgap via the and varies between 2 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Aluminium Antimonide
Aluminium antimonide (AlSb) is a semiconductor of the group III-V family containing aluminium and antimony. The lattice constant is 0.61 nm. The indirect bandgap is approximately 1.6 electron volt, eV at 300 K, whereas the direct band gap is 2.22 eV. Its electron mobility is 200 cm²·V−1·s−1 and hole mobility 400 cm²·V−1·s−1 at 300 K. Its refractive index is 3.3 at a wavelength of 2 μm, and its dielectric constant is 10.9 at microwave frequencies.K Seeger and E Schonherr "Microwave dielectric constant of aluminium antimonide" Semicond. Sci. Technol. 6 (1991) 301 AlSb can be chemical reaction, reacted with other III-V materials to produce ternary materials including aluminium indium antimonide, AlInSb, AlGaSb and AlAsSb. Aluminum antimonide is rather flammable because of the reducing tendency of the antimonide (Sb3−) ion. It burns to produce aluminum oxide and antimony trioxide. See also * Gallium antimonide * Indium antimonide ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Aluminium Phosphide
Aluminium phosphide is a highly toxic inorganic compound with the chemical formula AlP, used as a wide band gap semiconductor and a fumigant. This colorless solid is generally sold as a grey-green-yellow powder due to the presence of impurities arising from hydrolysis and oxidation. Properties AlP crystals are dark grey to dark yellow in color and have a zincblende crystal structure with a lattice constant of 5.4510 Å at 300 K. They are thermodynamically stable up to . Aluminium phosphide reacts with water or acids to release phosphine: :AlP + 3 H2O → Al(OH)3 + PH3 :AlP + 3 H+ → Al3+ + PH3 Preparation AlP is synthesized by combination of the elements: : 4Al + P4 → 4AlP : Caution must be taken to avoid exposing the AlP to any sources of moisture, as this generates toxic phosphine gas. Phosphine also poses fire hazards, as it is a dangerous pyrophoric compound, igniting easily in air. Uses Pesticide AlP is used as a rodenticide, insecticide, and fumigant for st ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Aluminium Nitride
Aluminium nitride ( Al N) is a solid nitride of aluminium. It has a high thermal conductivity of up to 321 W/(m·K) and is an electrical insulator. Its wurtzite phase (w-AlN) has a band gap of ~6 eV at room temperature and has a potential application in optoelectronics operating at deep ultraviolet frequencies. History and physical properties AlN was first synthesized in 1862 by F. Briegleb and A. Geuther. AlN, in the pure (undoped) state has an electrical conductivity of 10−11–10−13 Ω−1⋅cm−1, rising to 10−5–10−6 Ω−1⋅cm−1 when doped. Electrical breakdown occurs at a field of 1.2–1.8 V/mm (dielectric strength). The material exists primarily in the hexagonal wurtzite crystal structure, but also has a metastable cubic zincblende phase, which is synthesized primarily in the form of thin films. It is predicted that the cubic phase of AlN (zb-AlN) can exhibit superconductivity at high pressures. In AlN wurtzite crystal structure, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Aluminium Arsenide
Aluminium arsenide () is a semiconductor material with almost the same lattice constant as gallium arsenide and aluminium gallium arsenide and wider band gap than gallium arsenide. (AlAs) can form a superlattice with gallium arsenide ( GaAs) which results in its semiconductor properties. Because GaAs and AlAs have almost the same lattice constant, the layers have very little induced strain, which allows them to be grown almost arbitrarily thick. This allows for extremely high performance high electron mobility, HEMT transistors, and other quantum well devices. Properties It has the following properties: *Thermal expansion coefficient 5 µm/(°C*m) *Debye temperature 417 K * Microhardness 5.0 GPa (50 g load) *Number of atoms in 1 cm3: (4.42-0.17x)·1022Dierks, S"Aluminum Arsenide - Material Safety Data". The Fitzgerald Group, MIT, 1994. *Bulk modulus (7.55+0.26x)·1011 dyn cm−2 *Hardness on the Mohs scale: ~ 5 *Insolubility in H2O Uses Aluminium arsenide is a III-V comp ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Compound Semiconductor
Semiconductor materials are nominally small band gap insulators. The defining property of a semiconductor material is that it can be compromised by doping it with impurities that alter its electronic properties in a controllable way. Because of their application in the computer and photovoltaic industry—in devices such as transistors, lasers, and solar cells—the search for new semiconductor materials and the improvement of existing materials is an important field of study in materials science. Most commonly used semiconductor materials are crystalline inorganic solids. These materials are classified according to the periodic table groups of their constituent atoms. Different semiconductor materials differ in their properties. Thus, in comparison with silicon, compound semiconductors have both advantages and disadvantages. For example, gallium arsenide (GaAs) has six times higher electron mobility than silicon, which allows faster operation; wider band gap, which allows op ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
MOVPE
Metalorganic vapour-phase epitaxy (MOVPE), also known as organometallic vapour-phase epitaxy (OMVPE) or metalorganic chemical vapour deposition (MOCVD), is a chemical vapour deposition method used to produce single- or polycrystalline thin films. It is a process for growing crystalline layers to create complex semiconductor multilayer structures. In contrast to molecular-beam epitaxy (MBE), the growth of crystals is by chemical reaction and not physical deposition. This takes place not in vacuum, but from the gas phase at moderate pressures (10 to 760 Torr). As such, this technique is preferred for the formation of devices incorporating thermodynamically metastable alloys, and it has become a major process in the manufacture of optoelectronics, such as Light-emitting diodes. It was invented in 1968 at North American Aviation (later Rockwell International) Science Center by Harold M. Manasevit. Basic principles In MOCVD ultrapure precursor gases are injected into a reactor, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Metalorganic Vapour Phase Epitaxy
Metalorganic vapour-phase epitaxy (MOVPE), also known as organometallic vapour-phase epitaxy (OMVPE) or metalorganic chemical vapour deposition (MOCVD), is a chemical vapour deposition method used to produce single- or polycrystalline thin films. It is a process for growing crystalline layers to create complex semiconductor multilayer structures. In contrast to molecular-beam epitaxy (MBE), the growth of crystals is by chemical reaction and not physical deposition. This takes place not in vacuum, but from the gas phase at moderate pressures (10 to 760 Torr). As such, this technique is preferred for the formation of devices incorporating thermodynamically metastable alloys, and it has become a major process in the manufacture of optoelectronics, such as Light-emitting diodes. It was invented in 1968 at North American Aviation (later Rockwell International) Science Center by Harold M. Manasevit. Basic principles In MOCVD ultrapure precursor gases are injected into a reactor, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |