|
Tropone
Tropone or 2,4,6-cycloheptatrien-1-one is an organic compound with some importance in organic chemistry as a non-benzenoid aromatic. The compound consists of a ring of seven carbon atoms with three conjugated alkene groups and a ketone group. The related compound tropolone (2-hydroxy-2,4,6-cycloheptatrien-1-one) has an additional alcohol (or an enol including the double bond) group next to the ketone. Tropones are uncommon in natural products, with the notable exception of the 2-hydroxyl derivatives, which are called tropolones. Tropone has been known since 1951 and is also called ''cycloheptatrienylium oxide''. The name tropolone was coined by M. J. S. Dewar in 1945 in connection to perceived aromatic properties. Properties Dewar in 1945 proposed that tropones could have aromatic properties. The carbonyl group is more polarized as a result of the triene ring, giving a partial positive charge on the carbon atom (A) and a partial negative charge on oxygen. In an extreme case, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cycloheptatriene
Cycloheptatriene (CHT) is an organic compound with the formula C7H8. It is a closed ring of seven carbon atoms joined by three double bonds (as the name implies) and four single bonds. This colourless liquid has been of recurring theoretical interest in organic chemistry. It is a ligand in organometallic chemistry and a building block in organic synthesis. Cycloheptatriene is not aromatic, as reflected by the nonplanarity of the methylene bridge (-CH2-) with respect to the other atoms; however the related tropylium cation is. Synthesis Albert Ladenburg first generated cycloheptatriene in 1881 by the decomposition of tropine. The structure was finally proven by the synthesis of Richard Willstätter in 1901. This synthesis started from cycloheptanone and established the seven membered ring structure of the compound. Cycloheptatriene can be obtained in the laboratory by photochemical reaction of benzene with diazomethane or the pyrolysis of the adduct of cyclohexene and ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tropolone
Tropolone is an organic compound with the chemical formula . It is a pale yellow solid that is soluble in organic solvents. The compound has been of interest to research chemists because of its unusual electronic structure and its role as a ligand precursor. Although not usually prepared from tropone, it can be viewed as its derivative with a hydroxyl group in the 2-position. Synthesis and reactions Many methods have been described for the synthesis of tropolone. One involves bromination of 1,2-cycloheptanedione with ''N''-bromosuccinimide followed by dehydrohalogenation at elevated temperatures, while another uses acyloin condensation of the ethyl ester of pimelic acid the acyloin again followed by oxidation by bromine. : An alternate route is a +2cycloaddition of cyclopentadiene with a ketene to give a bicyclo .2.0eptyl structure, followed by hydrolysis and breakage of the fusion bond to give the single ring: : Thy hydroxyl group of tropolone is acidic, having a p''K' ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sigma-Aldrich
Sigma-Aldrich (formally MilliporeSigma) is an American chemical, life science, and biotechnology company that is owned by the German chemical conglomerate Merck Group. Sigma-Aldrich was created in 1975 by the merger of Sigma Chemical Company and Aldrich Chemical Company. It grew through various acquisitions until it had over 9,600 employees and was listed on the Fortune 1000. The company is headquartered in St. Louis and has operations in approximately 40 countries. In 2015, the German chemical conglomerate Merck Group acquired Sigma-Aldrich for $17 billion. The company is currently a part of Merck's life science business and in combination with Merck's earlier acquired Millipore Corporation, Millipore, operates as MilliporeSigma. History Sigma Chemical Company of St. Louis and Aldrich Chemical Company of Milwaukee were both American specialty chemical companies when they merged in August 1975. The company grew throughout the 1980s and 1990s, with significant expansion in fac ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Molecular Dipole Moment
In physics, a dipole () is an electromagnetic phenomenon which occurs in two ways: *An electric dipole deals with the separation of the positive and negative electric charges found in any electromagnetic system. A simple example of this system is a pair of charges of equal magnitude but opposite sign separated by some typically small distance. (A permanent electric dipole is called an electret.) *A magnetic dipole is the closed circulation of an electric current system. A simple example is a single loop of wire with constant current through it. A bar magnet is an example of a magnet with a permanent magnetic dipole moment. Dipoles, whether electric or magnetic, can be characterized by their dipole moment, a vector quantity. For the simple electric dipole, the electric dipole moment points from the negative charge towards the positive charge, and has a magnitude equal to the strength of each charge times the separation between the charges. (To be precise: for the definition of ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Potassium Hydroxide
Potassium hydroxide is an inorganic compound with the formula K OH, and is commonly called caustic potash. Along with sodium hydroxide (NaOH), KOH is a prototypical strong base. It has many industrial and niche applications, most of which exploit its caustic nature and its reactivity toward acids. An estimated 700,000 to 800,000 tonnes were produced in 2005. KOH is noteworthy as the precursor to most soft and liquid soaps, as well as numerous potassium-containing chemicals. It is a white solid that is dangerously corrosive. Properties and structure KOH exhibits high thermal stability. Because of this high stability and relatively low melting point, it is often melt-cast as pellets or rods, forms that have low surface area and convenient handling properties. These pellets become tacky in air because KOH is hygroscopic. Most commercial samples are ca. 90% pure, the remainder being water and carbonates. Its dissolution in water is strongly exothermic. Concentrated aqueous so ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Benzoic Acid
Benzoic acid is a white (or colorless) solid organic compound with the formula , whose structure consists of a benzene ring () with a carboxyl () substituent. It is the simplest aromatic carboxylic acid. The name is derived from gum benzoin, which was for a long time its only source. Benzoic acid occurs naturally in many plants and serves as an intermediate in the biosynthesis of many secondary metabolites. Salts of benzoic acid are used as food preservatives. Benzoic acid is an important precursor for the industrial synthesis of many other organic substances. The salts and esters of benzoic acid are known as benzoates . History Benzoic acid was discovered in the sixteenth century. The dry distillation of gum benzoin was first described by Nostradamus (1556), and then by Alexius Pedemontanus (1560) and Blaise de Vigenère (1596). Justus von Liebig and Friedrich Wöhler determined the composition of benzoic acid. These latter also investigated how hippuric acid is r ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
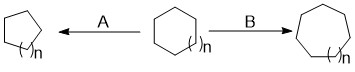
Ring Contraction
Ring expansion and ring contraction reactions in the course of organic synthesis refer to a set of reactions which can lead to the expansion or contraction of an existing ring. This often makes it possible to access structures that would be difficult if not impossible to synthesise with single cyclization reactions. Ring expansions are valuable because they allow access to larger systems that are difficult to synthesize through a single cyclization due to the slow rate of formation. Ring contractions are useful for making smaller, more strained rings from larger rings. Expansions are classified by the mechanism of expansion and the atom(s) added; contractions are characterized simply by the reactive intermediate which performs the contraction. Description In the course of an organic synthesis, a chemist often needs to form a new or alter an existing ring. Ring expansion and ring contraction reactions are used to expand or contract an existing ring, often making it possible to ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Bromination
In chemistry, halogenation is a chemical reaction that entails the introduction of one or more halogens into a compound. Halide-containing compounds are pervasive, making this type of transformation important, e.g. in the production of polymers, drugs. This kind of conversion is in fact so common that a comprehensive overview is challenging. This article mainly deals with halogenation using elemental halogens (F2, Cl2, Br2, I2). Halides are also commonly introduced using salts of the halides and halogen acids. Many specialized reagents exist for and introducing halogens into diverse substrates, e.g. thionyl chloride. Organic chemistry Several pathways exist for the halogenation of organic compounds, including free radical halogenation, ketone halogenation, electrophilic halogenation, and halogen addition reaction. The nature of the substrate determines the pathway. The facility of halogenation is influenced by the halogen. Fluorine and chlorine are more electrophilic an ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hofmann Elimination
Hofmann elimination is an elimination reaction of an amine to form alkenes. The least stable alkene (the one with the least number of substituents on the carbons of the double bond), called the Hofmann product, is formed. This tendency, known as the Hofmann alkene synthesis rule, is in contrast to usual elimination reactions, where Zaitsev's rule predicts the formation of the most stable alkene. It is named after its discoverer, August Wilhelm von Hofmann. The reaction starts with the formation of a quaternary ammonium iodide salt by treatment of the amine with excess methyl iodide (exhaustive methylation), followed by treatment with silver oxide and water to form a quaternary ammonium hydroxide. When this salt is decomposed by heat, the Hofmann product is preferentially formed due to the steric bulk of the leaving group causing the hydroxide to abstract the more easily accessible hydrogen. : In the Hofmann elimination, the least substituted alkene is typically favored due ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tropinone
Tropinone is an alkaloid, famously synthesised in 1917 by Robert Robinson as a synthetic precursor to atropine, a scarce commodity during World War I. Tropinone and the alkaloids cocaine and atropine all share the same tropane core structure. Its corresponding conjugate acid at pH 7.3 major species is known as tropiniumone. Synthesis The first synthesis of tropinone was by Richard Willstätter in 1901. It started from the seemingly related cycloheptanone, but required many steps to introduce the nitrogen bridge; the overall yield for the synthesis path is only 0.75%. Willstätter had previously synthesized cocaine from tropinone, in what was the first synthesis and elucidation of the structure of cocaine. Robinson's "double Mannich" reaction The 1917 synthesis by Robinson is considered a classic in total synthesis due to its simplicity and biomimetic approach. Tropinone is a bicyclic molecule, but the reactants used in its preparation are fairly simple: succinaldehyde, m ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Oxidation
Redox (reduction–oxidation, , ) is a type of chemical reaction in which the oxidation states of substrate change. Oxidation is the loss of electrons or an increase in the oxidation state, while reduction is the gain of electrons or a decrease in the oxidation state. There are two classes of redox reactions: * ''Electron-transfer'' – Only one (usually) electron flows from the reducing agent to the oxidant. This type of redox reaction is often discussed in terms of redox couples and electrode potentials. * ''Atom transfer'' – An atom transfers from one substrate to another. For example, in the rusting of iron, the oxidation state of iron atoms increases as the iron converts to an oxide, and simultaneously the oxidation state of oxygen decreases as it accepts electrons released by the iron. Although oxidation reactions are commonly associated with the formation of oxides, other chemical species can serve the same function. In hydrogenation, C=C (and other) bond ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |