|
Triangulane
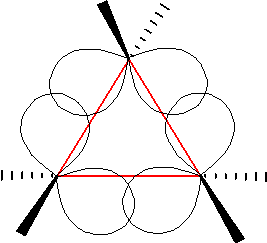
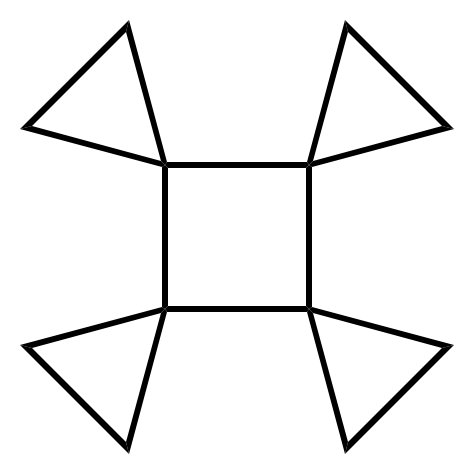
A triangulane is a hydrocarbon consisting exclusively of a series of spiro-linked cyclopropane rings. Triangulanes are named according to the rules of systematic nomenclature for spiro compounds. The pattern of their common names is " 'n''riangulane", where ''n'' is the number of cyclopropane units. The simplest such chemical, riangulane, is named spiro .2entane by systematic nomenclature. Chains consisting of four or more cyclopropane units— riangulane and higher—can form chiral helices. This property is unusual for a molecule that contains no stereogenic atoms; the chiral nature is due to restricted mobility of the chain ends analogous to helicene In organic chemistry, helicenes are ortho-condensed polycyclic aromatic compounds in which benzene rings or other aromatics are angularly annulated to give helically-shaped chiral molecules. The chemistry of helicenes has attracted continuing ... molecules. The rings can form a branched or cyclic patterns. For example, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Spiro(2
{{Disambiguation ...
Spiro(s) may refer to: * Spiro, Oklahoma, a town in the U.S. ** Spiro Mounds, an archaeological site * Spiro (band), a British music group * Spiro (name), including a list of people with the name * Špiro, South Slavic masculine given name * ARA ''Spiro'', two ships of the Argentine Navy * , an oil tanker * Euler spiral, or spiro, a curve * Spiro compound, a type of chemical structure * Spironolactone, a medicine, often used in feminizing hormone therapy See also * * * Spiro compound, a class of organic compound featuring two rings joined at one atom * Spirou (comics), a Belgian comic strip character * Spyro * Spira (other) Spira may refer to: * Spira (car), a three-wheeled motor vehicle * Spira (confectionery), a Cadbury chocolate bar in a helix shape * Spira (name), including a list of people with the name * Spira (''Final Fantasy''), the fictional world of the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrocarbon
In organic chemistry, a hydrocarbon is an organic compound consisting entirely of hydrogen and carbon. Hydrocarbons are examples of group 14 hydrides. Hydrocarbons are generally colourless and hydrophobic, and their odors are usually weak or exemplified by the odors of gasoline and lighter fluid. They occur in a diverse range of molecular structures and phases: they can be gases (such as methane and propane), liquids (such as hexane and benzene), low melting solids (such as paraffin wax and naphthalene) or polymers (such as polyethylene and polystyrene). In the fossil fuel industries, ''hydrocarbon'' refers to the naturally occurring petroleum, natural gas and coal, and to their hydrocarbon derivatives and purified forms. Combustion of hydrocarbons is the main source of the world's energy. Petroleum is the dominant raw-material source for organic commodity chemicals such as solvents and polymers. Most anthropogenic (human-generated) emissions of greenhouse gases are carbon di ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Spiro Compound
In organic chemistry, spiro compounds are compounds that have at least two molecular rings with only one common atom. The simplest spiro compounds are bicyclic (having just two rings), or have a bicyclic portion as part of the larger ring system, in either case with the two rings connected through the defining single common atom. The one common atom connecting the participating rings distinguishes spiro compounds from other bicyclics: from ''isolated ring compounds'' like biphenyl that have no connecting atoms, from ''fused ring compounds'' like decalin having two rings linked by two adjacent atoms, and from ''bridged ring compounds'' like norbornane with two rings linked by two non-adjacent atoms.For all four categories, see The specific chapters can be found aan respectively, same access date. For the description featuring adjacent atoms for all but the isolated category, see Clayden, op. cit. Spiro compounds may be fully carbocyclic (all carbon) or heterocyclic (havi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cyclopropane
Cyclopropane is the cycloalkane with the molecular formula (CH2)3, consisting of three methylene groups (CH2) linked to each other to form a ring. The small size of the ring creates substantial ring strain in the structure. Cyclopropane itself is mainly of theoretical interest but many of its derivatives are of commercial or biological significance. History Cyclopropane was discovered in 1881 by August Freund, who also proposed the correct structure for the substance in his first paper. Freund treated 1,3-dibromopropane with sodium, causing an intramolecular Wurtz reaction leading directly to cyclopropane. The yield of the reaction was improved by Gustavson in 1887 with the use of zinc instead of sodium. Cyclopropane had no commercial application until Henderson and Lucas discovered its anaesthetic properties in 1929; industrial production had begun by 1936. In modern anaesthetic practice, it has been superseded by other agents. Anaesthesia Cyclopropane was introduced into cli ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chemical Nomenclature
A chemical nomenclature is a set of rules to generate systematic names for chemical compounds. The nomenclature used most frequently worldwide is the one created and developed by the International Union of Pure and Applied Chemistry (IUPAC). The IUPAC's rules for naming organic and inorganic compounds are contained in two publications, known as the ''Blue Book''. . and the '' Red Book'',. respectively. A third publication, known as the '' Green Book'',. recommends the use of symbols for physical quantities (in association with the IUPAP), while a fourth, the ''Gold Book'',''Compendium of Chemical Terminology, IMPACT Recommendations (2nd Ed.)'', Oxford:Blackwell Scientific Publications. (1997) defines many technical terms used in chemistry. Similar compendia exist for biochemistry''Biochemical Nomenclature and Related Documents'', London: Portland Press, 1992. (the ''White Book'', in association with the IUBMB), analytical chemistry (the '' Orange Book''), macromolecular chemistr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Spiro Compound
In organic chemistry, spiro compounds are compounds that have at least two molecular rings with only one common atom. The simplest spiro compounds are bicyclic (having just two rings), or have a bicyclic portion as part of the larger ring system, in either case with the two rings connected through the defining single common atom. The one common atom connecting the participating rings distinguishes spiro compounds from other bicyclics: from ''isolated ring compounds'' like biphenyl that have no connecting atoms, from ''fused ring compounds'' like decalin having two rings linked by two adjacent atoms, and from ''bridged ring compounds'' like norbornane with two rings linked by two non-adjacent atoms.For all four categories, see The specific chapters can be found aan respectively, same access date. For the description featuring adjacent atoms for all but the isolated category, see Clayden, op. cit. Spiro compounds may be fully carbocyclic (all carbon) or heterocyclic (havi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Common Name (chemistry)
In chemistry, a trivial name is a nonsystematic name for a chemical substance. That is, the name is not recognized according to the rules of any formal system of chemical nomenclature such as IUPAC inorganic or IUPAC organic nomenclature. A trivial name is not a formal name and is usually a common name. Generally, trivial names are not useful in describing the essential properties of the thing being named. Properties such as the molecular structure of a chemical compound are not indicated. And, in some cases, trivial names can be ambiguous or will carry different meanings in different industries or in different geographic regions (for example, a trivial name such as ''white metal'' can mean various things). Trivial names are simpler. As a result, a limited number of trivial chemical names are retained names, an accepted part of the nomenclature. Trivial names often arise in the common language; they may come from historic usages in, for example, alchemy. Many trivial names p ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Spiropentane
Spiropentane is a hydrocarbon with formula . It is the simplest Spiro compound, spiro-connected cycloalkane, a triangulane. It took several years after the discovery in 1887 until the structure of the molecule was determined. According to the nomenclature rules for spiro compounds, the systematic name is spiro[2.2]pentane. However, there can be no isomer, constitutive isomeric spiropentanes, hence the name is unique without brackets and numbers. Synthesis After Gustavson produced cyclopropane by reacting with ground-up zinc metal, he tried the same reaction with (see formula scheme). The starting material is easily obtained by reacting pentaerythritol with hydrobromic acid. A molecule with the formula was obtained. It was called in the initial publication. In 1907, Fecht expressed the assumption that it must be spiropentane, a constitutional isomer of vinylcyclopropane. Further evidence for the structure of the hydrocarbon comes from the fact that it could also be obtained f ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chiral
Chirality is a property of asymmetry important in several branches of science. The word ''chirality'' is derived from the Greek (''kheir''), "hand", a familiar chiral object. An object or a system is ''chiral'' if it is distinguishable from its mirror image; that is, it cannot be superimposed onto it. Conversely, a mirror image of an ''achiral'' object, such as a sphere, cannot be distinguished from the object. A chiral object and its mirror image are called ''enantiomorphs'' (Greek, "opposite forms") or, when referring to molecules, '' enantiomers''. A non-chiral object is called ''achiral'' (sometimes also ''amphichiral'') and can be superposed on its mirror image. The term was first used by Lord Kelvin in 1893 in the second Robert Boyle Lecture at the Oxford University Junior Scientific Club which was published in 1894: Human hands are perhaps the most recognized example of chirality. The left hand is a non-superimposable mirror image of the right hand; no matter ho ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Stereogenic
In stereochemistry, a stereocenter of a molecule is an atom (center), axis or plane that is the focus of stereoisomerism; that is, when having at least three different groups bound to the stereocenter, interchanging any two different groups creates a new stereoisomer. Stereocenters are also referred to as stereogenic centers. A stereocenter is geometrically defined as a point (location) in a molecule; a stereocenter is usually but not always a specific atom, often carbon. Stereocenters can exist on chiral or achiral molecules; stereocenters can contain single bonds or double bonds. The number of hypothetical stereoisomers can be predicted by using 2''n'', with ''n'' being the number of tetrahedral stereocenters; however, exceptions such as meso compounds can reduce the prediction to below the expected 2''n''. Chirality centers are a type of stereocenter with four different substituent groups; chirality centers are a specific subset of stereocenters because they can only ha ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Helicene
In organic chemistry, helicenes are ortho-condensed polycyclic aromatic compounds in which benzene rings or other aromatics are angularly annulated to give helically-shaped chiral molecules. The chemistry of helicenes has attracted continuing attention because of their unique structural, spectral, and optical features. Structure and properties The systematic naming for this class of compounds is based on the number of rings: 'n''elicene is the structure consisting of ''n'' rings. According to IUPAC, only structures where ''n'' is at least 5 are considered helicenes. Some specific compounds also have alternate or trivial names. As the number of rings increases, starting at four, the structure becomes non-planar, but instead the planes of consecutive rings tilt to prevent steric collisions. The resulting helix is chiral. For helicenes with six benzene units, a 360° turn is completed. In the helicene series the dihedral angles between the extremities increases going from elic ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Rotane
A rotane is a hydrocarbon consisting of a central cycloalkane ring with cyclopropane units spiro-linked to each corner. The systematic naming pattern for these molecules is " 'n''otane", where ''n'' is the number of atoms in the central ring. The simplest such chemical, otane, consists solely of a branched array of spiro-cyclopropane units, and is thus a branched triangulane A triangulane is a hydrocarbon consisting exclusively of a series of spiro-linked cyclopropane rings. Triangulanes are named according to the rules of systematic nomenclature for spiro compounds. The pattern of their common names is " 'n''riangu .... References * Hydrocarbons Cyclopropanes Spiro compounds {{Hydrocarbon-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |