|
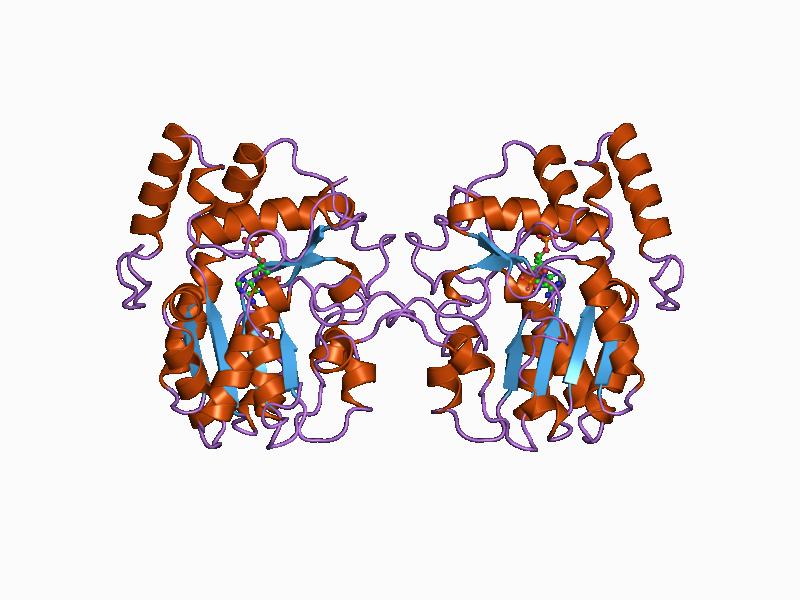
Thymidylate Synthase (FAD)
In enzymology, a thymidylate synthase (FAD) () is an enzyme that catalyzes the chemical reaction :5,10-methylenetetrahydrofolate + dUMP + FADH2 \rightleftharpoons dTMP + tetrahydrofolate + FAD The 3 substrates of this enzyme are 5,10-methylenetetrahydrofolate, dUMP, and FADH2, whereas its 3 products are dTMP, tetrahydrofolate, and FAD. This enzyme belongs to the family of transferases, to be specific those transferring one-carbon group methyltransferases. The systematic name of this enzyme class is 5,10-methylenetetrahydrofolate,FADH2:dUMP C-methyltransferase. Other names in common use include Thy1, and ThyX. This enzyme participates in pyrimidine metabolism and one carbon pool by folate. Most organisms, including humans, use the thyA- or TYMS-encoded classic thymidylate synthase whereas some bacteria use the similar flavin-dependent thymidylate synthase (FDTS) instead. Structural studies As of late 2007, 3 structures have been solved for this class of enzymes, with ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Enzymology
Enzymes () are proteins that act as biological catalysts by accelerating chemical reactions. The molecules upon which enzymes may act are called substrates, and the enzyme converts the substrates into different molecules known as products. Almost all metabolic processes in the cell need enzyme catalysis in order to occur at rates fast enough to sustain life. Metabolic pathways depend upon enzymes to catalyze individual steps. The study of enzymes is called ''enzymology'' and the field of pseudoenzyme analysis recognizes that during evolution, some enzymes have lost the ability to carry out biological catalysis, which is often reflected in their amino acid sequences and unusual 'pseudocatalytic' properties. Enzymes are known to catalyze more than 5,000 biochemical reaction types. Other biocatalysts are catalytic RNA molecules, called ribozymes. Enzymes' specificity comes from their unique three-dimensional structures. Like all catalysts, enzymes increase the reacti ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Transferase
A transferase is any one of a class of enzymes that catalyse the transfer of specific functional groups (e.g. a methyl or glycosyl group) from one molecule (called the donor) to another (called the acceptor). They are involved in hundreds of different biochemical pathways throughout biology, and are integral to some of life's most important processes. Transferases are involved in myriad reactions in the cell. Three examples of these reactions are the activity of coenzyme A (CoA) transferase, which transfers thiol esters, the action of N-acetyltransferase, which is part of the pathway that metabolizes tryptophan, and the regulation of pyruvate dehydrogenase (PDH), which converts pyruvate to acetyl CoA. Transferases are also utilized during translation. In this case, an amino acid chain is the functional group transferred by a peptidyl transferase. The transfer involves the removal of the growing amino acid chain from the tRNA molecule in the A-site of the ribosome a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Thymidylate Synthetase
Thymidylate synthase (TS) () is an enzyme that catalyzes the conversion of deoxyuridine monophosphate (dUMP) to deoxythymidine monophosphate (dTMP). Thymidine is one of the nucleotides in DNA. With inhibition of TS, an imbalance of deoxynucleotides and increased levels of dUMP arise. Both cause DNA damage. Function The following reaction is catalyzed by thymidylate synthase: : 5,10-methylenetetrahydrofolate + dUMP \rightleftharpoons dihydrofolate + dTMP By means of reductive methylation, deoxyuridine monophosphate (dUMP) and N5,N10-methylene tetrahydrofolate are together used to form dTMP, yielding dihydrofolate as a secondary product. This provides the sole de novo pathway for production of dTMP and is the only enzyme in folate metabolism in which the 5,10-methylenetetrahydrofolate is oxidised during one-carbon transfer. The enzyme is essential for regulating the balanced supply of the 4 DNA precursors in normal DNA replication: defects in the enzyme activity affectin ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Protein Data Bank
The Protein Data Bank (PDB) is a database for the three-dimensional structural data of large biological molecules, such as proteins and nucleic acids. The data, typically obtained by X-ray crystallography, NMR spectroscopy, or, increasingly, cryo-electron microscopy, and submitted by biologists and biochemists from around the world, are freely accessible on the Internet via the websites of its member organisations (PDBe, PDBj, RCSB, and BMRB). The PDB is overseen by an organization called the Worldwide Protein Data Bank, wwPDB. The PDB is a key in areas of structural biology, such as structural genomics. Most major scientific journals and some funding agencies now require scientists to submit their structure data to the PDB. Many other databases use protein structures deposited in the PDB. For example, SCOP and CATH classify protein structures, while PDBsum provides a graphic overview of PDB entries using information from other sources, such as Gene ontology. History Two ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tertiary Structure
Protein tertiary structure is the three dimensional shape of a protein. The tertiary structure will have a single polypeptide chain "backbone" with one or more protein secondary structures, the protein domains. Amino acid side chains may interact and bond in a number of ways. The interactions and bonds of side chains within a particular protein determine its tertiary structure. The protein tertiary structure is defined by its atomic coordinates. These coordinates may refer either to a protein domain or to the entire tertiary structure.Branden C. and Tooze J. "Introduction to Protein Structure" Garland Publishing, New York. 1990 and 1991. A number of tertiary structures may fold into a quaternary structure.Kyte, J. "Structure in Protein Chemistry." Garland Publishing, New York. 1995. History The science of the tertiary structure of proteins has progressed from one of hypothesis to one of detailed definition. Although Emil Fischer had suggested proteins were made of poly ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
TYMS
Thymidylate synthase (TS) () is an enzyme that catalyzes the conversion of deoxyuridine monophosphate (dUMP) to deoxythymidine monophosphate (dTMP). Thymidine is one of the nucleotides in DNA. With inhibition of TS, an imbalance of deoxynucleotides and increased levels of dUMP arise. Both cause DNA damage. Function The following reaction is catalyzed by thymidylate synthase: : 5,10-methylenetetrahydrofolate + dUMP \rightleftharpoons dihydrofolate + dTMP By means of reductive methylation, deoxyuridine monophosphate (dUMP) and N5,N10-methylene tetrahydrofolate are together used to form dTMP, yielding dihydrofolate as a secondary product. This provides the sole de novo pathway for production of dTMP and is the only enzyme in folate metabolism in which the 5,10-methylenetetrahydrofolate is oxidised during one-carbon transfer. The enzyme is essential for regulating the balanced supply of the 4 DNA precursors in normal DNA replication: defects in the enzyme activity affectin ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
One Carbon Pool By Folate
Folate, also known as vitamin B9 and folacin, is one of the B vitamins. Manufactured folic acid, which is converted into folate by the body, is used as a dietary supplement and in food fortification as it is more stable during processing and storage. Folate is required for the body to make DNA and RNA and metabolise amino acids necessary for cell division. As humans cannot make folate, it is required in the diet, making it an essential nutrient. It occurs naturally in many foods. The recommended adult daily intake of folate in the U.S. is 400 micrograms from foods or dietary supplements. Folate in the form of folic acid is used to treat anemia caused by folate deficiency. Folic acid is also used as a supplement by women during pregnancy to reduce the risk of neural tube defects (NTDs) in the baby. Low levels in early pregnancy are believed to be the cause of more than half of babies born with NTDs. More than 80 countries use either mandatory or voluntary fortification of ce ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pyrimidine Metabolism
Pyrimidine biosynthesis occurs both in the body and through organic synthesis. ''De novo'' biosynthesis of pyrimidine ''De Novo'' biosynthesis of a pyrimidine is catalyzed by three gene products CAD, DHODH and UMPS. The first three enzymes of the process are all coded by the same gene in CAD which consists of carbamoyl phosphate synthetase II, aspartate carbamoyltransferase and dihydroorotase. Dihydroorotate dehydrogenase (DHODH) unlike CAD and UMPS is a mono-functional enzyme and is localized in the mitochondria. UMPS is a bifunctional enzyme consisting of orotate phosphoribosyltransferase (OPRT) and orotidine monophosphate decarboxylase (OMPDC). Both, CAD and UMPS are localized around the mitochondria, in the cytosol. In Fungi, a similar protein exists but lacks the dihydroorotase function: another protein catalyzes the second step. In other organisms (Bacteria, Archaea and the other Eukaryota), the first three steps are done by three different enzymes. Pyrimidine ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
List Of Enzymes
This article lists enzymes by their classification in the International Union of Biochemistry and Molecular Biology's Enzyme Commission (EC) numbering system. * List of EC numbers (EC 5) * List of EC numbers (EC 6) :Oxidoreductases (EC 1) ( Oxidoreductase) * Dehydrogenase *Luciferase * DMSO reductase :EC 1.1 (act on the CH-OH group of donors) * :EC 1.1.1 (with NAD+ or NADP+ as acceptor) ** Alcohol dehydrogenase (NAD) ** Alcohol dehydrogenase (NADP) ** Homoserine dehydrogenase ** Aminopropanol oxidoreductase **Diacetyl reductase ** Glycerol dehydrogenase ** Propanediol-phosphate dehydrogenase **glycerol-3-phosphate dehydrogenase (NAD+) **D-xylulose reductase ** L-xylulose reductase ** Lactate dehydrogenase ** Malate dehydrogenase ** Isocitrate dehydrogenase **HMG-CoA reductase * :EC 1.1.2 (with a cytochrome as acceptor) * :EC 1.1.3 (with oxygen as acceptor) ** Glucose oxidase ** L-gulonolactone oxidase ** Thiamine oxidase ** Xanthine oxidase * :EC 1.1. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Flavin Adenine Dinucleotide
Flavin may refer to: Placename * Flavin, Aveyron, a commune in southern France Surname * Adrian Flavin (born 1979), a professional rugby player * Christopher Flavin, president of the Worldwatch Institute * Dan Flavin (1933–1996), a minimalist artist famous for using fluorescent light fixtures * Dan Flavin (politician), Louisiana politician * James Flavin (1906–1976), an American character actor * Jennifer Flavin (born 1968), a former model and wife of actor Sylvester Stallone * Martin Flavin (1883–1967), an American playwright and novelist * Martin Flavin (politician) (1841–1917), Irish Nationalist politician, Member of Parliament (MP) for Cork, 1891–1892 * Michael Joseph Flavin (1866-1944), Irish Nationalist politician, Member of Parliament (MP) for North Kerry, 1896-1918 * Mick Flavin Mick Flavin (born 3 August 1950) is an Irish country singer from Ballinamuck in County Longford. Flavin recorded his first album in Athlone in June 1986. His first big hit bei ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |