|
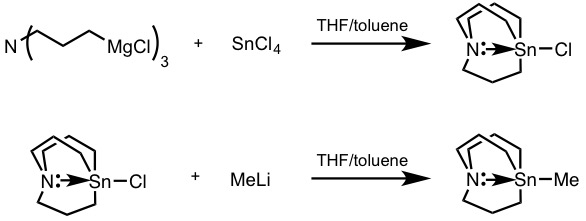
Stannatrane Lithiation
A stannatrane (IUPAC: 1-aza-5-stannabicyclo[3.3.3]undecane) is a tin-based atrane belonging to the larger class of Organotin chemistry, organostannanes. Though the term stannatrane is often used to refer to the more commonly employed carbastannatrane, azastannatranes have also been synthesized (prefix refers to the identity of the atom bound directly to tin center). Stannatrane reagents offer highly selective methods for the incorporation of "R" substituents in complex molecules for late-stage diversification. These reagents differ from their tetraalkyl organostannane analogues in that there is no participation of dummy ligands in the transmetalation step, offering selective alkyl transfer in Stille coupling, Stille Coupling reactions. These transmetalating agents are known to be air- and moisture-stable, as well as generally less toxic than their tetraalkyl counterparts. History and structural properties The first carbastannatrane was reported in 1984 by Jurkschat and Tzschach. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
General Stannatrane
A general officer is an officer of high rank in the armies, and in some nations' air forces, space forces, and marines or naval infantry. In some usages the term "general officer" refers to a rank above colonel."general, adj. and n.". OED Online. March 2021. Oxford University Press. https://www.oed.com/view/Entry/77489?rskey=dCKrg4&result=1 (accessed May 11, 2021) The term ''general'' is used in two ways: as the generic title for all grades of general officer and as a specific rank. It originates in the 16th century, as a shortening of '' captain general'', which rank was taken from Middle French ''capitaine général''. The adjective ''general'' had been affixed to officer designations since the late medieval period to indicate relative superiority or an extended jurisdiction. Today, the title of ''general'' is known in some countries as a four-star rank. However, different countries use different systems of stars or other insignia for senior ranks. It has a NATO r ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Bond Order
In chemistry, bond order, as introduced by Linus Pauling, is defined as the difference between the number of bonds and anti-bonds. The bond order itself is the number of electron pairs (covalent bonds) between two atoms. For example, in diatomic nitrogen N≡N, the bond order between the two nitrogen atoms is 3 (triple bond). In acetylene H–C≡C–H, the bond order between the two carbon atoms is also 3, and the C–H bond order is 1 ( single bond). In carbon monoxide , the bond order between carbon and oxygen is 3. In thiazyl trifluoride , the bond order between sulfur and nitrogen is 3, and between sulfur and fluorine is 1. In diatomic oxygen O=O the bond order is 2 (double bond). In ethylene the bond order between the two carbon atoms is also 2. The bond order between carbon and oxygen in carbon dioxide O=C=O is also 2. In phosgene , the bond order between carbon and oxygen is 2, and between carbon and chlorine is 1. In some molecules, bond orders can be 4 (quadr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Palladium
Palladium is a chemical element with the symbol Pd and atomic number 46. It is a rare and lustrous silvery-white metal discovered in 1803 by the English chemist William Hyde Wollaston. He named it after the asteroid Pallas, which was itself named after the epithet of the Greek goddess Athena, acquired by her when she slew Pallas. Palladium, platinum, rhodium, ruthenium, iridium and osmium form a group of elements referred to as the platinum group metals (PGMs). They have similar chemical properties, but palladium has the lowest melting point and is the least dense of them. More than half the supply of palladium and its congener platinum is used in catalytic converters, which convert as much as 90% of the harmful gases in automobile exhaust (hydrocarbons, carbon monoxide, and nitrogen dioxide) into nontoxic substances (nitrogen, carbon dioxide and water vapor). Palladium is also used in electronics, dentistry, medicine, hydrogen purification, chemical applications, groundwate ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electrophile
In chemistry, an electrophile is a chemical species that forms bonds with nucleophiles by accepting an electron pair. Because electrophiles accept electrons, they are Lewis acids. Most electrophiles are positively charged, have an atom that carries a partial positive charge, or have an atom that does not have an octet of electrons. Electrophiles mainly interact with nucleophiles through addition and substitution reactions. Frequently seen electrophiles in organic syntheses include cations such as H+ and NO+, polarized neutral molecules such as HCl, alkyl halides, acyl halides, and carbonyl compounds, polarizable neutral molecules such as Cl2 and Br2, oxidizing agents such as organic peracids, chemical species that do not satisfy the octet rule such as carbenes and radicals, and some Lewis acids such as BH3 and DIBAL. Organic chemistry Addition of halogens These occur between alkenes and electrophiles, often halogens as in halogen addition reactions. Common reactions i ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Lone Pair
In chemistry, a lone pair refers to a pair of valence electrons that are not shared with another atom in a covalent bondIUPAC ''Gold Book'' definition''lone (electron) pair''/ref> and is sometimes called an unshared pair or non-bonding pair. Lone pairs are found in the outermost electron shell of atoms. They can be identified by using a Lewis structure. Electron pairs are therefore considered lone pairs if two electrons are paired but are not used in chemical bonding. Thus, the number of electrons in lone pairs plus the number of electrons in bonds equals the number of valence electrons around an atom. Lone pair is a concept used in valence shell electron pair repulsion theory (VSEPR theory) which explains the shapes of molecules. They are also referred to in the chemistry of Lewis acids and bases. However, not all non-bonding pairs of electrons are considered by chemists to be lone pairs. Examples are the transition metals where the non-bonding pairs do not influence molecular ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tetramethyltin
Tetramethyltin is an organometallic compound with the formula (CH3)4Sn. This liquid, one of the simplest organotin compounds, is useful for transition-metal mediated conversion of acid chlorides to methyl ketones and aryl halides to aryl methyl ketones. It is volatile and toxic, so care should be taken when using it in the laboratory. Synthesis and structure Tetramethyltin is synthesized by reaction of the Grignard reagent methylmagnesium iodide, with tin tetrachloride, which is synthesized by reacting tin metal with chlorine gas. :4 CH3MgI + SnCl4 → (CH3)4Sn + 4 MgICl In tetramethyltin, the metal surrounded by four methyl groups in a tetrahedral structure is a heavy analogue of neopentane. Applications Precursor to methyltin compounds Tetramethyltin is a precursor to trimethyltin chloride (and related methyltin halides), which are precursors to other organotin compounds. These methyltin chlorides are prepared via the so-called Kocheshkov redistribution reaction. Thus ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Enantiomeric Excess
In stereochemistry, enantiomeric excess (ee) is a measurement of purity used for chiral substances. It reflects the degree to which a sample contains one enantiomer in greater amounts than the other. A racemic mixture has an ee of 0%, while a single completely pure enantiomer has an ee of 100%. A sample with 70% of one enantiomer and 30% of the other has an ee of 40% (70% − 30%). Definition Enantiomeric excess is defined as the absolute difference between the mole fraction of each enantiomer: :\ ee = , F_R - F_S, where :\ F_R + F_S = 1 In practice, it is most often expressed as a percent enantiomeric excess. The enantiomeric excess can be determined in another way if we know the amount of each enantiomer produced. If one knows the moles of each enantiomer produced then: Enantiomeric excess is used as one of the indicators of the success of an asymmetric synthesis. For mixtures of diastereomers, there are analogous definitions and uses for diastereomeric excess an ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Stannatrane Lithiation
A stannatrane (IUPAC: 1-aza-5-stannabicyclo[3.3.3]undecane) is a tin-based atrane belonging to the larger class of Organotin chemistry, organostannanes. Though the term stannatrane is often used to refer to the more commonly employed carbastannatrane, azastannatranes have also been synthesized (prefix refers to the identity of the atom bound directly to tin center). Stannatrane reagents offer highly selective methods for the incorporation of "R" substituents in complex molecules for late-stage diversification. These reagents differ from their tetraalkyl organostannane analogues in that there is no participation of dummy ligands in the transmetalation step, offering selective alkyl transfer in Stille coupling, Stille Coupling reactions. These transmetalating agents are known to be air- and moisture-stable, as well as generally less toxic than their tetraalkyl counterparts. History and structural properties The first carbastannatrane was reported in 1984 by Jurkschat and Tzschach. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
One-pot Method
In chemistry a one-pot synthesis is a strategy to improve the efficiency of a chemical reaction whereby a reactant is subjected to successive chemical reactions in just one reactor. This is much desired by chemists because avoiding a lengthy separation process and purification of the intermediate chemical compounds can save time and resources while increasing chemical yield. An example of a one-pot synthesis is the total synthesis of tropinone or the Gassman indole synthesis. Sequential one-pot syntheses can be used to generate even complex targets with multiple stereocentres, such as oseltamivir, which may significantly shorten the number of steps required overall and have important commercial implications. A sequential one-pot synthesis with reagents added to a reactor one at a time and without work-up is also called a telescoping synthesis. In one such procedure the reaction of 3-N-tosylaminophenol I with acrolein II affords a hydroxyl substituted quinoline III through 4 seq ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Triallylamine
Triallylamine is the organic compound with the formula N(CH2CH=CH2)3. It is a colorless liquid with an ammonia-like odor. It is multifunctional, featuring a tertiary amine and three alkene groups. Triallylamine (and mono- and diallyl amines) is produced by the treating allyl chloride with ammonia. Allylamines have particularly weak α-CH bonds, being near 80 kcal/mol.{{cite journal , doi=10.1021/JO9813843, title=Α-C−H Bond Dissociation Energies of Some Tertiary Amines, year=1999, last1=Dombrowski, first1=G. W., last2=Dinnocenzo, first2=J. P., last3=Farid, first3=S., last4=Goodman, first4=J. L., last5=Gould, first5=I. R., journal=The Journal of Organic Chemistry, volume=64, issue=2, pages=427–431 Related compounds *Allylamine *Diallylamine References [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Schwartz's Reagent
Schwartz's reagent is the common name for the organozirconium compound with the formula (C5H5)2ZrHCl, sometimes called zirconocene hydrochloride or zirconocene chloride hydride, and is named after Jeffrey Schwartz, a chemistry professor at Princeton University.This metallocene is used in organic synthesis for various transformations of alkenes and alkynes. Preparation The complex was first prepared by Wailes and Weigold. It can be purchased or readily prepared by reduction of zirconocene dichloride with lithium aluminium hydride: : (C5H5)2ZrCl2 + LiAlH4 → (C5H5)2ZrHCl + LiAlCl4 This reaction also affords (C5H5)2ZrH2, which is treated with methylene chloride to give Schwartz's reagent LiAl(O-t-Bu)3H can be used in place of LiAlH4. An alternative procedure that generated Schwartz's reagent from dihydride has also been reported. Moreover, it's possible to perform an ''in situ'' preparation of (C5H5)2ZrHCl from zirconocene dichloride by using LiH. This meth ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |

2.jpg)