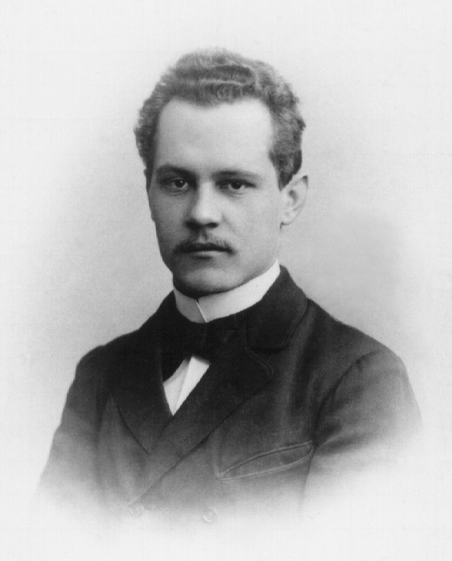|
Slater–Condon Rules
Within computational chemistry, the Slater–Condon rules express integrals of one- and two-body operators over wavefunctions constructed as Slater determinants of orthonormal orbitals in terms of the individual orbitals. In doing so, the original integrals involving ''N''-electron wavefunctions are reduced to sums over integrals involving at most two molecular orbitals, or in other words, the original 3''N'' dimensional integral is expressed in terms of many three- and six-dimensional integrals. The rules are used in deriving the working equations for all methods of approximately solving the Schrödinger equation that employ wavefunctions constructed from Slater determinants. These include Hartree–Fock theory, where the wavefunction is a single determinant, and all those methods which use Hartree–Fock theory as a reference such as Møller–Plesset perturbation theory, and Coupled cluster and Configuration interaction theories. In 1929 John C. Slater derived expressions for ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Computational Chemistry
Computational chemistry is a branch of chemistry that uses computer simulation to assist in solving chemical problems. It uses methods of theoretical chemistry, incorporated into computer programs, to calculate the structures and properties of molecules, groups of molecules, and solids. It is essential because, apart from relatively recent results concerning the hydrogen molecular ion (dihydrogen cation, see references therein for more details), the quantum many-body problem cannot be solved analytically, much less in closed form. While computational results normally complement the information obtained by chemical experiments, it can in some cases predict hitherto unobserved chemical phenomena. It is widely used in the design of new drugs and materials. Examples of such properties are structure (i.e., the expected positions of the constituent atoms), absolute and relative (interaction) energies, electronic charge density distributions, dipoles and higher multipole moments, vi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Edward Condon
Edward Uhler Condon (March 2, 1902 – March 26, 1974) was an American nuclear physicist, a pioneer in quantum mechanics, and a participant during World War II in the development of radar and, very briefly, of nuclear weapons as part of the Manhattan Project. The Franck–Condon principle and the Slater–Condon rules are co-named after him.''Edward Condon'' was elected as a member of the in 1944. He was the director of the |
Magnetic Dipole-dipole Interaction
Magnetism is the class of physical attributes that are mediated by a magnetic field, which refers to the capacity to induce attractive and repulsive phenomena in other entities. Electric currents and the magnetic moments of elementary particles give rise to a magnetic field, which acts on other currents and magnetic moments. Magnetism is one aspect of the combined phenomena of electromagnetism. The most familiar effects occur in ferromagnetic materials, which are strongly attracted by magnetic fields and can be magnetized to become permanent magnets, producing magnetic fields themselves. Demagnetizing a magnet is also possible. Only a few substances are ferromagnetic; the most common ones are iron, cobalt, and nickel and their alloys. The rare-earth metals neodymium and samarium are less common examples. The prefix ' refers to iron because permanent magnetism was first observed in lodestone, a form of natural iron ore called magnetite, Fe3O4. All substances exhibit some type of m ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Angular Momentum Coupling
In quantum mechanics, the procedure of constructing eigenstates of total angular momentum out of eigenstates of separate angular momenta is called angular momentum coupling. For instance, the orbit and spin of a single particle can interact through spin–orbit interaction, in which case the complete physical picture must include spin–orbit coupling. Or two charged particles, each with a well-defined angular momentum, may interact by Coulomb forces, in which case coupling of the two one-particle angular momenta to a total angular momentum is a useful step in the solution of the two-particle Schrödinger equation. In both cases the separate angular momenta are no longer constants of motion, but the sum of the two angular momenta usually still is. Angular momentum coupling in atoms is of importance in atomic spectroscopy. Angular momentum coupling of electron spins is of importance in quantum chemistry. Also in the nuclear shell model angular momentum coupling is ubiquitou ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electric Dipole Moment
The electric dipole moment is a measure of the separation of positive and negative electrical charges within a system, that is, a measure of the system's overall polarity. The SI unit for electric dipole moment is the coulomb-meter (C⋅m). The debye (D) is another unit of measurement used in atomic physics and chemistry. Theoretically, an electric dipole is defined by the first-order term of the multipole expansion; it consists of two equal and opposite charges that are infinitesimally close together, although real dipoles have separated charge.Many theorists predict elementary particles can have very tiny electric dipole moments, possibly without separated charge. Such large dipoles make no difference to everyday physics, and have not yet been observed. (See electron electric dipole moment). However, when making measurements at a distance much larger than the charge separation, the dipole gives a good approximation of the actual electric field. The dipole is represented by a v ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Kinetic Energy
In physics, the kinetic energy of an object is the energy that it possesses due to its motion. It is defined as the work needed to accelerate a body of a given mass from rest to its stated velocity. Having gained this energy during its acceleration, the body maintains this kinetic energy unless its speed changes. The same amount of work is done by the body when decelerating from its current speed to a state of rest. Formally, a kinetic energy is any term in a system's Lagrangian which includes a derivative with respect to time. In classical mechanics, the kinetic energy of a non-rotating object of mass ''m'' traveling at a speed ''v'' is \fracmv^2. In relativistic mechanics, this is a good approximation only when ''v'' is much less than the speed of light. The standard unit of kinetic energy is the joule, while the English unit of kinetic energy is the foot-pound. History and etymology The adjective ''kinetic'' has its roots in the Greek word κίνησις ''kinesis'', m ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Spin-orbital
In atomic theory and quantum mechanics, an atomic orbital is a Function (mathematics), function describing the location and wave-like behavior of an electron in an atom. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the Atomic nucleus, atom's nucleus. The term ''atomic orbital'' may also refer to the physical region or space where the electron can be calculated to be present, as predicted by the particular mathematical form of the orbital. Each orbital in an atom is characterized by a set of values of the three quantum numbers , , and , which respectively correspond to the electron's energy, angular momentum, and an angular momentum vector component (magnetic quantum number). Alternative to the magnetic quantum number, the orbitals are often labeled by the associated Spherical harmonics#Harmonic polynomial representation, harmonic polynomials (e.g., ''xy'', ). Each such orbital can be occupied by a maximum o ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Antisymmetrizer
In quantum mechanics, an antisymmetrizer \mathcal (also known as antisymmetrizing operatorP.A.M. Dirac, ''The Principles of Quantum Mechanics'', 4th edition, Clarendon, Oxford UK, (1958) p. 248) is a linear operator that makes a wave function of ''N'' identical fermions antisymmetric under the exchange of the coordinates of any pair of fermions. After application of \mathcal the wave function satisfies the Pauli exclusion principle. Since \mathcal is a projection operator, application of the antisymmetrizer to a wave function that is already totally antisymmetric has no effect, acting as the identity operator. Mathematical definition Consider a wave function depending on the space and spin coordinates of ''N'' fermions: : \Psi(1,2, \ldots, N)\quad\text \quad i \leftrightarrow (\mathbf_i, \sigma_i), where the position vector r''i'' of particle ''i'' is a vector in \mathbb^3 and σi takes on 2''s''+1 values, where ''s'' is the half-integral intrinsic spin of the fermion. For ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Per-Olov Löwdin
Per-Olov Löwdin (October 28, 1916 – October 6, 2000) was a Swedish physicist, professor at the University of Uppsala from 1960 to 1983, and in parallel at the University of Florida until 1993. A former graduate student under Ivar Waller, Löwdin formulated in 1950 the symmetric orthogonalization scheme for atomic and molecular orbital calculations, greatly simplifying the tight-binding method. This scheme is the basis of the zero-differential overlap (ZDO) approximation used in semiempirical theories. In 1956 he introduced the canonical orthogonalization scheme, which is optimal for eliminating approximate linear dependencies of a basis set. These orthogonalization procedures are widely used today in all modern quantum chemistry calculations. He is also credited with the use of fat symbols for matrices, making easy the derivation of several theorems of quantum mechanics. The famous 'Löwdin's pairing theorem' used in restricted open-shell Hartree–Fock (ROHF), unrestricted H ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
John C
John is a common English name and surname: * John (given name) * John (surname) John may also refer to: New Testament Works * Gospel of John, a title often shortened to John * First Epistle of John, often shortened to 1 John * Second Epistle of John, often shortened to 2 John * Third Epistle of John, often shortened to 3 John People * John the Baptist (died c. AD 30), regarded as a prophet and the forerunner of Jesus Christ * John the Apostle (lived c. AD 30), one of the twelve apostles of Jesus * John the Evangelist, assigned author of the Fourth Gospel, once identified with the Apostle * John of Patmos, also known as John the Divine or John the Revelator, the author of the Book of Revelation, once identified with the Apostle * John the Presbyter, a figure either identified with or distinguished from the Apostle, the Evangelist and John of Patmos Other people with the given name Religious figures * John, father of Andrew the Apostle and Saint Peter * Pope Jo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Wavefunction
A wave function in quantum physics is a mathematical description of the quantum state of an isolated quantum system. The wave function is a complex-valued probability amplitude, and the probabilities for the possible results of measurements made on the system can be derived from it. The most common symbols for a wave function are the Greek letters and (lower-case and capital psi, respectively). The wave function is a function of the degrees of freedom corresponding to some maximal set of commuting observables. Once such a representation is chosen, the wave function can be derived from the quantum state. For a given system, the choice of which commuting degrees of freedom to use is not unique, and correspondingly the domain of the wave function is also not unique. For instance, it may be taken to be a function of all the position coordinates of the particles over position space, or the momenta of all the particles over momentum space; the two are related by a Fourier trans ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Configuration Interaction
Configuration interaction (CI) is a post-Hartree–Fock linear variational method for solving the nonrelativistic Schrödinger equation within the Born–Oppenheimer approximation for a quantum chemical multi-electron system. Mathematically, ''configuration'' simply describes the linear combination of Slater determinants used for the wave function. In terms of a specification of orbital occupation (for instance, (1s)2(2s)2(2p)1...), ''interaction'' means the mixing (interaction) of different electronic configurations (states). Due to the long CPU time and large memory required for CI calculations, the method is limited to relatively small systems. In contrast to the Hartree–Fock method, in order to account for electron correlation, CI uses a variational wave function that is a linear combination of configuration state functions (CSFs) built from spin orbitals (denoted by the superscript ''SO''), : \Psi = \sum_ c_ \Phi_^ = c_0\Phi_0^ + c_1\Phi_1^ + where Ψ is ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |